Dr. Girish Bharat Velis, V16197, Dr. S Bala Murugan
Introduction
Acute retinal necrosis (ARN), described first by Urayama et al1 in 1971, is a severe sight threatening disease, affecting both immunocompetent as well as immunocompromised individuals. The causative organisms commonly implicated are Varicella zoster virus (VZV), Herpes simplex virus (HSV), Epstein-Barr virus (EBV) and Cytomegalovirus (CMV).2,3 The diagnostic criteria4 as per American Uveitis Society are: 1) one or more foci of retinal necrosis with discrete borders located in the peripheral retina; 2) rapid progression in the absence of antiviral therapy; 3) circumferential spread; 4) evidence of occlusive vasculopathy with arterial involvement; and 5) a prominent inflammatory reaction in the vitreous and anterior chamber. The visual outcome is generally poor due to its propensity to cause retinal breaks finally leading to retinal detachment (RD).5,6 Rhegmatogenous RD (RRD) occurs in 50-75% of eyes.7 Despite the proposition of prophylactic laser for RD, the final visual outcomes are conflicting. ARN is often overlooked or misdiagnosed as another uveitic entity due to inappropriate analysis of clinical features and lack of use of molecular diagnosis. Corticosteroid use without the cover of antivirals, inappropriate use of antivirals (late initiation, low dose, short duration of therapy) or failure to recognize resistance to antiviral therapy results in disease progression, bilateralisation, subsequent complications and ultimately blindness.
There is paucity of literature regarding the visual outcomes of ARN. We describe the spectrum, clinical profile and clinical outcome of eyes with acute retinal necrosis (ARN) syndrome.
Materials and Methods
Case records of 13 eyes of 10 patients newly diagnosed as ARN syndrome from 1st Jan 2013 to 31st Jan 2016 were retrospectively analysed. Inclusion criteria of enrolled patients were:
1) Cases diagnosed with acute retinal necrosis as per American Uveitis Society criteria;
2) Complete clinical records of best-corrected visual acuity (BCVA), slit lamp biomicroscopic examination, intraocular pressure, treatment records at baseline and follow-up visits, complications if any;
3) Cases with complete evaluation to rule out TB and syphilis (like Mantoux test, TPHA);
4) All other known causes of infectious and noninfectious uveitis and known masquerading uveitic syndromes excluded by clinical features or relevant tests, and
(5) A minimum of 6 months of follow-up from the initiation of treatment.
Cases of ARN with inappropriate follow up details were excluded. The data collected included demographics, clinical profile and history of immunocompromised status. The clinical parameters (signs of panuveitis, retinitis, retinal necrosis, occlusive vasculitis, retinal detachment), visual acuity at presentation and during follow up (1 week, 1 month, 3 month, 6 month and last follow up), aqueous or vitreous tap for diagnostic PCR DNA analysis, extent of retinal involvement were documented. The occurrence of RD, its type and time of occurrence, rate of re-detachment, presence of other sequelae, prophylactic laser received or not, treatment details resulting in final functional and anatomical outcomes were meticulously analyzed.
Patients were treated with intravenous antiviral therapy (Acyclovir 10 mg/kg thrice daily) for 7–10 days, followed by oral antiviral therapy (Valacyclovir 1 g thrice daily) for next 6–8 weeks. Patients who developed side effects with acyclovir and cases in which serous RD was involving the posterior pole were administered intravitreal Ganciclovir (2-5mg/0.1ml BD per week). Systemic corticosteroids were started 48–72 hours after institution of antiviral therapy in immunocompetent patients. The initial steroid dosing of 1 mg/kg/day, was gradually tapered by 10 mg, based on clinical titration of inflammatory activity. During surgery for RRD the experienced surgeon performed standard 23G 3-port pars plana vitrectomy, with appropriate induction of a posterior vitreous detachment and 360-degree encirclage with silicone oil or gas tamponade. A favorable functional outcome was considered as best-corrected visual acuity greater than 6/60 at final follow-up.
Results
The mean age at presentation was 39.9 years (range 15-58 years). Eight patients were males and 2 females (male to female ratio = 4:1). The follow up ranged from 2-24 months (mean = 6 months). Six cases (60%) presented with immunosuppression. Five patients (50%) had history of chickenpox. One patient had history of intravitreal triamcinolone acetonide administration. At presentation, all patients had typical peripheral retinitis with no macular involvement. Bilateral acute retinal necrosis (BARN) was seen in 3 cases (30%), two of them were immunocompromised while one was immunocompetent. One of the cases with BARN had bilateral uveal effusion. Polymerase chain reaction results from vitreous aspirate of four eyes yielded positivity in two (Cytomegalovirus -1 and Varicella Zoster virus -1). Among three eyes (25%) with retinal detachment, one had serous RD at presentation and two had rhegmas developed after 1.5 months of treatment. Two of the patients who underwent silicon oil removal had redetachment and so it was reimplanted. Within a week of retinal surgery, one of the patient developed ischemic stroke and succommed to it. Intravitreal Ganciclovir was given in 2 eyes. Prophylactic laser photocoagulation was performed in 2 (16%) eyes and no retinal detachment was noted in the follow up till 18 months. Favourable
functional outcome of BCVA > 6/60 was observed in 5 (38.4%) eyes.
| Case
No. |
Age | Gender | Presenting
BCVA |
H/o pox | Immuno
suppression |
PCR | Laser Px | RD | Timing of RD | Re-RD | Final VA |
| 1 | 57 | Female | 6/9 | yes | Yes, IVTA
for IU |
VZV | No | yes, Rheg | after 1.5 month | Yes | 5/60 |
| 2 | 50 | Female | 6/36 | yes | No | Not done | Yes | No | No | No | 6/9 |
| 3 | 32 | Male | PL | no | No | CMV | No | yes, exudative | at presentation | Yes | HM |
| 4 | 25 | Male | HM (RE)
PL (LE) |
yes | yes, HIV | Not done | No | RE RRD, LE no RD | after 1.5 months | No | RE 4/60 LE No PL |
| 5 | 30 | Male | 2/60 | no | No | Neg | Yes | No | No | No | 6/12 |
| 6 | 52 | Male | 1/60 RE 6/36 LE | no | yes, IV steroids | Not done | No | No | No | No | 6/24 RE 6/6 LE |
| 7 | 58 | Male | 6/24 | yes | yes, HIV | Not done | No | No | no | No | 5/60 |
| 8 | 52 | Male | 6/9 | no | yes, HIV | Not done | No | No | No | No | 5/60 |
| 9 | 15 | Male | 6/60 (RE) 6/36 (LE) | yes | yes, HIV | Neg | No | No | No | No | 3/60 |
| 10 | 28 | Male | 6/36 | no | No | Not done | No | No | No | No | 6/6 |
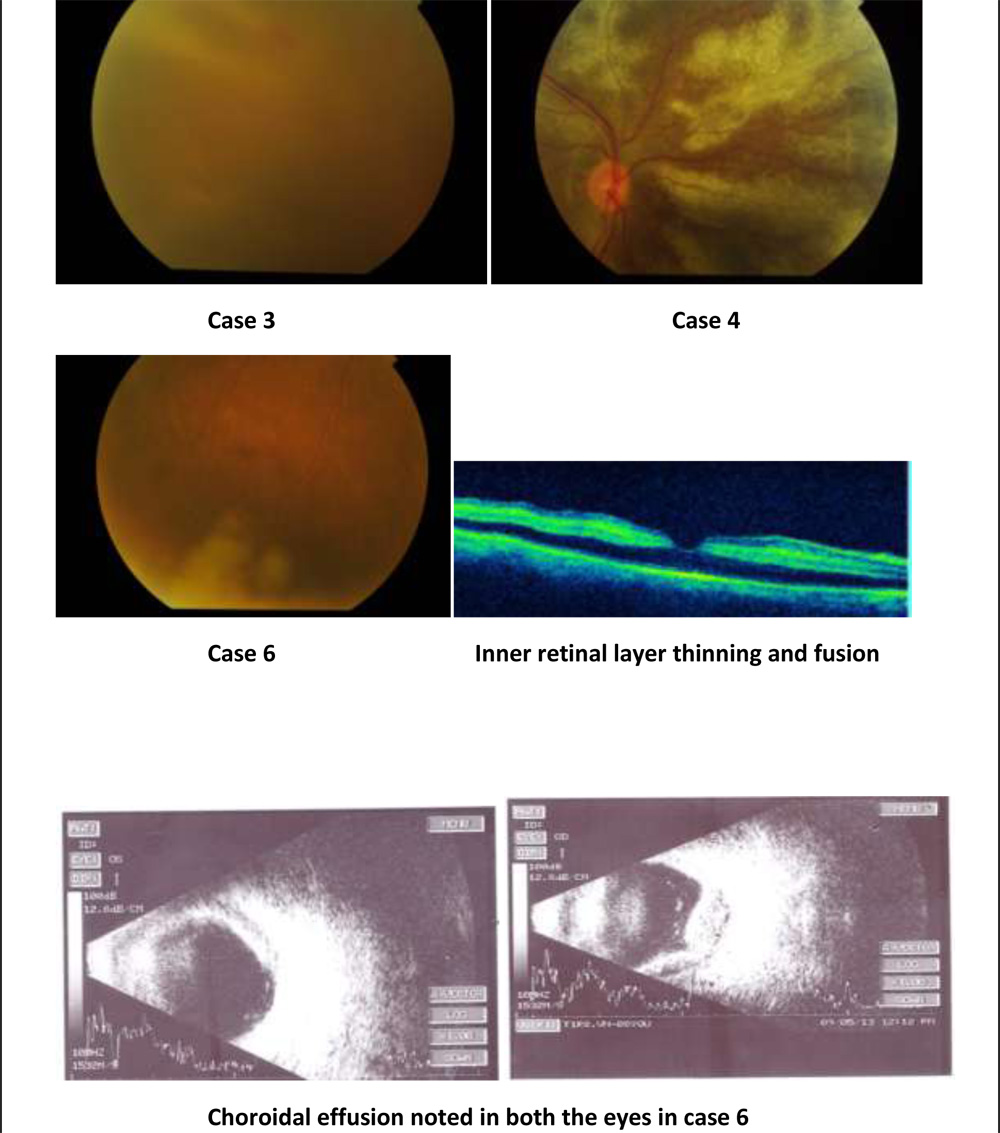
Choroidal effusion noted in both the eyes in case 6
Discussion
In this retrospective study, we analyzed the clinical outcomes of 13 eyes of 10 patients. Usually ARN is not known to have gender predilection, however in our study males outnumbered females by a ratio of 4:1. The age group ranged from 15 to 58 years with mean age of 39.9 years consistent with the findings of Roy et al8 and Hui Ping Chen et al.9
ARN usually occurs in immunocompetent state, however it is not uncommon in immunocompromised patients. In our study, 6 patients (60%) had history of immunosuppression which is higher as compared to the literature. Out of 6 patients with immunosuppresion, 4 were seropositive, one patient was administered intravenous steroids for 3 days before presenting to us and one patient had history of intravitreal triamcinolone injection indicating that local immunosuppression also predisposes to ARN.
Bilateral involvement occurs in approximately one third of patients.10 Our study also showed similar rates of bilateral involvement (3 patients, 30%), two of them were immunocompromised while one was immunocompetent. Roy et al8 reported bilateral involvement in 17% (9 out of 53) patients and proposed that lower dose of antivirals or treatment defaulters as the most common predisposing factor for bilateral involvement. Our cases however had bilaterality at presentation itself and we could not identify any systemic cause for the same despite thorough evaluation. One of the possible explanation for this could be the late presentation and thus delay in institution of acyclovir therapy.11 One of the cases with BARN had bilateral 360 degree drug induced uveal effusion (history of tablet diamox intake to lower the intraocular pressure) that resolved on discontinuation of the drug.
PCR was done in 4 cases and results were positive in 2 cases. One was Varicella zoster virus and the other was positive for CMV. History of chicken pox, in 5 patients, points towards probable VZV etiology, consistent with the findings of Lau et al5 and Hillenkamp et al6, that VZV is the most frequently implicated virus in causing ARN (6 out of 10 cases in our case series). The incidence of RD in our study was 30% which is lower as compared to that reported by Roy et al8 (66%) and Lau et al (60%).5 The probable reason could be the early presentation and early institution of antiviral therapy. Prophylactic laser was done in 2 eyes and was found to be effective to prevent RD. Roy et al8 found no role of prophylactic laser to prevent RD. Systemic corticosteroids are necessary in cases of ARN to control secondary inflammation like vitritis, retinal vasculitis, optic neuropathy and to prevent subsequent retinal and optic disc damage and should be avoided in immunocompromised patients. In our case series, steroid therapy was initiated after stabilization of retinitis under antiviral treatment. This could probably explain the low incidence of RD and better functional visual outcome in this study.
| Our study
(n=13 eyes) |
R. Roy et al8
(n=62 eyes) |
Hui-Ping Chen et al (n=11 eyes) | |
| Age at presentation | 15-58 years (Mean=39.9 years) | 6–70 years
(Mean=36.04 years) |
26-60 years
(Mean=39.4 years) |
| M:F | 8 M, 2 F | 38 M, 15 F | 9 M, 2 F |
| Immunosuppresion | 6 | 2 | NA |
| Chickenpox | 5 | 4 | NA |
| IVTA | 1 | NA | NA |
| Bilateral | 3 | 9 | 2 |
| Peripheral retinitis | 10 | 52 | NA |
| Retinal detachment | 3 | 41 | 8 |
| PCR | 1 VZV, 1 CMV | 28 VZV, 19 HSV | NA |
| Favourable Visual Outcome | 5 eyes | 28 eyes | 3 eyes |
Limitations
The limitations of the study include retrospective design, small sample size, and the procedures like PCR and prophylactic laser could not be uniformly performed in all subjects due to feasibility constraints.
Conclusion
To conclude, prompt diagnosis, early intervention, focused treatment of the sequelae of ARN and meticulous follow-ups helps in regaining better visual outcome and reduces the ocular morbidity of ARN syndrome.
References
1. Urayama A, Yamada N, Sasaki T, et al. Unilateral acute uveitis with retinal periarteritis and detachment. Jpn J Clin Ophthalmol 1971;25:607–19.
2.Ganatra JB et al. Viral Causes of the Acute Retinal Necrosis Syndrome. Am J Ophthalmol 2000;129:166–172.
3.Gavin Walters, Teifi E. James. Viral causes of the acute retinal necrosis syndrome. Curr Opin Ophthalmol 2001, 12:191–195.
4.Holland GN. Standard diagnostic criteria for the acute retinal necrosis syndrome. Executive Committee of the American Uveitis Society. Am J Ophthalmol 1994;117:663–7.
5.Lau CH, Missotten T, Salzmann J, et al. Acute retinal necrosis features, management, and outcomes. Ophthalmology 2007;114:756e62.
6.Hillenkamp J, Nolle B, Bruns C, et al. Acute retinal necrosis: clinical features, early vitrectomy, and outcomes. Ophthalmology 2009;116:1971e5.e2.
7.Clarkson JG, Blumenkranz MS, Culbertson WW, et al. Retinal detachment following the acute retinal necrosis syndrome. Ophthalmology. 1984;91:1665–1668.
8.Rupak Roy, Bikramjit P. Pal, Gaurav Mathur et al. Acute Retinal Necrosis: Clinical Features, Management and Outcomes – a 10 Year Consecutive Case Series. Ocular Immunology and Inflammation. 2014; 22: 170–174.
9.Hui-Ping Chen, Hsi-Kung Kuo, Shih-Hao Tsai et al. Acute Retinal Necrosis Syndrome: Clinical Manifestations and Visual Outcomes. Chang Gung Med 2004;27:193-200
10.Duker JS, Blumenkranz MS Surv Ophthalmol 35:327-343, 1991.
11.Palay DA, Sternberg P Jr, Davis J, et al. Decrease in the risk of bilateral acute retinal necrosis by acyclovir therapy. Am J Ophthalmol. 1991;112:250–255.

