Dr. Rebika, R16797, Dr.Rachna Meel, Dr. Sharanya Devi, Dr. (Mrs ) RadhikaTandon
Rebika Dhiman, Rachna Meel, Saranya Devi, Radhika Tandon
Abstract
This is a prospective observational case series of the clinico-demographic profile of 52 eyes in 50 cases of clinically diagnosed OSSN presenting to a tertiary eye care centre in 1 year. The median age was 55 years with a male:female ratio of 4.5:1. Systemic predisposing conditions were xeroderma pigmentosa(2), post-kidney transplant immunosuppression(1), active pulmonary tuberculosis(1), HIV(1) and both HIV and TB(1). These patients had a younger median age of onset of 33 years. Bilateral involvement was seen in 2 cases that had some systemic predisposing condition. There were 12 recurrent cases. Impression cytology was positive in 75% cases. Clinical characteristics of OSSN were: commonest location – limbus; commonest growth pattern – nodular; commonest clinical presentation – gelatinous and papilliform. Intraocular extension was seen in 7 cases. Nodular metastasis was seen in 1 case. As per the AJCC staging,- there were 6 cases of T1, 38 cases of T3 and 8 cases of T4. Recurrence was seen in 2 cases. In conclusion, age>50 years, male gender and immunosuppression are risk factors for OSSN. Although associated with older age, a younger onset of OSSN and bilateral involvement is seen in patients with systemic predisposing conditions.
Introduction
Ocular surface squamous neoplasia (OSSN) is a term that encompasses a spectrum of epithelial squamous malignancies, ranging from dysplasia to invasive carcinoma.[1] OSSN is a common ocular surface malignancy with a reported incidence of 0.02 to 3.5 cases per 100 000 worldwide.[2] The incidence increases with decreasing latitude, being higher in countries located close to the equator.[3] The average age of presentation is usually in the sixth and seventh decades. Based on literature, two types of disease patterns of OSSN have been identified – one occurring in older males, and the other in younger immunocompromised individuals having equal incidence in both the sexes. [4, 5]
Riskfactors for OSSN include ultraviolet radiation, human immunodeficiency virus (HIV)infection, human papillomavirus infection, [6] heavy cigarette smoking, [7, 8] male gender and age. [9] Several studies have reported a dramatic increase in the incidence of OSSN following the outbreak of HIV.[10, 11]
The conventional treatment modality for OSSN is wide surgical excision with “no-touch” technique [12] often combined with cryotherapy and/or chemotherapy. There has been a change in treatment strategy during the last decade with the focus shifting from surgical to non-surgical management with chemotherapeutic agents [interferon-a 2b, fluorouracil (5-FU) and mitomycin C].[13]
In the wake of changing trends in the incidence and management options for OSSN, there is little knowledge about the clinic-demographic profile of these patients in the Indian scenario. So the purpose of this study was to evaluate the clinico-demographic profile and treatment outcome of OSSN patients presenting to a tertiary eye care centre.
Methodology
A prospective observational study was conducted on all patients clinically diagnosed with OSSN presenting to our centre from March 2015 to March 2016. Institutional ethical approval was obtained. Informed consent was obtained from all patients.
Demographic details and clinical history of the patients were taken and included the following – age at presentation, gender, laterality, occupation, socioeconomic status (using modified Kuppuswami scale[14]),duration of symptoms, risk factors (history of ocular trauma or surgery, sunlight exposure, associated ocular or systemic predisposing conditions and genetically predisposed state),and previous treatment history. Ahistory of prolonged sun exposure was determined by self-reportedhistory and by the occupation of the patient.Previous treatment history included details of types of previous interventions (surgery/chemotherapy), number of recurrences and available histopathological reports.Pterygium surgery was included in the previous treatment history as it had the potential to be a case of missed OSSN that was treated as pterygium.
Clinical details of the lesion were based on gross and slit-lamp examinations done by the same ocular oncologist. Clinical characteristics of tumor documented were location, extent, tissues involved, multiplicity, presence of feeder vessels, growth pattern (nodular/sessile),clinical type (papilliform, gelatinous, fungating, ulcerative), presence of leukoplakia, presence of pigmentation, and nodal or systemic
metastasis.Based on the number of quadrants of conjunctival involvement or the clock hours of limbal involvement, tumor was classified as small (≤1 quadrant of conjunctival involvement or ≤3 clock hours of limbal involvement), large (≤ 2 quadrants of conjunctival involvement or ≤6 clock hours of limbal involvement) and diffuse (>2 quadrants of conjunctival involvement or >6 clock hours of limbal involvement) (Figure 1).
Intraocular involvement was assessed by gonioscopy, direct fundus examination, ultrasound biomicroscopic examination (UBM) and ocular ultrasonography in cases with media opacity. UBM was used to measure the height of the lesion (Figure 2). Orbital imaging was done to rule out orbital extension of the tumor. The AJCC-TNMclassification of conjunctival carcinoma was used to stage all casesof OSSN (7th edition of AJCC [15]). Impression cytology and complete hemogram were done in all cases. HIV status was tested for patients less than 50 years of age.
Statistical analysis
Analysis was done following compilation of data using SPSS (version 11). Descriptive statistics were used for demographic characteristics and the data being presented as percentages, mean and standard deviation.
Results
Clinical and demographic details
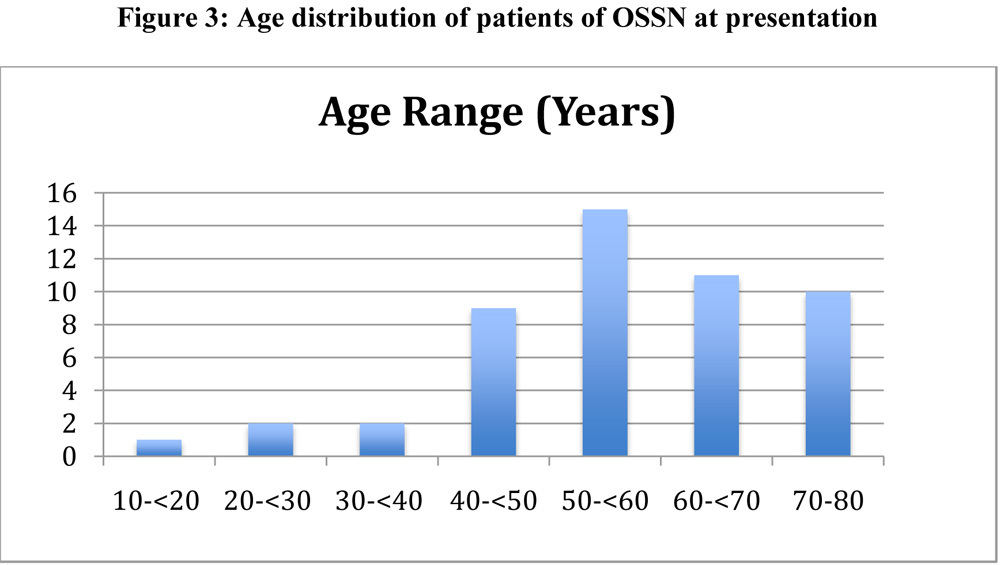
A total of 52 eyes of 50 patients were included in the study with the age range of 10 to 80 years. Median age of presentation was 55 years and the median age of onset of symptoms was 53 years.Figure 3 shows the age distribution of cases with the highest frequency noted in the age group of 50 to 60 years. Twenty eight percent of patients (n=14) were in the age group ≤50 years. There was a marked male preponderance with male:female ratio of 4.5:1. Seventy percent patients (n=35) were involved in indoor occupation and rest 30% (n=15) had an outdoor occupation. Amongst the risk factors, 34% (n=17) cases had prolonged sun exposure, 34% cases (n=17) had history of smoking and 14% (n=7) cases had previous history of ocular trauma or surgery unrelated to OSSN.
The right eye was involved in 25 patients and left eye in 23 patients. Bilateral involvement was seen in 2 cases. Previous treatment history was present in 48% (n=24) cases that included 4 cases of management with topical mitomycin C, 8 cases of pterygium excision and 12 cases of excision biopsy for OSSN. Of 12 cases with a history of excision biopsy, histopathological reports were available in 8 patients and revealed well-differentiated squamous cell carcinoma in 3 cases, moderately differentiated squamous cell carcinoma in 2 cases and poorly differentiated invasive squamous cell carcinoma in 3 cases. Regarding socioeconomic status, 80% of the patients belonged to the upper lower and lower class on assessment of socioeconomic status using modified Kuppuswami scale.
Associated ocular pathologies seen in the affected eye were cataract (n=10), ptosis (n=2), lagophthalmos (n=1) and symblepharon due to a previous surgery. Pterygium was noted in the fellow eye in 8 cases. Systemic associations were xeroderma pigmentosum (n=2), human immunodeficiency virus (HIV) (n=1), active pulmonary tuberculosis (TB) (n=1), both HIV and active TB (n=1), renal transplantation (n=1) and Berger’s disease (n=1). OSSN was the presenting feature in one of the HIV patients. Median age of presentation of six patients with underlying systemic predisposing conditions (HIV, xeroderma pigmentosum,renal transplant, active pulmonary tuberculosis) was 33 years (Range – 10-51 years).
Staging
Mean duration of symptoms was 1.03± 2.3 years.Most common presenting symptom was a mass or a swelling (78%) followed by redness (9%). The tumor characteristics of 52 eyes have been described in Table 1. Majority of tumors had a gelatinous appearance (57.7%) and a nodular growth pattern(57.7%). Limbus (50%) followed by the bulbar conjunctiva (48%) was the commonest tumor location. Based on the clinical characteristics, the AJCC grading of tumor included 6 eyes (11.5%) in T1, 38 eyes (73.1%) in T3 and 8 eyes (15.4%) in T4 stage. Intraocular involvement was seen in 7 cases on UBM, of which 2 cases had an associated orbital involvement (4 cases-T3, 2 cases-T4). Leukoplakia was seen in 46% cases and pigmentation in 27% cases. One case had a regional lymph node involvement confirmed by fine needle aspiration cytology. None of the cases had distant metastasis at presentation. Clinical characteristics of immunocompromised patients have been described in Table 2.
Investigations
Impression cytology and ultrasound biomicroscopic (UBM) examination were done for all except one paediatric patient. Impression cytology showed dysplasia in 75% (n=39/51) cases. Mean tumor height on UBM was 3.5±0.7mm.Intraocular involvement in 7 cases was evident as scleral infiltration and ciliary body thickening underlying the tumor. Orbital imaging (contrast enhanced computed tomography or magnetic resonance imaging) was ordered in all cases with forniceal involvement and an orbital extension was noted in 8 cases.
Treatment and follow-up
Treatment advised included conservative management (with mitomycin C or Interferon alfa -2b) in 32 cases, wide surgical excision with cryotherapy in 5 cases, enucleation with amniotic membrane transplantation with adjunctive treatment [topical ± external beam radiotherapy(EBRT)]in 4 cases, and exenteration in 3 cases. Six cases were lost to follow-up after an initial visit. Clinical outcome of 45 cases were assessed on follow-up. Mean duration of follow-up was 8.2 ± 4.6 months. Recurrence was seen in 2 cases. One patient had an orbital recurrence 5 months after the surgery, as he defaulted and did not get EBRT post-operatively.Another patient came with a lymph node metastasis one month after completion of treatment (enucleation+EBRT). None of the patients developed systemic metastasis or died of disease on follow-up.
Discussion
OSSN is a common lesion in India. In a recent study from India, extensive OSSN was found to be the commonest malignancy resulting in exenteration. [16]The pathogenesis is multifactorial. Risk factors for OSSN in our study were ultraviolet radiation, cigarette smoking, old age, male gender, ocular trauma/surgery and immunosuppression with similar findings reported in literature.[6-9] In our study, highest occurrence of OSSN was seen in the older age group of 5th to 6th decade with a marked male preponderance (82%). Male gender is associated with greater outdoor activity and thus greater sun exposure, which is an established risk factor for OSSN. Similar findings have been reported in most other studies from Indian and other subcontinents, [17, 18, 19, 20] excepting the studies from Africa [9, 10, 21]where a younger age of onset and female predominance have been associated with HIV prevalence. Younger mean ages in HIV positive individuals are also being recently reported from India.[22, 23] This observation was also seen in our study where the mean age of presentation of immunocompromised patients was much lower (33 years). Bilateral cases in our series had systemic predisposing conditions that included xeroderma pigmentosum (n=1) and active pulmonary TB (n=1).
Chauhan et al. [19] in their series of 64 cases of histopathologically proven OSSN reported a nodular growth pattern in 65% cases, gelatinous appearance in 59% cases and limbus involvement in 55% cases. Our study reports similar observation with majority of tumors being nodular (58%), gelatinous (58%) and having limbal involvement (50%). In a retrospective cross sectional study, Dandala et al. [22] reported that a nodule at the limbus as the commonest OSSN presentation. Papilliform sessile lesions were seen in 63.6% cases in a retrospective study done in Korean eyes by Kim et al. [17]
In our cohort, according to AJCC classification, majority of tumors (75%) belonged to T3 stage. There were no lesions in T2 stage. With limbus being the commonest site of origin, the lesions that are >5mm in size mostly are large enough to involve the adjacent cornea. Hence the percentage of tumours in T2 stage is expected to be less. The representation of T2 stage OSSN in studies by Shields et al. [24] and Shah et al. [25] were 7% and 0% respectively. Hence, we support the recommendations of Sheilds et al. to modify the existing classification scheme for OSSN. [25]Also, there should be a separate categorization of tumours with intraocular involvement as even smaller tumours may have an intraocular extension that may be missed clinically and they require a more aggressive management. We propose that OSSN that are >5mm in diameter and with <2mm of radial corneal involvement should be graded as T2.
We devised our clinical classification scheme (small, large and diffuse) based on the number of quadrants of conjunctival involvement or the clock hours of limbal involvement. Fifty percent of the tumors were large. We found that 75% (6 out of 8) tumors with orbital extension were large and 67%(4 out of 6) tumors with intraocular extension were diffuse. The mean duration of symptoms of symptoms was longer in these cases (1.89 ± 1.2 years). Mean tumor height of OSSN on UBM with orbital and intraocular involvement were 5.9mm and 4.3mm respectively, that was more than the overall mean height (3.5mm). Hence, large and thick tumors are more aggressive in nature, as was also seen by Kao et al. in a retrospective study of 612 OSSN cases,[20]and have a longer duration of pathology.
Immunosuppression has been speculated to play a role in the pathogenesis of OSSN. Eight percent (n=4) of our patients were immunocompromised with a younger mean age of presentation. OSSN was a presenting feature in one of the patients who was subsequently diagnosed as a case of HIV. Sptizer et al.[21] conducted a study to evaluate the prevalence of undiagnosed and asymptomatic HIV infection in patients of OSSN. Seventy nine percent (30/38) patients were HIV positive. None of the patients had previous HIV testing or was aware of having symptoms of HIV. Seventy percent of the HIV-positive patients had no other symptoms suggestive of HIV infection or any other disease. Porges et al.[4] did a study to evaluate the prevalence of HIV seropositivity among patients with OSSN in a group of African black patients. Conjunctival malignancy was the first presenting sign for HIV in 50% of their patients. Although the HIV positivity rate is expected to be high in studies from African subcontinent, conjunctival tumor may be the primary and only apparent manifestation of HIV especially in young onset OSSN. A study from South India reported HIV positivity in 21% OSSN patients. [26] The tumors are found to be more invasive in HIV positive cases. [23] Therefore, an HIV test should be performed in all cases of OSSN who are <50 years of age.
Four percent (n=2) of our cases had xeroderma pigemntosa. Gupta N et al.[27] in their retrospective study on 7 patients of OSSN with xeroderma pigmentosum (XP) concluded that OSSN tends to be more aggressive and occurs at a younger age in such patients. Table 2 shows that majority of the cases with a systemic predisposition were high grade tumors (T3, T4), were larger in size (large/diffuse) and, with invasion of adjacent structures like lid, sclera and globe. Bilaterality was also seen in these cases.
Metastasis is rare in OSSN with a reported incidence of 0-16% in cases of squamous cell carcinoma. [28] Systemic metastasis was not seen in our patients and nodal involvement was seen in one case. Another patient subsequently developed a nodal recurrence on follow-up. Four percent cases (n=2) showed recurrence during a mean follow up of around 8 months in our study. Higher recurrence rate of 10% to 36% during a follow-up of 1 to 2.5 years have been reported in several other studies.[17, 19, 29, 30] Lower recurrence rate in our study could be due to several reasons. Our study included cases that were treated with topical chemotherapeutic agents. They have the advantage of treating the entire ocular surface with less dependence on defining tumor margins. Hence, a better outcome and lower recurrence rate is expected. Also, we have a shorter duration of follow-up.
There are a few strengths of our study. First, we utilized prospective data collection. Second, we have included cases with complete range of management options for OSSN including those who were treated with chemotherapeutic agents like mitomycin C and interferon alpha2b. Hence, our study gives the clinico-demographic profile and outcome of OSSN in the current scenario. The limitation of our study is a short follow-up of cases. Longer follow-up is needed to assess the recurrence and long term outcome of OSSN.
In conclusion, OSSN is a common ocular surface tumor occurring in older age. Younger age of onset and bilaterality may be seen in patients with underlying systemic predisposing conditions. Larger and thicker tumors are aggressive, with propensity to involve the orbit or the globe.
References
- Lee GA, Hirst LW. Ocular surface squamous neoplasia. Surv Ophthalmol. 1995;39:429–450.
- Yang J, Foster CS. Squamous cell carcinoma of the conjunctiva. IntOphthalmol Clin. 1997;37(4):73–85.
- Gichuhi S, Sagoo, MS, Weiss HA, Burton, MJ Epidemiology of ocular surface squamous neoplasia in Africa. Trop Med Int Health 2013;18:1424-1443.
- Porges Y, Groisman GM. Prevalence of HIV with conjunctivalsquamous cell neoplasia in an African provincial hospital.Cornea 2003;22:1–4.
- Balint GA. Situation analysis of HIV/AIDS in sub-SaharanAfrica. East Afr Med J 1998;75:684–686.
- Gichuhi S, Ohnuma S, Sagoo MS, Burton MJ. Pathophysiology ofocular surface squamous neoplasia. Exp Eye Res 2014; 129: 172–182.
- Napora C, Cohen EJ, Genvert GI, et al. Factors associated with conjunctival intraepithelial neoplasia: a case control study. Ophthalmic Surg. 1990; 21:27–30.
- Lee GA, Hirst LW. Retrospective study of ocular surface squamous neoplasia. Aust N Z JOphthalmol. 1997; 25:269–276.
- Gichuhi S, Macharia E, Kabiru J, Zindamoyen AM, Rono H, Ollando E, Wanyonyi L, Wachira J, Munene R, Onyuma T, Sagoo MS, Weiss HA,Burton MJ.Clinical Presentation of Ocular Surface Squamous Neoplasia in Kenya.JAMA Ophthalmol.2015 Nov;133(11):1305-13.
- Ateenyi-Agaba C. Conjunctival squamous-cell carcinomaassociated with HIV infection in Kampala, Uganda. Lancet1995 Mar 18;345(8951):695–696.
- Guech-Ongey M, Engels EA, Goedert JJ, Biggar RJ & MbulaiteyeSM. Elevated risk for squamous cell carcinoma of the conjunctiva among adults with AIDS in the United States. Int J Cancer2008 Jun 1;122(11):2590–2593.
- Shields JA, Shields CL, De Potter P. Surgical management of conjunctival tumors. The 1994 Lynn B. McMahan Lecture. Arch Ophthalmol 1997; 115:808–815.
- Nanji AA, Moon CS, Galor A, Sein J, Oellers P, Karp CL. Surgical versus medicaltreatment of ocular surface squamous neoplasia: a comparisonof recurrences and complications. Ophthalmology 2014;121:994–1000.
- Mishra D, Singh HP. Kuppuswamy’s Socioeconomic Status Scale: a revision[letter]. Indian J Pediatr. 2003;70(3):273–274.
- Edge SB, Byrd DR, Compton CA, et al. AJCC Ophthalmic Oncology Task Force. Carcinoma of the conjunctiva:AJCC Cancer StagingManual. 7th ed. New York, NY: Springer; 2010:531–537.
- Ali MJ, Pujari A, Dave TV, Kaliki S, Naik MN.Clinicopathological profile of orbital exenteration: 14 years of experience from a tertiary eye care center in South India.Int Ophthalmol.2016 Apr;36(2):253-8.
- Kim BH, Kim MK, Wee WR, Oh JY.Clinical and pathological characteristics of ocular surface squamous neoplasia in an Asian population.Graefes Arch Clin Exp Ophthalmol.2013;251(11):2569-73.
- Shields CL, Demirci H, Karatza E, Shields JA. Clinical survey of 1643 melanocytic and nonmelanocytic conjunctival tumors. Ophthalmology 2004;111(9):1747-54.
- Chauhan S, Sen S, Sharma A, Tandon R, Kashyap S, Pushker N, Vanathi M, Sharma N. American Joint Committee on Cancer Staging and clinicopathological high-risk predictors of ocular surface squamous neoplasia: a study from a tertiary eye center in India.Arch Pathol Lab Med.2014 Nov;138(11):1488-94.
- Kao AA1, Galor A, Karp CL, Abdelaziz A, Feuer WJ, Dubovy SR.Clinicopathologic correlation of ocular surface squamous neoplasms at Bascom Palmer Eye Institute: 2001 to 2010.2012 Sep;119(9):1773-6.
- Spitzer MS, Batumba NH, Chirambo T, Bartz-Schmidt KU, Kayange P, Kalua K, Szurman P.Ocular surface squamous neoplasia as the first apparent manifestation of HIV infection in Malawi.Clin Exp Ophthalmol. 2008;36(5):422-5.
- Dandala PP, Malladi P, Kavitha.Ocular Surface Squamous Neoplasia (OSSN): A Retrospective Study.J Clin Diagn Res.2015 Nov;9(11):NC10-3.
- Kamal S, Kaliki S, Mishra DK, Batra J, Naik MN.Ocular Surface Squamous Neoplasia in 200 Patients: A Case-Control Study of Immunosuppression Resulting from Human Immunodeficiency Virus versus Immunocompetency. 2015 Aug;122(8):1688-94.
- Shields CL, Kaliki S, Kim HJ, et al. Interferon for ocularsurface squamous neoplasia in 81 cases: outcomes based on the American Joint Committee on Cancer classification. Cornea 2013;32:248–56.
- Shah SU, Kaliki S, Kim HJ, et al. Topical interferon alpha-2b for management of ocular surface squamous neoplasia in 23 cases: outcomes based on American Joint Committee on Cancer classification. Arch Ophthalmol. 2012;130:159–164.
- Pradeep TG, Gangasagara SB, Subbaramaiah GB, et al.Prevalence of undiagnosed HIV infection in patients with ocular surface squamous neoplasia in a tertiary center in Karnataka, South India. Cornea 2012 Nov; 31(11):1282-4.
- Gupta N, Sachdev R, Tandon R. Ocular surface squamous neoplasia in xeroderma pigmentosum: clinical spectrum and outcome. Graefes Arch Clin Exp Ophthalmol 2011;249(8):1217-21.
- McKelvie PA, Daniell M, McNab A, et al. Squamous cell carcinoma of theconjunctiva: a series of 26 cases. Br J Ophthalmol. 2002;86:168–173.
- Ramberg I, Heegaard S, Prause JU, Sjö NC, Toft PB. Squamous cell dysplasia and carcinoma of the conjunctiva. A nationwide retrospective study of Danish patients.Acta Ophthalmol.2015 Nov;93(7):663-6.
- Maudgil A, Patel T, Rundle P, Rennie IG, Mudhar HS. Ocular surface squamous neoplasia: analysis of 78 cases from a UK ocular oncology centre.Br J Ophthalmol.2013 Dec;97(12):1520-4.
Table 1- Summary of clinical features of 52 eyes of OSSN
| Clinical characteristics | Number of cases (%)
N=52 eyes |
| Clinical appearance
– Gelatinous – Pappilliform – Ulcerative – Fungating |
30 (57.7) 16 (30.8) 5 (9.6) 1 (1.9) |
| Growth pattern
– Nodular – Sessile |
30 (57.7) 22 (42.3) |
| Multiplicity
– Single – Multiple |
47 (90.4) 5 (9.6) |
| Structures involved
– Limbus – Bulbar conjunctiva – Tarsal conjunctiva – Fornices – Cornea – Lid – Intraocular |
50 (96.1) 48 (92.3) 8 (15.4) 12 (23.1) 49 (94.2) 5 (9.6) 7 (13.5) |
| Feeder vessels
– Present – Absent |
52 (100) 0 (0) |
| Leukoplakia
– Present – Absent |
24 (46.1) 27 (51.9) |
| Pigmentation
– Present – Absent |
14 (26.9) 38 (73.1) |
| Impression cytology
– Positive – Negative – Not Done (Pediatric patient) |
39 (75) 12 (23.1) 1(1.9) |
| Based on the quadrant involvement
– Small – Large – Diffuse |
16 (30.8) 26 (50) 10 (19.2)
|
| AJCC staging
– T1 – T2 – T3 – T4 |
6 (11.5) 0 (0) 38 (73.1) 8 (15.4) |
| Fresh/Recurrent cases
– Fresh – Recurrent |
40 (76.9) 12 (23.1) |
Table 2: Clinical characteristics of patients of OSSN with underlying systemic predisposing condition
| 1 | 2 | 3 | 4 | 5 | 6 | |
| Age | 33 | 27 | 10 | 24 | 40 | 51 |
| Gender | Male | Male | Female | Male | Male | Male |
| Laterality | OD | Bilateral | OS | OS | OS | Bilateral |
| Duration of symptoms | 6 months | 8 months | 12 months | 18 months | 12 months | 10 months |
| Underlying systemic condition | Post-renal transplant | XP | XP | HIV | HIV, TB | TB |
| Previous treatment history | – | + | – | – | – | + |
| Clinical appearance | Gelatinous | Papilliform | Gelatinous | Gelatinous | Papilliform | Ulcerative |
| Leucoplakia | – | + | + | – | – | – |
| Pigmentation | – | + | – | + | – | _ |
| Growth pattern | Sessile | Sessile | Nodular | Nodular | Nodular | Nodular |
| Structures involved | Cornea, Limbus | Cornea, bulbar & palbebral conjunctiva, limbus, lid | Cornea, limbus, bulbar conjunctiva | limbus, bulbar conjunctiva | Cornea, limbus, bulbar conjunctiva, intraocular | Corneal melt, limbal, bulbar conjunctiva, scleral melt, intraocular |
| Our classification | Small | Large | Small | Small | Diffuse | Diffuse |
| AJCC stage | T3 | T3 | T3 | T1 | T3 | T4 |
Figure 1:Clinical classification based on the number of quadrants of conjunctival involvement or the clock hours of limbal involvement as (A) Small, (B) Large, and (C) Diffuse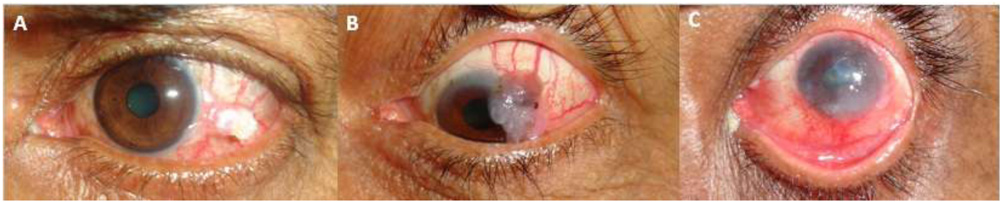 Figure 2: Measurement of the height of the lesion using ultrasound biomicroscopy
Figure 2: Measurement of the height of the lesion using ultrasound biomicroscopy
Figure 2: Measurement of the height of the lesion using ultrasound biomicroscopy
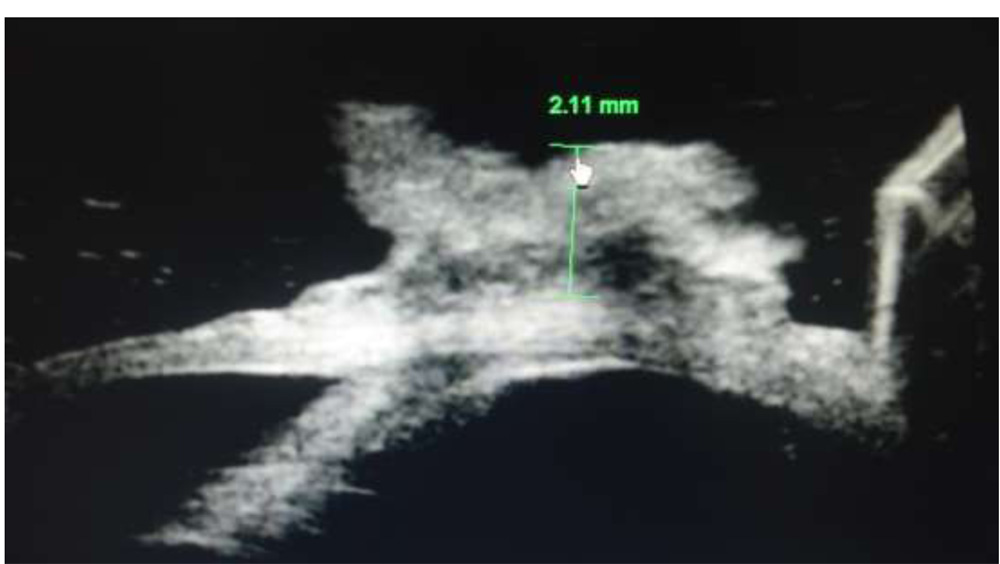 Figure 3: Age distribution of patients of OSSN at presentation
Figure 3: Age distribution of patients of OSSN at presentation