Dr. Rutul R Patel, P18478, Dr. Giridhar
Anantharaman, Dr. Mahesh G, Dr. Bindu K
Appukuttan
ABSTRACT CODE NO: FP733
CHIEF AND PRESENTING AUTHOR:DR RUTUL PATEL
CO- AUTHORS: DR. A GIRIDHAR, DR. MAHESH GOPALAKRISHNAN ,DR BINDU K A
COMMUNICATION ADDRESS: GIRIDHAR EYE INSTITUTE, PONNETH TEMPLE ROAD, KADAVANTHRA, KOCHI – 682020.
Mobile Number: 9624722995
ABSTRACT–
Purpose – To evaluate optical coherence tomography (OCT),fundus fluorescein angiography(FFA) and indocyanine green angiography(ICG) features of extra large pigment epithelial detachment(X large PED) and assess their response to treatment.
Methods– 50 eyes of 50 patients with X large PED(>350µ vertical height)were evaluated for FFA,ICG,OCT features and response to treatment.
Results– Mean age was 70 years.Mean height of PED was 797 µ (350-700µ-46%,700-1000µ-32%,>1000µ-22%).IPCV,wet AMD and CSCR were the main etiological diagnosis(80%,16% and 4% eyes respectively).Photodynamic therapy(PDT),focal thermal laser, and intravitreal VEGF were modalities of treatment.Reduction in PED height following PDT or focal laser was significant(p=0.04) though visual acuity change was not statistically significant.
Conclusion -IPCV was the major etiology in X large PED and PDT or focal laser resulted in significant reduction in PED height compared to intravitreal anti-VEGF.
INTRODUCTION
Retinal pigment epithelial detachment is a pathological condition in which retinal pigment epithelium is separated from underlying bruch’s membrane. This separation can be due to presence of either serous fluid, blood or neovascular membrane in Sub-RPE space (space between RPE and bruch’s membrane).1
Retinal pigment epithelium detachments (PEDs) have been associated most commonly with age-related macular degeneration (AMD), central serous chorioretinopathy (CSCR), and polypoidalchoroidalvasculopathy (PCV )2,3. Clinically PED appears as dome shaped elevation of RPE. It may be associated with serous retinal detachment, retinal edema or hard exudates according to the underlying mechanism of PED.
PED was initially classified on basis of fundus fluorescence angiography (FFA). Serous PED shows pooling of dye in area of RPE detachment while fibrovascular PED shows typical stippled type of hyper fluorescence on fundusfluorescence angiography( FFA).
After advent of Spectral domain optical coherence tomography (SD-OCT) , PED was classified according to characteristics on OCT in 4 categories namely serous, fibrovascular , haemorrhagic and drusenoid PED2. Serous PED shows dome shaped elevation of RPE with sub RPEoptically clear space. Fibrovascular PED has sub RPE hyperreflective material. HaemorrhagicPED showssub RPE shadowing effect. Drusenoid PED will show dome shaped elevation of PED with hyperreflectivedrusenoid material below RPE.
Presence of PED and its type are important factors for diagnosis and management. Change in morphology of PED is one of the important predictive factors for progression of the disease.
In a subgroup analysis in Harbor study Dr Eichenbaum and colleaguescategorized pigment epithelial detachments by vertical height as small (35-164μm), medium (164.5-233μm), large (233.25- 351μm), or extralarge (352- 1395.5μm) on SD-OCT.5Management of large and extra large PED is controversial due to variable response to treatment and variable natural course.It may remain stable or it can increase in size or collapse, or can convert to RPE atrophy. Height of PED is also an important risk factor RPE rip following intravitrealinjection.Jordymones et al has reported a single case of large PED with RAP lesion where they have injected half dose of ranibizumab twice in a month to prevent RPE rip.6
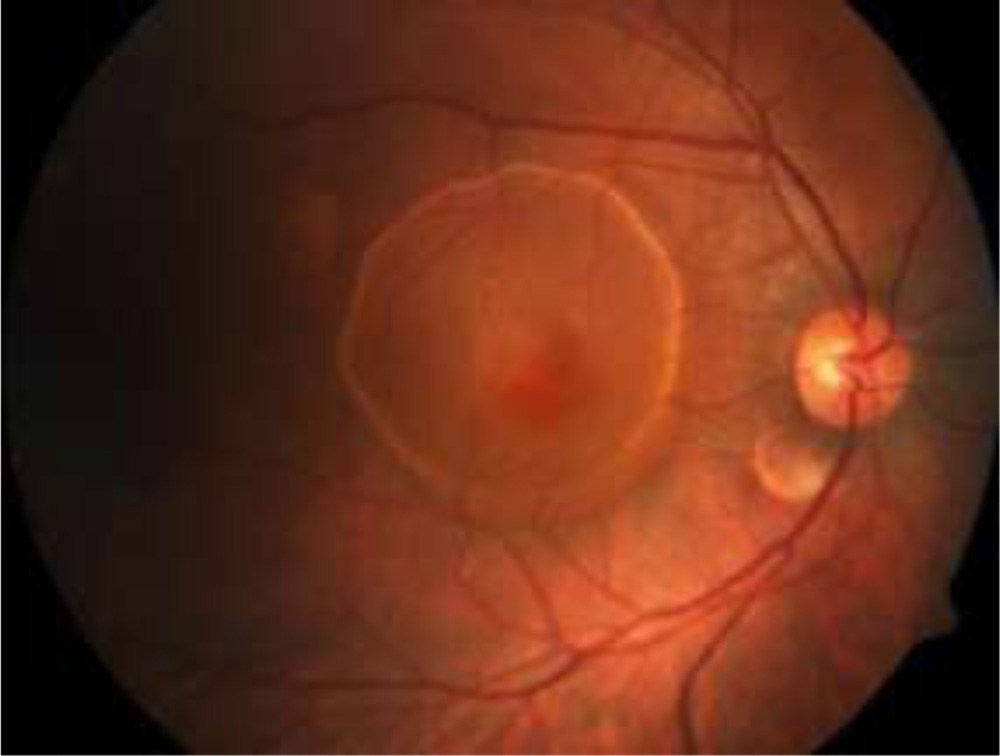 Figure 1 –Extra large PED
Figure 1 –Extra large PED
So , purpose of this study is to evaluate features of extra large PED with multimodal imaging like SD-OCT , FFA and ICG and evaluate their response to various modalities of treatment.
MATERIALS & METHODS
This is a retrospective study of 50 eyes of 50 patients with extra large PED on SD-OCT examination. Parametres recorded were demographic details , Best corrected visual acuity ( BCVA ), Spectral domain optical coherence tomography ( SD-OCT ) , fundus fluorescence angiography ( FFA) , and indocyanine green angiography ( ICG ). Duration of follow up was ranging from 2 months to 18 months with mean duration of 7 months.
BCVA was evaluated at baseline and at last follow up with help of snellen’s visual acuity chart and later converted to LogMAR for calculation. Characteristics of PED observed in SD- OCT were location , maximal vertical height , type of PED and presence of notch in PED. PED height was measured manually with help of caliper provided inbuilt in Heidelberg software by measuring distance between RPE and Bruch’s membrane. And as per the critreriasmentioned by Dr Eichenbaum and colleagues in subgroup analysis of Harbor study PED with maximum vertical height of > 352 micron were considered as Extra large PED.5
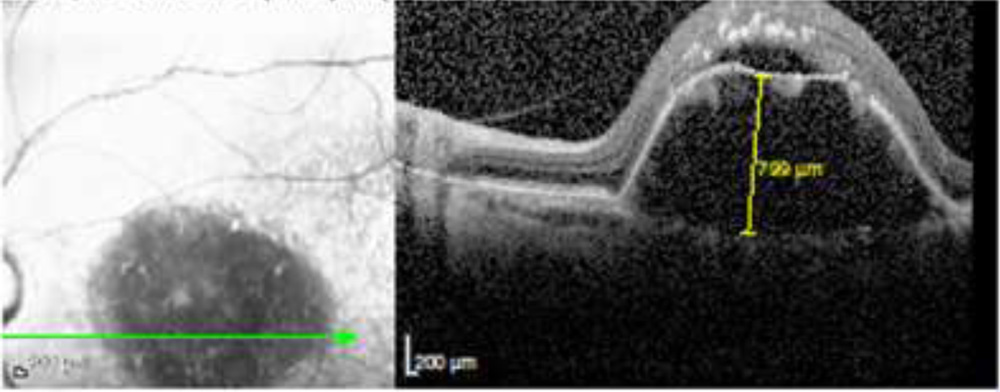 Figure 2 – Measurement of maximum vertical height of PED on SD-OCT
Figure 2 – Measurement of maximum vertical height of PED on SD-OCT
PED location was divided in two groups based on involvement of fovea. Other SD-OCT features observed were presence of subretinalfluid ,intraretinalhyperreflective dots , intraretinal edema , subfovealchoroidal thickness and presence of double layer sign ( DLS). Presence of RPE rip was assessed at baseline and throughout the treatment
FFA and ICG were done at baseline before starting the treatment. ICG were observed for any polypoidal changes. Final diagnosis was made based on all multimodal imaging features.Out of 50 patients, 8 patients were lost to follow up after diagnosis so they were excluded from analysis for measuring response to treatment. Remaining 42 patientsreceived treatment at our hospital and were followed up regularly.
Treatment modalities used wereintravitreal anti – VEGF(monotherapy) , focal green laser, and photodynamic therapy ( PDT)with Intravitreal VEGF(combination therapy) . Some patients were not willing for treatment , so they were observed without treatment.
Response to treatment wasassessed based on change in vertical height of PED after treatment.PED height was measured at each visit and change in PED height was calculated.
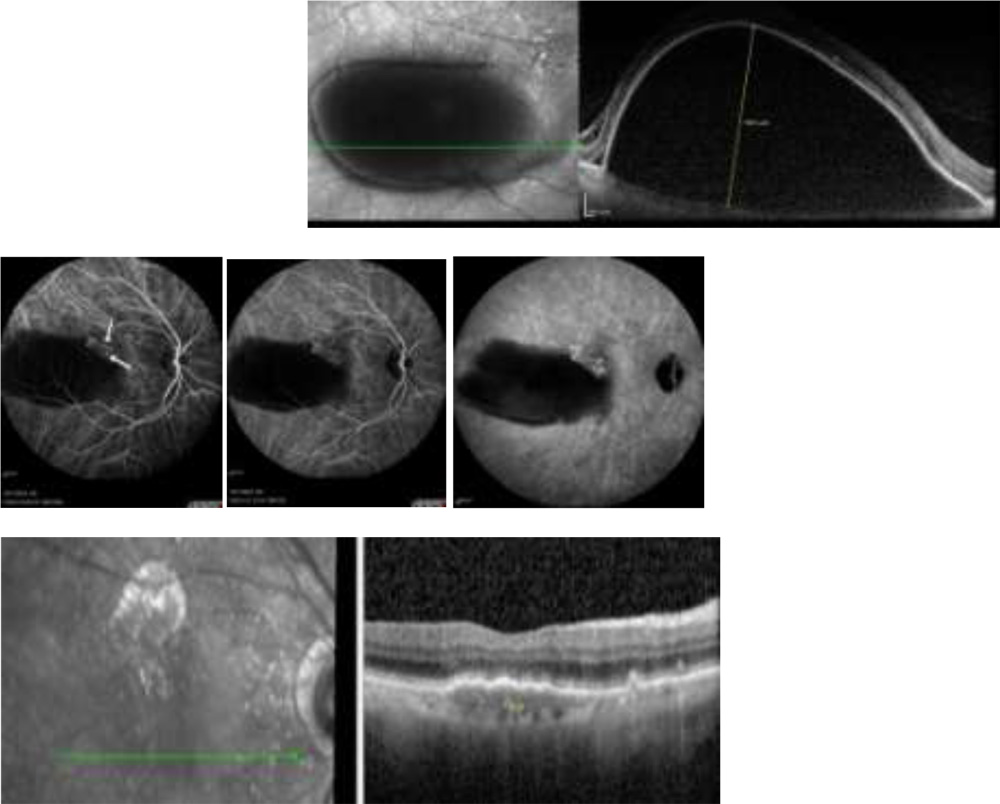 Figure 3 -A case of 64 year female with PED height of 1650 microns and bunch of polyps at notch of PED in ICG showed complete resolution of PED after treatment with focal green laser to a feeder vessel
Figure 3 -A case of 64 year female with PED height of 1650 microns and bunch of polyps at notch of PED in ICG showed complete resolution of PED after treatment with focal green laser to a feeder vessel
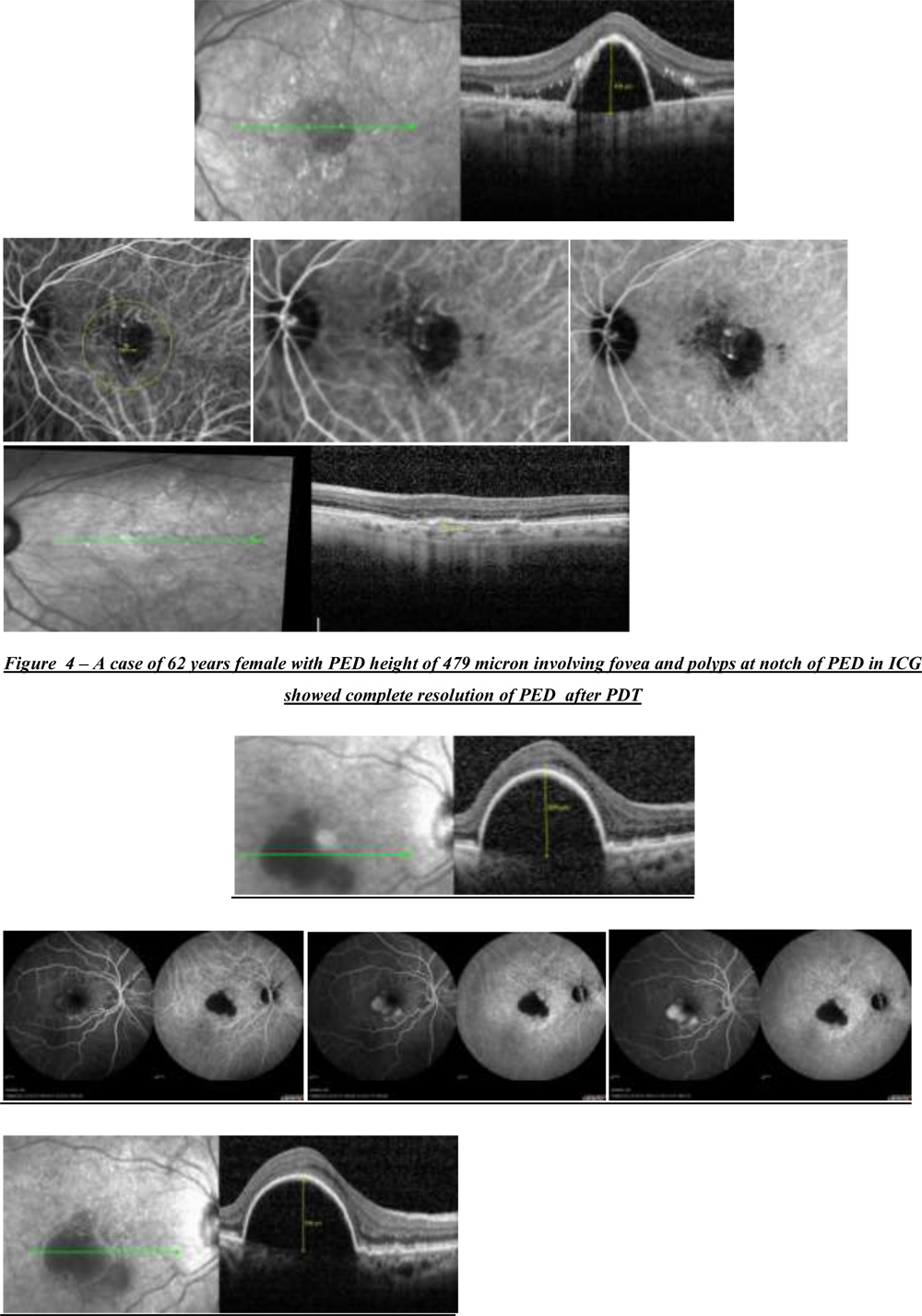 Figure 5 – A case of 75 years female with PED height of 625 microns showing features of occult CNVM on FFA like stippled Hyper Fluorescence with pooling of dye in PED which didn’t opt for any treatment and was observed
Figure 5 – A case of 75 years female with PED height of 625 microns showing features of occult CNVM on FFA like stippled Hyper Fluorescence with pooling of dye in PED which didn’t opt for any treatment and was observed
RESULTS
Extra largePED was evaluated in 50 eyes of 50 patients. Out of all 50 patients , 26 were female ( 52 %) and 24 male ( 48 % ).(Table1.2) Patients were in age group of 43 – 87 years with Mean age of 64.18 ± 11.7 years (Table1.1). Mean BCVA at baseline was 0.55± 0.44 LogMAR which improved to 0.48± 0.4 at the final follow up after treatment which was statistically significant ( p = 0.02)
Table 1.1: Distribution of cases based on Age
| Age Group (Years) | Frequency | Percent |
| <60 | 21 | 42.0 |
| 60-70 | 12 | 24.0 |
| > 70 | 17 | 34.0 |
| Total | 50 | 100.0 |
Table 1.2: Distribution of cases based on Gender
| Gender | Frequency | Percent |
| Female | 26 | 52.0 |
| Male | 24 | 48.0 |
| Total | 50 | 100.0 |
Average vertical height of PED was 797 ± 292 micronsat baseline. Out of total 50 eyes 23 eyes (46%) had PED height in range of 350-700 microns , 16 eyes ( 32%) in range of 700-1000 microns and 11 eyes ( 22%) with > 1000 microns.(Table1.4)According to OCT based classification 16 eyes ( 32%) were having tall peaked PED (TPP),11 eyes ( 22 % ) with dome shaped PED ,10 eyes (20%) with thumb shaped PED (TLP), 7 eyes ( 14%) with haemorrhagic PED , and 6 eyes ( 12 %) were having irregular PED.(Table1.3) PED involving fovea was seen in 33 eyes ( 66%) (Table1.5).Presence of subretinalfluid ( SRF ) was very consistent finding which was present in 45 eyes ( 90 %).(Table1.6)Another characteristic feature was absence of intraretinaledema.Intraretinal edema on OCT was seen only in 10 eyes ( 20 % ). Double layer sign ( DLS ) which is described as two distinct and parallel hyper-reflective lines of RPE and bruch’s membrane was present in 29 eyes ( 58%). Other significant OCT features like presence of intraretinal hyper reflective dots on OCT was seen in 40 eyes ( 80 % ) and characteristic notch in PED in 36 eyes ( 72 %) (Table1.7 ,1.8)
Choroidal thickness measured by enhanced depth imaging showed subfoveal thickness at baseline of < 200 microns in 16 eyes ( 32 %) , 200-350 microns in 24 eyes ( 48 % ) and > 350 microns in 10 eyes ( 20 % ).(Table1.9)
Table 1.3: Distribution of cases based on Type of PED
| Type of PED | Frequency | Percent |
| Tall peaked PED | 16 | 32.0 |
| Dome shaped PED | 11 | 22.0 |
| Thumb shaped PED | 10 | 20.0 |
| Haemorrhagic PED | 7 | 14.0 |
| Irregular PED | 6 | 12.0 |
| Total | 50 | 100.0 |
Table 1.4: Distribution of cases based on PED Height
| PED Height (µ) | Frequency | Percent |
| 350-700 | 23 | 46.0 |
| 700-1000 | 16 | 32.0 |
| >1000 | 11 | 22.0 |
| Total | 50 | 100.0 |
Table 1.5: Distribution of cases based on PED Involving Fovea
| PED Involving Fovea | Frequency | Percent |
| Yes | 33 | 66.0 |
| No | 17 | 34.0 |
| Total | 50 | 100.0 |
Table 1.6: Distribution of cases based on presence of subretinal fluid
| SUBRETINAL FLUID | Frequency | Percent |
| Yes | 45 | 90.0 |
| No | 5 | 10.0 |
| Total | 50 | 100.0 |
Table 1.7: Distribution of cases based on presence of Intra retinal Hyper Reflective Dots
| Intraretinal Hyper Reflective Dots | Frequency | Percent |
| Yes | 40 | 80.0 |
| No | 10 | 20.0 |
| Total | 50 | 100.0 |
Table 1.8: Distribution of cases based on the Notch in PED
| Notch in PED | Frequency | Percent |
| Yes | 36 | 72.0 |
| No | 14 | 28.0 |
| Total | 50 | 100.0 |
Table 1.9: Distribution of cases based on Enhanced Depth Imaging
| Enhanced Depth Imaging (µ) | Frequency | Percent |
| <200 | 16 | 32.0 |
| 200-350 | 24 | 48.0 |
| >350 | 10 | 20.0 |
| Total | 50 | 100.0 |
FFA and ICG were done at baseline in all patients which was showing polypoidal changes ( single/multiple polyps+- BVN) in 40 eyes ( 80 % ) , occult choroidal neovascularization features in 4 eyes ( 8 %) , focal RPE leak with pachychoroid features in 2 eyes ( 4%) and nonspecific features in remaining 4 eyes ( 8%).(Table1.10) Another characteristic feature notedin ICG was presence of multiple polyps in notch of X large PED which was present in 36 eyes ( 72 %)(Table1.11)
Based on multimodal imaging 40 eyes ( 80%) were diagnosed as IPCV , 8 eyes ( 16 %) as wet ARMD and 2 eyes ( 4%) as CSCR.(Table1.12) After diagnosis 8 eyes were lost to follow up .
Table 1.10: Distribution of cases based on FFA/ICG features
| FFA or ICG | Frequency | Percent |
| POLYP/BVN | 37 | 74.0 |
| CNVM/OCCULT | 4 | 8.0 |
| OTHERS | 9 | 18.0 |
| Total | 50 | 100.0 |
Table 1.11: Distribution of cases based on presence of polyp at notch in PED in ICG
| POLYP AT NOTCH IN PED IN ICG | Frequency | Percent |
| Present | 31 | 62.0 |
| Absent | 19 | 38.0 |
| Total | 50 | 100.0 |
Table 1.12: Distribution of cases based on the Diagnosis
| Diagnosis | Frequency | Percent |
| CSCR | 2 | 4.0 |
| IPCV | 40 | 80.0 |
| WET AMD | 8 | 16.0 |
| Total | 50 | 100.0 |
Treatment modalities used for remaining 42 eyes were focal green laser , photodynamic therapy ( PDT) with anti-VEGF(combination therapy) , intravitreal anti – VEGF monotherapyand observation . 5 eyes ( 11 %) were treated with focal laser , 11 eyes (38%) with PDT , 23 eyes ( 54 %) with I/V anti- VEGF .Remaining 3 eyes were observed without treatment as patients were not willing for treatment.Response to treatment was measured as change in vertical height of PED on OCT on last follow up. Mean decrease in PED height was 577 microns for focal laser , 467 microns for PDT and 353 microns for I/V anti VEGF and 15 microns for eyes which were only observed.(Table 2.1)So , decrease in PED height was maximum with focal laser and PDT was maximum compared to eyes which were observed.The difference was statistically significant with p < 0.05.(Table 2.2)
Table 2.1: Determination of relation of PED Difference and Mode of Treatment
| PED Difference | Mode of Treatment | P Value | |||
| Focal | PDT | I/V | Others | ||
| Mean | -577.0 | -467.40 | -353.08 | -15.66 |
0.04** |
| SD | 677.46 | 194.47 | 332.01 | 21.07 | |
Kruskal Wallis test was used to find the P value
** indicates a significant association (p-value <0.05)
Table 2.2: Pairwise comparison of the mode of treatment using Bonferroni method
| Mode of treatment | Mean Difference (PED) | P value |
| 1 Vs 2 | -109.60 | 0.71 |
| 1 Vs 3 | -223.91 | 0.74 |
| 1 Vs 4 | -561.33 | 0.05 |
| 2 Vs 3 | -114.31 | 0.17 |
| 2 Vs 4 | -451.73 | 0.01 |
| 3 Vs 4 | -337.42 | 0.01 |
Bonferroni method was used to find the P value
The difference between 2 Vs 4 is more significant (451.73) as compared to the other possible combinations.
Table 3: Assessment of BCVA – Baseline and Follow up
| Baseline | Follow up |
P value |
|||
|
Mean |
SD |
Mean |
SD |
||
|
BCVA |
0.547 |
0.442 |
0.482 |
0.481 |
0.025** |
Table 3.1: Determination of relation of BCVA Difference and Mode of Treatment
| BCVA Difference | Mode of Treatment | P Value | |||
| Focal | PDT | I/V | Others | ||
| Mean | -0.02 | -0.12 | -0.03 | 0 |
0.56 |
| SD | 0.18 | 0.15 | 0.35 | 0 | |
Kruskal Wallis test was used to find the P value
Table 4.1:Determination of relation of PED Difference and the Diagnosis
| PED Difference | Diagnosis | P Value | ||
| PCV | WET AMD | CSCR | ||
| Mean | -441.34 | -221.42 | -27.0 | 0.02** |
| SD | 357.50 | 360.28 | 32.52 | |
Kruskal Wallis test was used to find the P value
** indicates a significant association (p-value <0.05)
Table 4.2:Pair wise comparison of the Diagnosis methods using Bonferroni method
| Diagnosis | Mean Difference (PED) | P value |
| 1 Vs 2 | -219.91 | 0.04 |
| 1 Vs 3 | -414.34 | 0.04 |
| 2 Vs 3 | -194.42 | 0.55 |
Bonferroni method was used to find the P value
The difference between 1 Vs 3 is more significant (414.34) as compared to the other possible combinations
Table 5:Determination of relation of BCVA Difference after treatment and the Diagnosis
| BCVA Difference | Diagnosis | P Value | ||
| PCV | WET AMD | CSCR | ||
| Mean | -0.06 | -0.05 | 0.12 | 0.31 |
| SD | 0.25 | 0.40 | 0.16 | |
Kruskal Wallis test was used to find the P value
DISCUSSION
In this study ,outof 50 eyes with extra large PED , mean age was 70 years with almost equal M:F ratio. Out of 50 pigment epithelial detachment 16 eyes ( 32%) were having tall peaked PED (TPP), 11 eyes ( 22 % ) with dome shaped PED ,10 eyes (20%) with thumb shaped PED (TLP), 7 eyes ( 14%) with haemorrhagic PED , and 6 eyes ( 12 %) were having irregular PED. Subretinal fluid was present in 90% of eyes with absence of intraretinal edema in 80 % of eyes. Other characteristic features on OCT were intraretinal hyper reflective dots which were present in 41 eyes (82%).Choroidal thickness was also measured at fovea which showed 80% of eyes with thickness <350 microns.
FFA and ICG findings were showing , 40 eyes ( 80 %) with polypoidal changes like single or multiple polyp with or without branch vascular network ( BVN ).One characteristic ICG feature noted was presence of bunch of polyps in notch of PED which was seen in 62 % of eyes.Presence of polyps at the notch in PED has already been documented by AkitakaTsujikawa et al.7 And presence of bunch of polyps rather than single polyp can explain the extra large height of PED because of excess sub RPE exudation in all these cases.
Based on multimodal imaging features, 40 eyes (80%) were diagnosed as IPCV, 8 eyes ( 16%) as wet age related macular degeneration and 2 eyes ( 4%) as central serouschorioretinopathy. Exudative form of IPCV has beendocumented to be more prevalent in asian population compared to haemorrhagicform.8Exudative form of IPCV isassociated with excess fluid collection sub-RPE and hence large PED. Presence of multiple polyps can explain the extra large height of PED in our patients. Incidence of IPCV in extra large PED cases is similar to a study by Richard et al.Richard et al has documented 85 % incidence of IPCV based on ICG in 40 eyes with large haemorrhagic or exudative PED. 9But to best of author’s knowledge till now there are no studies documenting extra large PED withits multimodal imaging features and its response to various mode of management.
Management of large and X large PED is always difficult because variable course of PED after treatment and chances of complication like RPE rip.10 In a subgroup analysis of HARBOR study authors have documented 102 eyes (25 % ) with X-large PED (352-1395.2 microns vertical height )at baseline.Authors have shown Ranibizumab 0.5 mg to be effective for X-large PED in PRN or monthly basis.(Presented in ASRS Meeting 2015)
Shima C et al has documented PDT to be effective in reducing PED height with stabilization of vision in 22 eyes with serous PED.11
Our study shows out of all 50 eyes , 8 eyes were lost to follow up after diagnosis. Out of remaining 42 eyes 5 eyes(12 %) were treated with focal green laser, 11 eyes(26%) were treated with photodynamic therapy(PDT) with I/V anti-VEGF( combination therapy) ,23 eyes ( 54%) with intravitreal Anti-VEGF monotherapyand 3 eyes(8%) were observed or patients refused for treatment.
Response to treatment was analysed on basis of change in height of PED and change in baseline BCVA after treatment at final follow up.All the patients who received treatment in form of focal laser , PDT or intravitreal anti-VEGF showed decrease in PED height compared to patients who were observed or who refused for treatment. This difference was statisticaly significant ( p = 0.04). Among various treatment modalities focal laser and PDT were found to have more decrease in PED height compared to anti-VEGF at final follow up.
In a subgroup analysis comparing response to treatment in form of reduction in PED height among wet AMD, PCV and CSCR subgroups, patients in PCV subgroup were having significantly better reduction in PED height compared to wet AMD and CSCR ( p= 0.02).
After taking treatment in the form of laser , PDT or anti- VEGF ,patients experienced significant improvement in mean BCVA at final follow up which was statistically significant ( p = 0.025) .There was no significant difference in BCVA improvement within subgroups of IPCV , wet AMD ,and CSCR.
Presence of RPE rip was seen in 4 eyes ( 8%) after treatment and contrary to common belief none of the patients treated with PDT developed RPE rip.All 4 patients were treated with anti VEGF who developed RPE rip after treatment.
Our study shows that polypoidalchoroidalvasculopathy can present with extra large PED due to excess of exudation because to multiple polyps.Multimodal imaging is very useful to study features of extra large PED and their response to various treatment.Further evaluation for more characteristic features and treatment response is needed with larger studies involving more number of patients.
CONCLUSION
This is a unique study where we have evaluated Extra large PED with help of multimodal imaging to Idiopathic polypoidalchoroidalvasculopathy( IPCV) was the major etiology with characteristic double layer sign in SD-OCT and presence of multiple polyps in notch of PED in ICG. Focal green laser and photodynamic therapy ( PDT) were the treatment modalities which showed significant reduction in height of PED without producing any RPE rip compared to I/V anti-VEGFmonotherapy.
REFERENCES
- Gass JD. Pathogenesis of tears of the retinal pigment epithelium. Br J Ophthalmol.1984;68:513-519.
- Zayit-Soudry S, Moroz I, Loewenstein A. Retinal pigment epithelial detachment. 2007;52(3):227-243.
- Pepple K Mruthyunjaya P.- Retinal pigment epithelial detachments in
- age related macular degeneration:classification and therapeutic options.SeminOphthalmol. 2011;26:198-208.
- Pauleikhoff D, Löffert D, SpitalG,et al.- Pigment epithelial detachment in the elderly. Clinical differentiation, natural course and pathogenetic implications. Graefes Arch ClinExpOphthalmol.2002;240:533-538.
- Allen C. Ho,Brandon G. Busbee et al – Twenty-four-Month Efficacy and Safety of 0.5 mg or 2.0 mg Ranibizumab in Patients with SubfovealNeovascular Age-Related Macular Degeneration. Ophthalmology 2014;121:2181-2192
- JordiMonés , Marc Biarnés ,JosepBadal – Bimonthly half-dose ranibizumab in large pigment epithelial detachment and retinal angiomatous proliferation with high risk of retinal pigment epithelium tear: a case report- Dove Press Journal – Clinical Ophthalmology – 6/6/2013 AkitakaTsujikawa et al – Pigment Epithelial Detachment in PolypoidalChoroidalVasculopathy- Am J Ophthalmol 2007; 143:102–111
- .Wong C W et al -Age-related macular degeneration and polypoidalchoroidalvasculopathy in Asians – ProgRetin Eye Res. 2016 Jul;53:107-39
- Richard M Ahuja–Polypoidalchoroidalvasculopathy in exudative and haemorrhagic pigment epithelial detachments – Br J Ophthalmol 2000;84:479–484
- BozhoTodorich et al- Treatment of pigment epithelial detachments in age-related macular degeneration – Retinal Phyician-11/1/2012
- Shima C, Gomi F, Sawa M, Sakaguchi H, Tsujikawa M, Tano Y. One-year results of combined photodynamic therapy and intravitrealbevacizumab injection for retinal pigment epithelial detachment secondary to age-related macular degeneration. Graefes Arch ClinExpOphthalmol. 2009;247:899-90

