Unnikrishnan Nair*,Manoj S, Lata Iyoob*
*Chaithanya Eye Hospital, Kesavadasapuram, Thiruvananthapuram, Kerala, India.
No Financial Disclosures: Nil
Conflict of interest: None stated
ABSTRACT
AIMS:
To evaluate the choroidal thickness in different variants of neovascular AMD like occult choroidal neovascularisation (occult AMD) & Polypoidal choroidal vasculopathy (PCV) and study the change in thickness during anti-VEGF treatment.
METHODS:
This prospective study conducted in Chaithanya Eye Hospital in 2014 included 60 eyes with neovascular AMD including occult AMD& PCV treated with 0.5mg intravitreal Ranibizumab or 1.25mg intravitreal Bevacizumab on PRN basis and followed up for 6 months. Patients with choroidal neovascularisation secondary to myopia or other causes and eyes in which meaningful EDI image could not be obtained were excluded. Subfoveal choroidal thickness (SFCT) was measured by Enhanced Depth Imaging on spectralis SD-OCT before and after injection at monthly intervals.
RESULTS:
31 eyes (51.7%) were occult AMD & 29 eyes (48.3%) had PCV. The mean SFCT of occult AMD group was 189.68±58 and PCV group was 210.93±69. The SFCT decreased from 199.53±63 at baseline to 195.97±64 at 1 month (p=0.49), 186.55±64 at 3 month (p=<0.001), 179.43 ± 60 at 6 month (p=<0.001). The average SFCT in month 4,5 & 6 in the repeat injection group was more compared to that in the group that didn’t require repeats. Subgroup analysis revealed PCV eyes had earlier increase in SFCT and less reductions in SFCT with further injections
CONCLUSIONS:
This may indicate that SFCT may be used as a surrogate marker to determine continuation of Anti-VEGF therapy and also that the effect of Anti-VEGF on the thickened choroid in PCV may be transient.
INTRODUCTION
Age related macular degeneration (AMD) is a chronic progressive generally irreversible disorder of the macula resulting in gradual loss of central vision commonly beyond 50 years of age. Clinical findings, such as geographic macular atrophy, neovascularisation, drusen, hyperpigmentation, and hypopigmentation help to confirm the diagnosis of AMD. Diagnostic modalities include stereoscopic slit-lamp biomicroscopic examination, optical coherence tomography (OCT), fundus fluorescein angiography, indocyanine angiography and fundus autofluorescence imaging. Noninvasive imaging modalities like OCT present an exciting avenue for the management of AMD as it helps in early detection of the disease and can monitor the effect of treatment precisely. The introduction of OCT has revolutionised the way these pathologies are managed as demonstrated in the various anti-VEGF multicentric trials in the management of wet AMD. The characteristic OCT features described in wet AMD include thickening of the retina, retinal pigment epithelial abnormalities like pigment epithelial detachment with/without internal reflectivity, subretinal space fluid/hemorrhage, intraretinal fluid, inner-segment outer-segment layer abnormalities and changes in the Bruch’s membrane and underlying choroid.
Imaging of the choroid with conventional commercial OCT has been difficult because of difficulty in signal transmission. This is because the wave length of the light source used to image retina is not long enough to penetrate into the choroid1. The common commercially available OCTs employ a wavelength of 800nm, whereas those capable of choroidal imaging are in a range of 1060nm1,2. One would have to use a light source nearer to the infrared region to penetrate into the choroid. A recent modification to the standard technique, termed Enhanced depth imaging optical coherence tomography (EDI-OCT) is able to image the choroid using SD-OCT. Several workers have analysed the use of EDI-OCT in normal eyes as well as in typical neovascular AMD and other disorders.3,4,5,6 The results from these few studies present conflicting data.7,8 There has been no general consensus on the pattern of data from EDI-OCT evaluation in AMD resulting in dearth of formal guidelines for the same. This study is designed to prospectively evaluate choroidal thickness using EDI-OCT in patients with AMD receiving anti-Vascular Endothelial Growth Factor (anti-VEGF).
AIMS AND OBJECTIVES
To evaluate the choroidal thickness in different variants of neovascular AMD like occult choroidal neovascularisation (occult AMD) & Polypoidal choroidal vasculopathy (PCV) and to study the change in thickness during anti-VEGF treatment.
MATERIALS AND METHODS
The study was conducted as a prospective follow up cohort study at Chaithanya Eye Hospital and Research Institute, Thiruvananthapuram inpatients diagnosed to have Age Related Macular Degeneration and those who received Intravitreal anti-VEGF injection between August 2013 and January 2015.The main study was preceded by a pilot study which was conducted on 20 patients with AMD on follow up at our institute in 2013 to establish baseline parameters of the affected individuals in south India, in the absence of previous data on the population concerned. The sample size for the index study was derived by statistical measures from data obtained from the pilot study as 60 cases.
Inclusion criteria: Patients with AMD those receive anti-VEGF therapy-intravitreal Bevacizumab, Ranibizumab or Pegaptanib sodium with/without PDT will be included. Patients with AMD includes eyes with choroidal neovascularisation- typical AMD features as described by international AMD classification, eyes with variants of AMD like Polypoidal choroidal vasculopathy (PCV) diagnosed by ICG.
Exclusion criteria: 1.Patients with choroidal neovascularisation secondary to myopia or other causes, 2. Eyes in which meaningful EDI image not obtained due to media opacities or other causes, 3.Patients who have not followed up periodically during the period of the study.
The diagnosis of Wet AMD was based on clinical examination and angiographic evaluation (FFA and ICG Angiography whenever necessary). All the eligible patients were subjected to EDI- OCT before starting the treatment and at different intervals during follow up till 6 months.The OCT machine used for analysis was the SPECTRALIS spectral domain OCT. Choroidal thickness is defined as the distance between the hyper-reflective line corresponding to Bruch’s membrane beneath the retinal pigment epithelium and the inner surface of the sclera which can be identified by the change in reflectivity characteristics between the choroid and sclera (fig 1). This thickness was measured by using calliper at the subfoveal region. The primary outcome measure was estimation of the subfoveal choroidal thickness (SFCT) in different types of AMD. The secondary outcome measures included the change in subfoveal choroidal thickness during follow up especially before and after anti-VEGF therapy. Our study hypothesis was that administration of anti-VEGF therapy in AMD would cause a decrease in SFCT and SFCT could be used as a guide for predicting disease activity of AMD. The study conducted after clearance from the institute ethical committee.
STATISTICAL ANALYSIS:
Statistical analysis was done by SPSS v20. Categorical data are displayed as proportions and continuous data as mean ± standard deviation. Pearson chi-square test and Fisher exact test were used for comparison of discrete variables as appropriate. The t-test was used for comparison of continuous variables. All p-values were two – tailed and a value of less than 0.05 was considered statistically significant.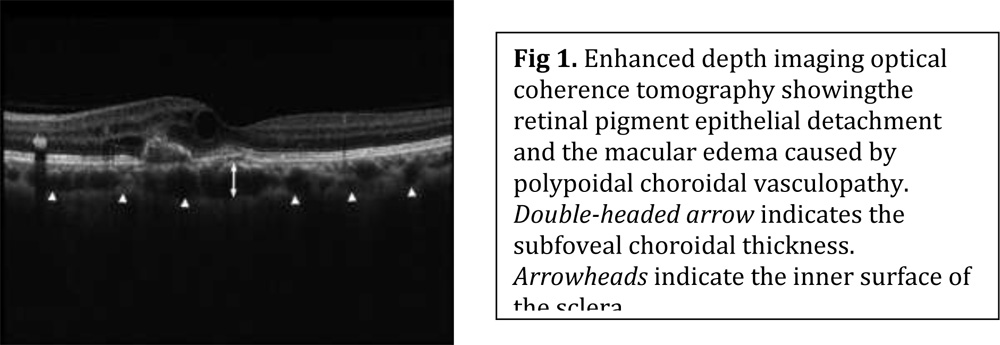
RESULTS
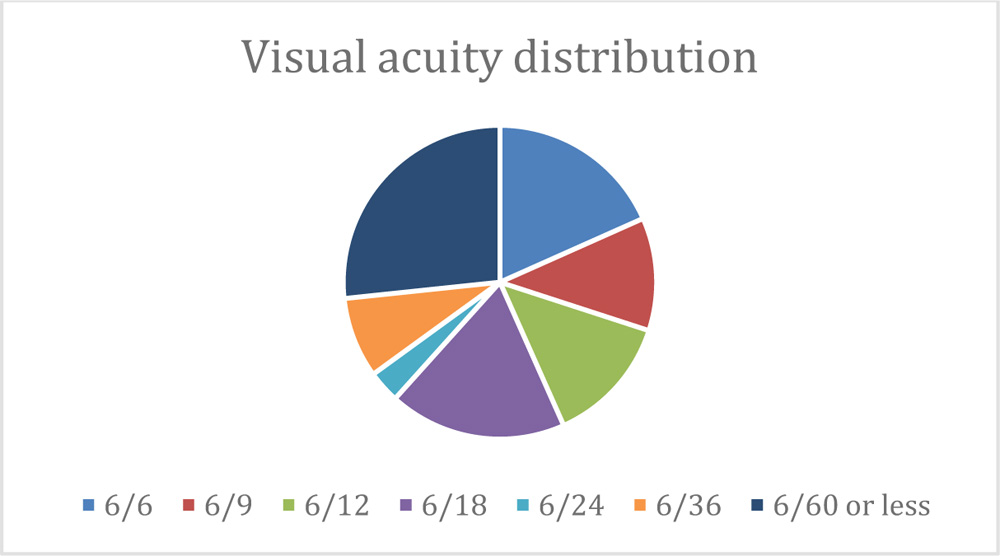
60 patients with AMD who underwent intravitreal anti-VEGF injections were included in the study cohort. There was male preponderance with a mean age of 69.15 years. The mean age of wet AMD group was 69.48years and that of PCV group was 69.75years. 31 eyes(51.7%) were wet AMD, 29 eyes(48.3%) had PCV. 26.7% of the population were having 6/60 or less vision, 18.3% were having 6/6 and 6/18 vision, 13.3% with 6/12 vision, 11.7% with 6/9 vision, 8.3% with 6/36 vision and the rest with 6/24 vision. Average greatest linear dimension of choroidal neovascular membranein the patients was 3291.37microns.Their average central foveal thickness was 438microns. In regard to the subtypes of neovascular AMD, the mean SFCT at baseline was 189.68±58microns in 31eyes with typical neovascular AMD and 210.93±69micron in 29 eyes with PCV. However, the difference between the 2 subtypes was not significant (p= 0.208).
All the patients received monthly injections for the first 3 months. Subsequent injections were given based on clinical indications. Maximum number of injections received was 5 and minimum was 3. On 6 months follow up, 18 patients showed improvement in visual acuity at least by one line in Snellen’s visual acuity chart, 31 patients no change in VA, 11 patients showed decrease in VA. In the study cohort, 10 patients developed complications following intravitreal anti-VEGF injections. 7 of them developed severe subconjunctival haemorrhage and 3 patients developed secondary rise in intraocular pressure.
Of the 60 affected eyes, the mean subfoveal choroidal thickness as measured by EDI-OCT images significantly reduced from 199.53 ± 63 microns at baseline to 195.97± 64 microns at 1 month follow up (p=0.49), 186.55 ± 64 micron at 3 months (p=<0.001), and 179.43 ±60 microns at 6 months (p=<0.001) . SFCT demonstrated a decreasing trend with anti-VEGF injection independent of the subtype of neovascular AMD (fig 3).
Fig 3: Change in subfoveal choroidal thickness after administration of anti-VEGF therapy in different subsets of AMD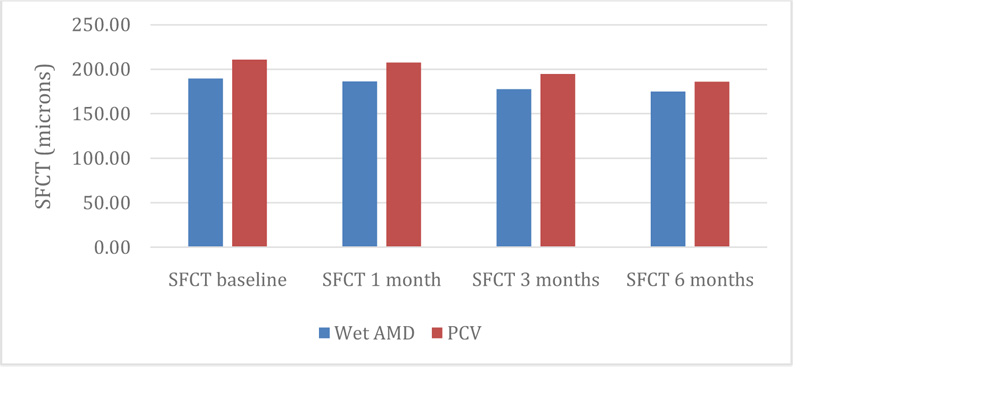
Administration of anti-VEGF therapy after three loading doses based on clinical indications (increase in central foveal thickness) without a routine choroidal thickness evaluation by EDI-OCT allowed a comparison of those who received subsequent injections and those who did not. Those who received repeat injections of anti-VEGF also had significant increase in choroidal thickness unlike those who did not (fig 4). This difference was consistent among even on subgroup analysis of these patients.
Fig 4: Comparison of monthly change in choroidal thickness measured by EDI-OCT in those who underwent anti-VEGF injections by clinical indications: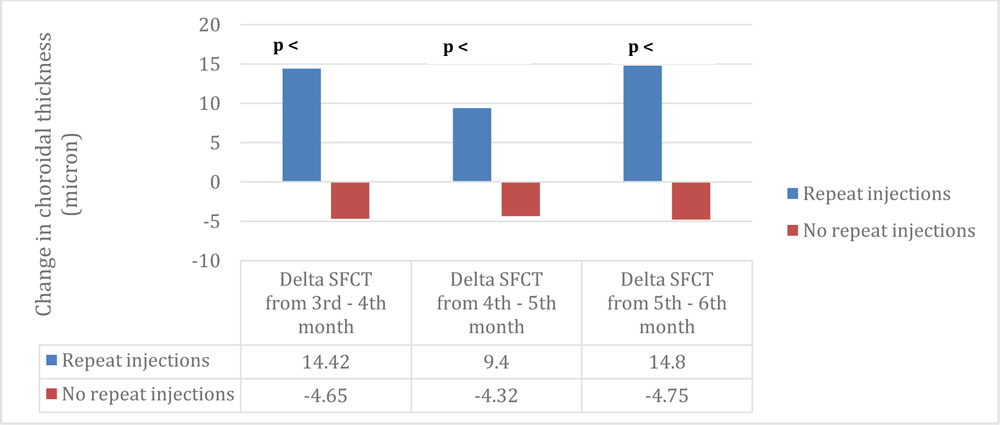
Of the 29 eyes with PCV , there was early flattening of the macula in 18 eyes after the loading dose of 3 injections. 11 eyes had persistent activity at the fovea and required more injections. These eyes had angiographic evidence of macular involvement of the polyps and increased SFCT. In these patient there was less reduction in SFCT with subsequent injections and the SFCT was persistently high.
All eyes in the Repeat Injection group showed elevated SFCT measurements. Only 39 of the 47 eyes were re-injected at 4 months based on the clinical / OCT parameters. 8 eyes had resolution of clinical and tomographic features but continued to have thickened SFCT. 47 eyes were re-injected at Month 5 (Table 1).
Table1
| Number of eyes in each group | Repeat Injection N=47 |
| No Repeat Injection N=13 | |
| Number of eyes injected at Month 4 | N=39 |
| Number of eyes injected at Month 5
|
N=47 |
| Number of eyes injected at Month 6
|
N=43 |
| Persistently higher SFCT | All eyes in Repeat Injection group |
DISCUSSION
We evaluated the choroidal thickness in different variants of neovascular AMD and studied the change in thickness during anti-VEGF treatment.The subfoveal choroidal thickness in eyes treated with intravitreal anti-VEGFshowed significant decrease over 6 months.Results of the present study indicate the possibility that intravitreal Anti-VEGF influenced the choroidal structure under the neovascular membrane in neovascular AMD.Recent studies have reported that eyes with PCV have a thicker choroid under the fovea than those with typical neovascular AMD9,10In the present study, subfoveal choroidal thickness in eyes with PCV at baseline was not significantly thicker than that in eyes with typical neovascular AMD. Subfoveal choroidal thickness in eyes with the subtypes of neovascular AMD, namely, typical neovascular AMD & PCV, showed a similar trend toward decreasing during the following 6-month period, which may implicate that intravitreal injections has a constant effect on the choroid, regardless of the subtype of neovascular AMD and the treatment histories.
Vascular endothelial growth factor-A provides various pharmacologic actions on the choroid, such as increases in microvascular permeability, angiogenesis, and survival for the vascular endothelial cells.11,12Vascular endothelial growth factor-A is also considered to play a key role in the pathogenesis of neovascular AMD. In fact, a significantly increased expression of VEGF was measured in the aqueous humor of eyes with neovascular AMD, and a significant decrease of VEGF was observed after Intravitreal Ranibizumab.13In rabbits, Ranibizumab rapidly penetrates after intravitreal injectionthrough all retinal layers to reach the choroid14and may have the potential to inhibit the activity of all VEGF-A isoforms.11,12 One reason for the decreased choroidal thicknessin the current study might be related to the reduction ofchoroidal vascular permeability, as seen in eyes with central serous chorioretinopathy treated with Intravitreal Bevacizumab.15
The re-injection criteria was based on an OCT/clinical evaluation based PRN protocol. The measurement of SFCT was an observatory tool and did not affect any treatment decisions. 8 eyes that were not injected at Month 4 , required to be injected at Month 5. These eyes did not require injections at month 4 based on the absence of clinical and tomographic evidence of activity , but noteworthy was the fact that they still had elevated SFCT. Thus the persistence of elevated SFCT at Month 4 probably predated the retinal changes requiring reinjection.
Our study shows that, beside central foveal thickness, evaluation for increase in choroidal thickness by EDI-OCT could also be used to guide the need for repeat anti-VEGF injections in neovascular AMD. This could possibly be explained by the basic pathology of AMD which involves the formation of a choroidal neovascular membrane and consequent increase in SFCT and a rise in central foveal thickness.
The study cohort in our trial was small and the generalizability of its results may have consequent limitations. However, the prospective design of our trial with periodic follow up with spectral – domain OCT is unique. Choroidal thickness in all OCT images was evaluated manually because of lack of software for automated measurement of choroidal thickness in current OCT equipment. However, the observations were compared with that of another independent observer to minimise inter-observer variability.
CONCLUSIONS
Subfoveal choroidal thickness decreased after intravitreal injections in eyes with neovascular age related macular degenerationirrespective of the pathologic subtypes. Increase insubfoveal choroidal thickness could be considered as a marker of disease activity in these patients as SFCT alterations often predated clinical and tomographic signs of activity. SFCT reduction was seen to transient in cases of macular PCV.
REFERENCES
1.Povazay B, Hermann B, Unterhuber A, et al. Three-dimensional optical coherence tomography at 1050 nm versus 800 nm in retinal pathologies: enhanced performance and choroidal penetration in cataract patients. J Biomed Opt. 2007;1204-1211
2.Ikuno Y, Kawaguchi K, Nouchi T, Yasuno Y. Choroidal thickness in healthy Japanese subjects. Invest Ophthalmol Vis Sci. 2010;51:2173–2176.
3.Spaide RF, Koizumi H, Pozonni MC. Enhanced depth imaging spectral-domain optical coherence tomography. Am J Ophthalmol 2008;146:496 –500.
4.Margolis R, Spaide RF. A pilot study of enhanced depth imaging optical coherence tomography of the choroid in normal eyes. Am J Ophthalmol 2009;147:811–5.
5.Koizumi H, Yamagishi T, Yamazaki T, et al. Subfovealchoroidal thickness in typical age-related macular degeneration and polypoidalchoroidalvasculopathy. Graefes Arch ClinExpOphthalmol 2011;249:1123– 8.
6.Chung SE, Kang SW, Lee JH, Kim YT. Choroidal thickness in polypoidalchoroidalvasculopathy and exudative age-related macular degeneration. Ophthalmology 2011;118:840–5.
7.Waheeda Rahman, Fred Kuanfu, Jonathan Yeoh. Enhanced depth imaging of the choroid in patients with neovascular age-related macular degeneration treated with Anti-VEGF therapy versus untreated patients.Clinical and experimental ophthalmology. june 2013, volume 251 pp1483-1488
8.Jonas JB,Forster TM,SteinmetzP,SchlichtenbredeFC,Harder BC Choroidal thickness in age related macular degeneration. Retina 2013 Nov 11.(Epub ahead of print)
9.Koizumi H, Yamagishi T, Yamazaki T, et al. Subfoveal choroidalthickness in typical age-related macular degenerationand polypoidal choroidal vasculopathy. Graefes Arch ClinExp Ophthalmol 2011;249:1123– 8.
10.Chung SE, Kang SW, Lee JH, Kim YT. Choroidal thickness in polypoidal choroidal vasculopathy and exudative age-related macular degeneration. Ophthalmology 2011;118:840–5.
11.Lowe J, Araujo J, Yang J, et al. Ranibizumab inhibits multiple forms of biologically active vascular endothelial growth factor in vitro and in vivo. Exp Eye Res 2007;85:425–30.
12.Ferrara N, Damico L, Shams N, et al. Development of ranibizumab, an anti-vascular endothelial growth factor antigen binding fragment, as therapy for neovascular age-related macular degeneration. Retina 2006;26:859 –70.
13.Funk M, Karl D, Georgopoulos M, et al. Neovascular agerelated macular degeneration: intraocular cytokines and growth factors and the influence of therapy with ranibizumab.Ophthalmology 2009;116:2393–9.
14.Gaudreault J, Fei D, Beyer JC, et al. Pharmacokinetics andretinal distribution of ranibizumab, a humanized antibody fragment directed against VEGF-A, following intravitreal administration in rabbits. Retina 2007;27:1260–6.
15.Lim SJ, Roh MI, Kwon OW. Intravitreal bevacizumab injection for central serous chorioretinopathy. Retina 2010;30:100–6.

