Dr. Deepak Bhojwani, B13467, Dr. Shail, Dr. Viraj Abhayakumar Vasavada, Dr. Vasavada Abhaykumar Raghukant
Use of Ozurdex (Dexamethasone Intravitreal Implant) in patients undergoing cataract surgery with Chronic posterior uveitis.
Abstract
AIM:
To describe the outcomes of concurrent Ozurdex implantation during cataract surgery in patients with chronic posterior uveitis(CPU).
METHODS:This prospective interventional case series comprises of 20 eyes of 20 patients with medically controlled CPU with no recurrent episodes for past 3 months and visually significant cataract. Standardized cataract surgery(phacoemulsification) was followed by intravitreal OZURDEX implantation.
RESULTS:18 eyes(90%)had<grade 2 AC cells- flare on day 1;19 eyes(95%)had no cells-flare at 1 month & at 3 months(P=0.031).Seventeen (85%)eyes had grade 2 or less vitreous cells on day 1;19(95%)eyes had no vitreous cells at 1 month & at 3 months(P=0.001).Mean BCVA increased from preop 0.97 LogMAR to 0.27 LogMAR at 1 month & 0.26 LogMARat 3 months(P=0.04).Mean central macular thickness decreased from 261.3u(preop)to 203.2u(3months)(P>0.05).
CONCLUSION:
Ozurdex safely & effectively controls postoperative inflammation when concurrently implanted during cataract surgery.
INTRODUCTION:
Cataract is a frequent sequelae of uveitis, primarily as a result of disease process and its longterm treatment with systemic corticosteroids.1
The main problem following cataract surgery on uveitic patients is the exacerbation of inflammation. Cystoid macular edema (CME) is an important cause of decreased vision after cataract surgery and can prove especially challenging in patients with uveitis. The incidence of CME after uveitic cataract surgery ranges between 21% and 50%, a much higher rate than in nonuveitic patients undergoing cataract surgery.2 Macular edema in the uveitic patient can be difficult to treat, especially in the perioperative setting. Therefore peripoerative topical,local and systemic corticosteroids are routinely preferred by most of the clinicians.
Current evidence supports reliable safety and favorable efficacy profile of Ozurdex implant in controlling inflammation in uveitic eyesalong with significant improvement in visual acuity.3
We report a novel approach of controlling postoperative inflammation with intraoperative use of OZURDEX implant along with cataract surgery in uveitic patients.
AIM / PURPOSE:
To describe the outcomes of concurrent (Intraoperative) Ozurdex implantation during cataract surgery in patients with medically controlled chronic posterior uveitis.
MATERIALS & METHODS:
Design:
The study was a prospective, interventional pilot trial approved by our institutional ethics committee.
Methods:
This interventional case series comprises of 21 eyes (20 patients) with medically controlled posterior uveitis and visually significant cataract with no episodes of recurrence for past 3 months. Phacoemulsification was performed using standardized technique followed by in the bag intraocular lens implantation. We preferred to inject Intravitreal dexamethasone implant (Ozurdex®,AllerganInc) at the end of surgery. The idea was to avoid injecting implants in compromised posterior capsular support cases to avoid perioperative IOL or implant related complications. No oral steroids were given perioperatively.
Postoperatively our primary outcome measures were anterior chamber cells ,flare & vitreous cells graded as per SUN classification. Secondary outcomes measures were central macular thickness on OCT , best corrected visual acuity and intraocular pressure.
Patients were followed on Day 1, 1 week, 1 and 3 months.
Preoperative and follow-up assessment included visual acuity using Early Treatment Diabetic Retinopathy Study charts, intraocular pressure by Goldmann applanation tonometry, slit lamp examination for anterior and vitreous inflammation, fundus photography (Carl Zeiss FF 500, plus, Zeiss Meditech, Dublin, CA). Central macular thickness (CMT) was measured by optical coherence tomography (HD 5000 Cirrus OCT, Carl Zeiss).
RESULTS:
In our series, 1 eye had intraoperative posterior capsular tear and so didn’t received concurrentOZURDEX which was excluded from study.
Eighteeneyes(90%) had grade 2 or less AC cells- flare on postoperative day 1. Subsequently at 1 week followup 10 (50%)eyes had no anterior chamber cells or flare(P=0.021). And finally at the end of 3 months 19 eyes (95%) had no cells-flare(P=0.031).
Seventeen eyes (85%) had grade 2 or less vitreous cells on day 1 &18 (90%) eyes had grade 1 vitreous cells at 1 week. 19 (95%) eyes has no vitreous cells at 1 month and at 3 month (P=0.001).Only one eye had grade 1 anterior chamber cells and vitreous cells at 1 month and 3 months. No eye had recurrence of inflammation until 3 months.
Majority of patients had significant improvement in their visual acuity. Mean BCVA increased from mean preoperative 0.97 LogMARto 0.27 LogMAR at 1 month (P=0.06) & 0.26 LogMARat 3 months(p=0.04).
Mean central macular thickness decreased from preop (261.3u) to 3 months(203.2u). We did not note any case of macular edema posteratively at any followup visit till months in our series.
There was no significant change in mean IOP from preoperative (10mmHg) to 1 week (14mmHg), 1 month (13mmHg) and 3 months(12mmHg) (P>0.05).Twoeyes(10%) had transient IOP spike of >30 mmHg at 2 weeks which were controlled with single anti-glaucoma medication. At the end of 3 months no eye required antiglaucoma medication.
DISCUSSION:
The outcome of cataract surgery in uveitic patients depends upon several factors, namely the diagnosis, proper perioperative management and meticulous surgery. It is now well recognized that chronic inflammation, even low grade, can irreversibly damage the retina and optic nerve and therefore control of inflammation, both pre and postoperatively, is vital.4
Jancevski and Foster recommended the routine use of supplementary perioperative anti-inflammatory therapy to prevent exacerbation of inflammation, CME and reducing damage to ocular structures.5
Also postsurgical macular edema is a complication more frequently encountered in uveitic patients compared to the general population.Several methods of treating postsurgical CME have been described, including topical nonsteroidal anti-inflammatory drugs (NSAIDs), oral acetazolamide, topical steroids, and periocular and intraocular steroids, as well as intravitreal anti–vascular endothelial growth factor (VEGF) injections.
Finding ways to prevent macular edema in the postoperative cataract setting is essential. A systematic review by Kessel and associates found high-quality evidence that topical NSAIDS are more effective than topical steroids in preventing pseudophakic CME.6Previous studies have shown that the incidence of CME after cataract surgery is decreased in patients whose inflammation was controlled for the 3 months prior to surgery. Preoperative oral corticosteroids have also been shown to be effective in decreasing the incidence of CME.7
Several studies have demonstrated the effectiveness of the DEX implant in the treatment of postsurgical macular edema. A retrospective case series conducted by Bellocq and associates, composed of 50 eyes, showed significant functional and anatomic improvements of postsurgical macular edema treated with the DEX implant.8 However, patients with uveitis were excluded in this study. In a prospective trial by Williams and associates, the DEX implant was used to treat 27 patients with macular edema from either uveitis (9 eyes) or Irvine-Gass syndrome (18 eyes), but not uveitic eyes that had undergone cataract surgery.9
The effectiveness of the DEX implant in the treatment of uveitic macular edema outside the perioperative setting has also been established. As part of the CHROME study, Lam and associates included 23 eyes with CME secondary to uveitis treated with the DEX implant. In this subset, 81% of study eyes gained >1 line of vision following treatment with first DEX implant, and the mean peak improvement in central retinal thickness was −274.2 μm.10 In a retrospective chart review by Cao and associates, 27 eyes treated with DEX implant for persistent uveitic macular edema in the absence of active inflammation showed a statistically significant improvement in visual acuity and reduction in mean CMT compared to baseline.11Patients with intraocular surgery within 6 months were excluded from this study.
It has been proposed to insert the DEX implant at the same time as phacoemulsification in patients predisposed to macular edema to prevent postsurgical CME. Agarwal and associates studied 18 eyes with cataract and diabetic retinopathy in a prospective randomized controlled trial. Nine eyes received intravitreal DEX implant at the beginning of phacoemulsification and 9 eyes served as controls. The gain in visual acuity was significantly higher in the group that received DEX implant, and the mean decline in CMT was lower compared to the controls.12 In a retrospective case series, Sze and associates describe a total of 24 eyes with macular edema
secondary to diabetes or retinal vein occlusion (12 eyes each) that underwent phacoemulsification and received DEX implant at time of surgery. Their results also show improved visual acuity and decrease in CMT from baseline.13 It can be postulated that similar inflammatory mediators released during ocular surgery prompt the development of CME in both diabetic and uveitic subjects. Given that DEX is effective for both DME and uveitic CME, it is reasonable to infer that the DEX implant would have a similar effect on postsurgical macular edema in patients with a history of uveitic CME.
We present a case series of 20 eyes treated with intraoperative DEX implant following uneventful cataract surgery. Intravitreal DEX implant was injected intravitreally at the end of cataract surgery with intact posterior capsule without any adverse events in all patients in our case series.
We noted excellent postoperative intraocular inflammation control in majority of cases. Only 1 eye out of 20 had some residual inflammation even at the end of 1month. The same eye had developed retinal detachment secondary to atrophic holes at 6 weeks followup and underwent vitrectomy and silicone oil tamponade for the same.
All patients had significant improvement in their visual acuity from preoperative mean BCVAof 0.97logmar to 0.26 logMAR. The improvement in visual acuity was found to be statistically significant on every visit.
The mean CMT on OCT in our series reduced from preoperative (261μm) to postoperative (203 μm), suggesting that the DEX implant prevents the occurrence or recurrence or worsening of CME in uveitic patients after phacoemulsification.
Only 2 eyes had transient drug related spike in IOP which was medically controlled. None of those eyes were using anti-glaucoma medications at the end of 3 months.
Previous studies show the effect of intravitreal DEX implant to be 4–6 months duration, in regard to treating noninfectious intermediate and posterior uveitis14 and CME from retinal vein occlusion.15 In our series we noted the presence of DEX implant even at the end of 3 months. So thereby our study also supports the existing evidence on duration of intravitreal DEX implant to be more than 3 months.
CONCLUSION:
Single Intraoperative intravitreal dexamethasone implant during phacoemulsification in uveitis patients with cataract is an effective alternative to postoperative oral steroids and immunotherapy for combating exacerbation of inflammation.
Ozurdex safely & effectively controls postoperative inflammation in eyes with chronic posterior uveitis when concurrently implanted during cataract surgery in our small case series.
Similar large clinical studies with longer followup are warranted to demonstrate long term benefit of intraocular inflammation control in uveitic patients.
References:
1: Mehta S, Linton MM, Kempen JH. Outcomes of cataract surgery in patients with uveitis: a systematic review and meta-analysis. Am J Ophthalmol. 2014;158(4):676–692.
2: Baheti U, Siddique SS, Foster CS. Cataract surgery in patients with history of uveitis. Saudi J Ophthalmol 2012;26(1):55–60.
3: Karim R, Sykakis E, Lightman S, Fraser-Bell S. Interventions for the treatment of uveitic macular edema: a systematic review and meta-analysis. Clin Ophthalmol. 2013;7:1109–1144.
4: Rupesh Agrawal, Somashiela Murthy, Sudha K. Ganesh, Chee Soon Phaik, Virender Sangwan, and Jyotimai Biswas, “Cataract Surgery in Uveitis,” International Journal of Inflammation, vol. 2012, Article ID 548453, 16 pages, 2012.
5: M. Jancevski and C. S. Foster, “Cataracts and uveitis.,” Discovery Medicine, vol. 9, no. 44, pp. 51–54, 2010.
6: Kessel L, Tendal B, Jørgensen KJ, et al. Post-cataract prevention of inflammation and macular edema by steroid and nonsteroidal anti-inflammatory eye drops: a systematic review. Ophthalmology 2014;121(10):1915–1924.
7.Be´lair ML, Kim SJ, Thorne JE, et al. Incidence of cystoid macular edema after cataract surgery in patients with and without uveitis using optical coherence tomography. Am J Ophthalmol 2009;148(1):128–135.
8.Bellocq D, Korobelnik JF, Burillon C, et al. Effectiveness and safety of dexamethasone implants for post-surgical macular oedema including Irvine-Gass syndrome: the EPISODIC study. Br J Ophthalmol 2015;99(7):979–983.
9.Williams GA, Haller JA, Kuppermann BD, et al. Dexamethasone posterior-segment drug delivery system in the treatment of macular edema resulting from uveitis or Irvine-Gass syndrome. Am J Ophthalmol 2009;147(6): 1048–1054.
10.Lam WC, Albiani DA, Yoganathan P, et al. Real-world assessment of intravitreal dexamethasone implant (0.7 mg) in patients with macular edema: the CHROME study. Clin Ophthalmol 2015;10(9):1255–1268.
11: Cao JH, Mulvahill M, Zhang L, et al. Dexamethasone intravitreal implant in the treatment of persistent uveitic macular edema in the absence of active inflammation. Ophthalmology 2014;121(10):1871–1876.
12.Agarwal A, Gupta V, Ram J, et al. Dexamethasone intravitreal implant during phacoemulsification. Ophthalmology 2013;120(1). 211–211.
13Sze AM, Luk FO, Yip TP, et al. Use of intravitreal dexamethasone implant in patients with cataract and macular edema undergoing phacoemulsification. Eur J Ophthalmol 2015; 25(2):168–172.
14: Palla S, Biswas J, Nagesha CK. Efficacy of Ozurdex implant in treatment of noninfectious intermediate uveitis. Indian Journal of Ophthalmology. 2015;63(10):767-770. doi:10.4103/0301-4738.171505.
15:Garweg JG, Zandi S. Retinal vein occlusion and the use of a dexamethasone intravitreal implant (Ozurdex®) in its treatment. Graefe’s Archive for Clinical and Experimental Ophthalmology. 2016;254:1257-1265. doi:10.1007/s00417-016-3350-x.
TABLE: 1 Demographics of patients in our study
| PARAMETER | Number of patients |
| Males | 8 |
| Females | 12 |
| Eyes | 20 |
| Mean AGE | |
| Diagnosis | |
| Idiopathic posterior or panuveitis | 9 |
| Intermediate Uveitis | 4 |
| Sarcoidosis | 2 |
| Behcets | 1 |
| Eales disease | 2 |
| Sytemic therapy | |
| Immunomodulators | 3 |
| Steroids | 14 |
| No systemic therapy | 6 |
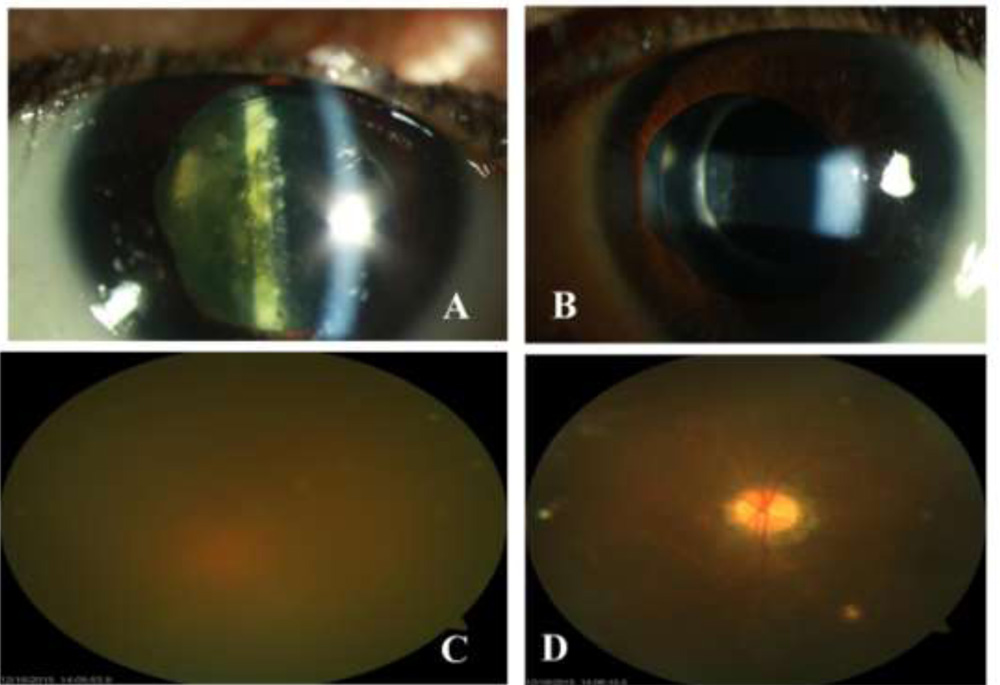
Figure1:
A: External photograph showing typical uveitic cataract with nondilating pupil with posterior synaechiae.
B: Ultrasonography B-scan documenting moderately reflective echoes in vitreous cavity a sign of inflammation.
C: Same eye 2 weeks after cataract surgery and intravitreal DEX implant. (Note the quiet uniflammed eye resembling a routine cataract scenario.
D: Fundus photograph of same eye post cataract surgery with DEX implant documenting clearity of media and absence of vitreous inflammation.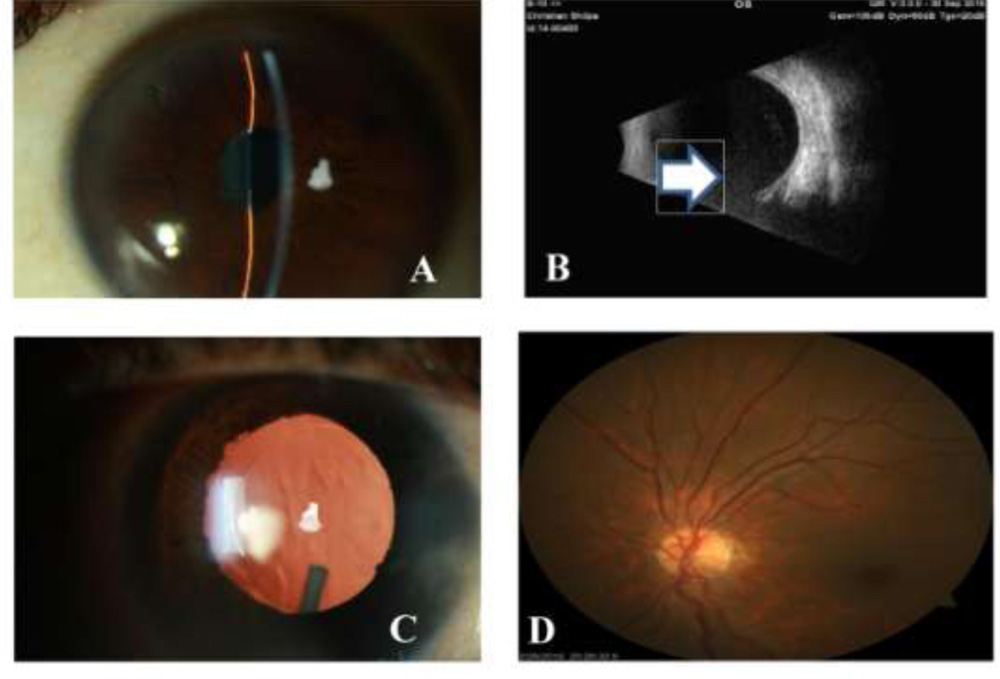
Figure 2
A & C: Represents right eye of case 9 which underwent cataract surgery 5 years back under cover of systemic steroids.
B & D: Represents left eye of case 9 which underwent cataract surgery with intravitreal DEX implant as a part of our study.
(note the obvious superiority of DEX implant in controlling postoperative inflammation following cataract surgery)