Dr. Shruti Bonda, B18477, Dr. Sumita Mohapatra, Dr. Harshavardhan V K, Dr. Parul Priyambada
Co-authors: Prof DrSumitaMohapatra, Harshavardhan VK, ParulPriyambada
INTRODUCTION:
Degenerative myopia[ also known as pathologic myopia or high myopia] is defined as a condition when myopic refractive error is >6.0 D or >26.5mm of axial length associated with degenerative fundus changes. Congenital scleral weakness is the main feature of the disease which eventually leads to globe enlargement, axial lengtheningand posterior staphyloma formation.
The scleral stretching may give rise to various degenerative changes such as progressive atrophy of the choriocapillaries and choroid, linear ruptures of the bruch’s membrane[lacquer cracks]. Other typical features of myopic degeneration include vitreous degeneration and high frequency of peripheral lesions such as lattice degeneration and retinal tears.
Vision in high myopia is not only affected by these causes but also due to damage to the posterior retina in the form of traction induced by the epiretinal membrane (ERM) or residual focal vitreoretinal adhesion (vitreomacular traction), which in these eyes is combined by the presence of posterior staphyloma and progressive global scleral stretching. This unique combination of retinal traction mixed with the complex and distinctive anatomy of degenerative myopia leads to the frequent presence of myopic traction maculopathy (MTM) such as retinoschisis, lamellar holes, or shallow detachment which can further progress to macular hole formation and posterior retinal detachment.
Because of the characteristic and confounding features of the choroid, retina, and vitreoretinal interface in degenerative myopia (tigroid fundus, thin retina, areas of choriocapillary atrophy, retinal pigment epithelium (RPE) hypopigmentation and/or hyperpigmentation, posterior staphyloma), the early stages of traction maculopathy can be easily underestimated by biomicroscopy, angiography or ultrasonography and consequently, its presence can remain undiagnosed. Optical coherence tomography (OCT) thus facilitates the study of the posterior vitreoretinalanatomy of such patients and allows the detection of subtle macular changes that go unnoticed in vast majority of patients.
The purpose of this study is to describe the incidence and features of macular abnormalities in high myopic eyes detected by SD-OCT.
PATIENTS AND METHODS:
This was a prospective observational study done on 88 eyes of 88 patients of high myopia complaining of recent diminution of vision. All the patients attending the outpatient clinic of SCB medical college and hospital were recruited for the study.
The research protocol was approved by the ethics committee of the SCB medical college and informed consent was obtained for each patient.
Inclusion criteria-
- minimum spherical equivalent of -6D
- minimum axial length of 26mm
- diminution of vision is in the last 6 months duration
Exclusion criteria-
- best corrected visual acuity <6/9
- previous intraocular surgery or ocular trauma
- systemic diseases that could affect the retina like diabetes mellitus etc
- intraocular pressure more than 21mm
- gonioscopic findings of angle closure
- Clinical evidence of pseudo-exfoliation, uveitis or pigment dispersion syndrome, corneal or media opacities, retinal pathology or neurological diseases
- Family history in a first degree relative of glaucoma or other optic neuropathy.
All patients were subjected to a
- detailed history taking, refraction using Topcon autorefractometer and best corrected visual acuity (VA) measurement.
- Slit lamp examination, Goldmann applanation tonometry, gonioscopy examination with a Goldmann three-mirror lens
- dilated fundus examination with a 90 diopter fundus lens and indirect ophthalmoscopy with 20D.
- Axial length measurements- Ultrasound A scan is performed for each eye. Axial length is ascertained from the average of six consistent recordings. Care is taken to locate the fovea especially in cases with posterior staphyloma, by ensuring fixation of the A scan probe light.
- Fundus photographywiththeTopcon camera
Optical coherence tomography examination was done through dilated pupils, OCT examination was done through a dilated pupil using commercially available Spectralis OCT Heidelberg Engineering, Heidelberg, Germany. We used the vertical and horizontal 6-mm line scans. The OCT ophthalmoscope produces longitudinal retinal B-scans and coronal C-scans. Therefore, it provides information not readily available by conventional imaging techniques or fundus examination. B-scan produces cross-sectional images of retinal morphology which bears strong resemblance to histology. OCT C-scans are represented as 2-dimensional transverse slices at any given depth through the retina, thereby enabling visualization of the lateral extent of different structures. With C-scan OCT, localization and relation of retinal lesions can be precisely defined, especially in myopic eyes.
Myopic posterior staphyloma, with its outward globe bulge, results in a deep concave B-scan OCT and distorted retinal structures. Because of the smaller base curvature of the horizontal section and larger base curvature of vertical section, macular structures can be defined more easily by vertical sections [especially when the macula lies in the slope of the staphyloma]
All abnormal macular findings were documented, and all the images were evaluated by two-blinded graders.
- Macular retinoschisis was defined as a separation of intraretinal layers, predominantly outer layers by a low reflective space that is separated by an erect columnar microstructure.
- The ERMs observed in this study appeared as noticeable layer over the inner surface of the retina, and at the same time, internal limiting membrane (ILM) could be distinguished as well at the same cut below the ERM. ILM detachment was associated with Muller cell columns that bridge from ILM to the rest of the retinal layers.
- Dome shaped macula was characterized as an inward convexity of the macula that occurred within the concavity of a posterior staphyloma.
- Myopic choroidal neovascularization (CNV) was defined by OCT as the neovascular tuft with a highly reflective dome-shape elevation above the RPE.
RESULTS:
A total of 88 eyes of 88 patients were studied. We included only eyes with recent complaint of diminution of vision i.e within 6 months of duration. There were 40 males and 48 females. 60 % of the studied eyes were the right eyes. The mean age of patients is 56 ± 11.4 [range: 24-65] years. Mean spherical equivalent in these eyes was 16.5 ± 5.6 [range: 8.75-29.75] D. Mean axial length was 29.1 ± 2.1mm [range: 26.5-32.5] The mean best corrected VA was 0.235 ± 0.2 (0.05-0.5).
All eyes had one or more chorioretinal features typical of degenerative myopia (tigroid fundus, stretched vascular arcades, peripapillary atrophy, chorioretinal atrophy, and lacquer cracks).
On biomicroscopy, complete posterior vitreous detachment with a Weiss ring which was mobile in the vitreous cavity was obvious in 15 eyes. Posterior staphyloma was detected in 72 eyes. Greyish lesion at the macula was detected in 11 eyes.
Optical coherence tomography macular findings were recorded. ERM was present in 38 eyes. Vitreomacular traction (anteroposterior traction) was detected in 9 eyes. Macular retinoschisis was present in 18 eyes. Multiple columnar structures were seen widely within the retinoschisis as long straight highly reflective lines at the fovea and throughout the retinoschisis.
Lamellar macular hole was present in 10 eyes. Full-thickness macular hole was present in 4 eyes. Posterior sensory retinal detachment was present in 2 eyes. ILM detachment was present in 7 eyes. Dome shaped macula was detected in 5 eyes. Myopic CNV was present in 17 eyes. Atrophy of retinal layers was detected in 5 eyes.
OCT FINDINGS IN RELATON TO DEGREE OF MYOPIA
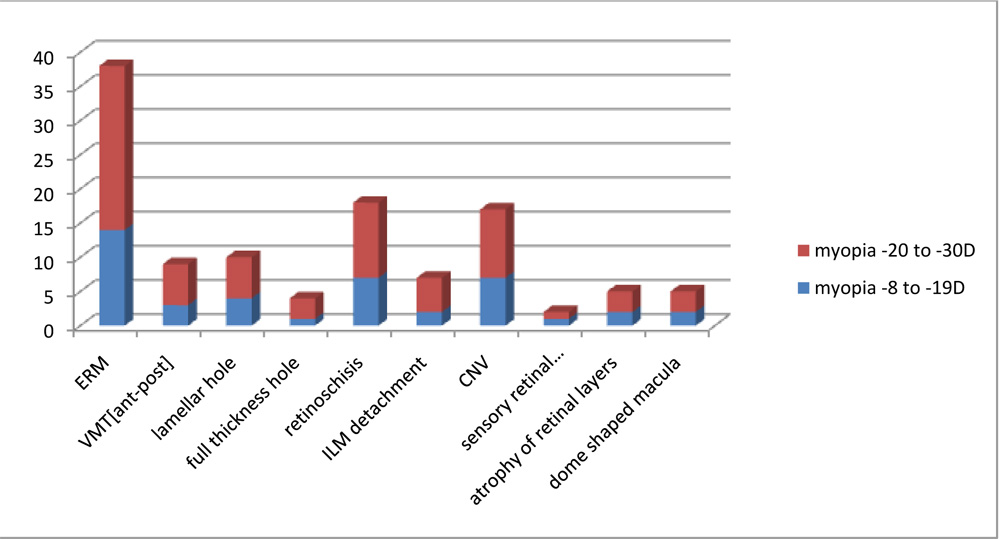
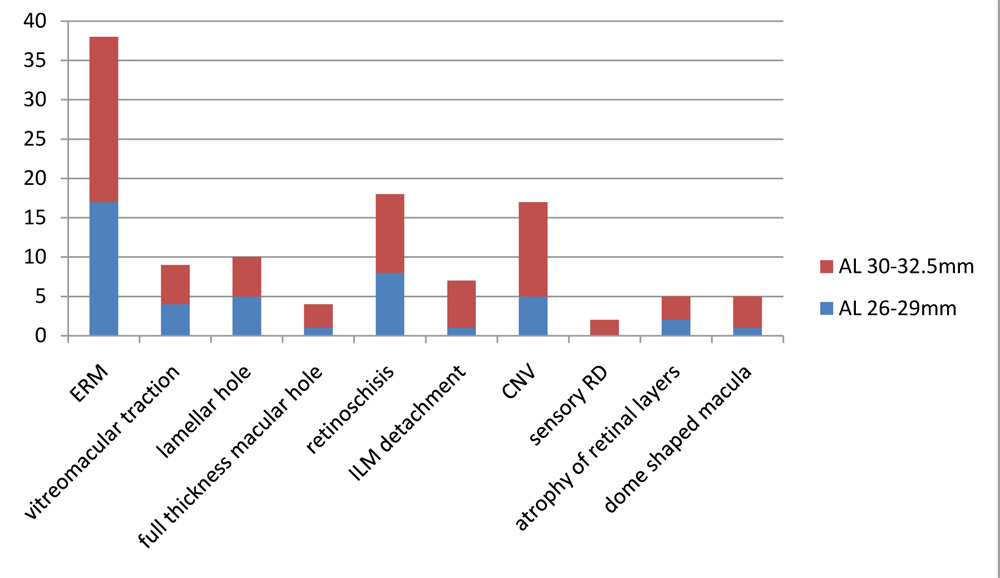 *ERM-epiretinal membrane, VMT-vitreomacular traction, ILM-internal limiting membrane, CNV-choroidal neovascularization
*ERM-epiretinal membrane, VMT-vitreomacular traction, ILM-internal limiting membrane, CNV-choroidal neovascularization
DISCUSSION:
Spectral domain (SD)-OCT has made it possible to discover macular changes which go unseen in conventional biomicroscopic examination and which give rise to unexplained low VA values.
In 2004, Panozzo and Mercanti first used the term ‘myopic traction maculopathy’ MTM to refer to these pathologies of the posterior pole in high myopia, such as macular retinoschisis, shallow retinal detachment without retinal holes, lamellar macular holes, and macular holes with or without retinal detachment. Tangential traction in the form of ERM was the most common finding in our study group.
In 2007, Panozzo suggested that the term ‘retinoschisis’ used in the literature for high myopic maculopathy was not appropriate as the term schisis suggested a complete separation between the retinal layers that resulted in the irreversible and total loss of retinal function. This retinal damage is not a schisis but rather is a retinal swelling.
Smiddy in 2009 supported this notion and suggested that myopic maculoschisis and vitreoschisis should all fall under the family of MTM.
Myopic foveoschisis[or macular retinoschisis] may remain stable for many years without affecting vision (Gaucher et al., 2007) ,however it can result in metamorphopsia and blurring of vision (Timothy, 2007) , vision worsening increases when there are associated premacular structures such as epiretinal membranes or a partially detached vitreous cortex (Johnny et al., 2010).
PATHOGENESIS OF ‘MTM’:
1) Thickening and stiffening of the internal limiting membrane (ILM), which can prevent the retina from adapting to high myopia posterior scleral staphyloma that produce centripetal traction and tangential traction to the retina.
2) choroidal atrophy, which can lead to the shortage in blood supply in the retina that reduces adhesion between the retinal layers.
3) the retinal pigment epithelium and choroid atrophy, thus reducing the adhesion between the retinal pigment epithelium and neuroretina.
4) Incomplete posterior vitreous detachment or adhesion between the remaining vitreous and the retina, which can produce centripetal traction and tangential traction to the retina.
5) One subretinal factor is the formation of posterior scleral staphyloma from the extension of the axis of the eye, which produces centrifugal traction on the retina.
Macular retinoschisis and lamellar macular holes occur in the early stages of MTM, while high myopic macular holes and sensory detachment occur in the advanced stages of MTM. Myopic macular retinoschisis is often stable for many years without progression, but it can progress to retinal detachment or macular hole. Development of a foveal retinal detachment in myopic macular retinoschisis impairs vision more severely. RD is an intermediate-stage change in development of FTMH in some MRS cases.
Enhanced SD-OCT images enabled improved visualization of the fine structures associated with macular retinoschisis, such as multiple columnar structures. Shimada et al. {11} suggested in a longitudinal study of five eyes with retinoschisis that progressed to a foveal retinal detachment using time-domain OCT that inward traction was transmitted to the outer retina through the foveal columnar structures in the retinoschisis layer.
Differentiation of ILM detachment from epiretinal membrane is based on the presence of columns that bridge the membrane to the retinal surface. It seems that tractional myopic maculopathy described by Panozzo in 2004 is a variation of longstanding tractional ILM detachment with loss of column bridges due to extreme traction and retinal atrophy. The cause of this detachment mainly thought to be due to inward tractional forces of retinal vasculature due to globe elongation. In this study we found 7 patients with ILM detachment all extrafoveal in location. ILM detachment in the superior and/or inferior peripheral macula is a sign of persistent strong traction that can cause a foveal retinal detachment, which is likely transmitted to the outer retina through the dense columnar structures in the retinoschisis at each retinal level.
Takano and Kishi {2} found a much higher prevalence of foveal retinal detachment, with 34% of highly myopic eyes with posterior staphyloma showing foveal retinal detachment or retinoschisis. However, the study group was much smaller, only 32 eyes, and also included pseudophakic patients, who are at a greater risk of retinal detachment following cataract surgery. In our study, we excluded pseudophakic cases and we had posterior sensory retinal detachment in only two eyes.
Choroidal neovascularization (CNV) secondary to pathological myopia is the leading cause of vision impairment in patients younger than 50 years. They are located in the subretinal space, as opposed to the sub-RPE space in AMD. Subretinal or sub-RPE fluid, hemorrhage or exudates are uncommon. Macular holes usually exist at the edge of the old CNV so periodic OCT examination is recommended. In our study there were 17 patients with CNV.
Based on OCT, a dome-shaped macula was first described by Gaucher et al.{13} as an unexpected finding in myopic staphyloma and was characterized as an inward convexity of the macula that occurred in highly myopic eyes within the concavity of a posterior staphyloma. They suggested that the dome-shaped macula may be the result of changes in choroidal thickness or to changes in scleral shape in highly myopic eyes. In our study, we found dome shaped macula in five eyes.
CONCLUSION:
In summary, in this large case series, we showed that retinal abnormalities in the macular area can be a frequent finding in eyes with degenerative myopia. Epiretinal traction probably plays a major role in this condition. Optical coherence tomography can greatly contribute in the examination of eyes with high myopia to detect early changes so should be routinely recommended for all high myopic patients.
REFERENCES:
1.Balacco-Gabrieli C. Aetiopathogenesis of degenerative myopia. A hypothesis. Ophthalmologica 1982
2.Takano M, Kishi S. Foveal retinoschisis and retinal detachment in severely myopic eyes with posterior staphyloma. Am J Ophthalmol
3.Panozzo G, Mercanti A. Optical coherence tomography findings in myopic traction maculopathy. Arch Ophthalmol 2004
4.Smiddy WE, Kim SS, Lujan BJ, Gregori G. Myopic traction maculopathy: spectral domain optical coherence tomographic imaging and a hypothesized mechanism. Ophthalmic Surg Lasers Imaging 2009
5.Panozzo G, Mercanti A. Vitrectomy for myopic traction maculopathy. Arch Ophthalmol 2007
6.Bando H, Ikuno Y, Choi JS, Tano Y, Yamanaka I, Ishibashi T. Ultrastructure of internal limiting membrane in myopic foveoschisis. Am J Ophthalmol 2005
7.Matsumura N, Ikuno Y, Tano Y. Posterior vitreous detachment and macular hole formation in myopic foveoschisis. Am J Ophthalmol 2004
8.Sayanagi K, Ikuno Y, Tano Y. Reoperation for persistent myopic foveoschisis after primary vitrectomy. Am J Ophthalmol 2006
9.Shimada N, Ohno-Matsui K, Baba T, Futagami S, Tokoro T, Mochizuki M. Natural course of macular retinoschisis in highly myopic eyes without macular hole or retinal detachment. Am J Ophthalmol 2006
10.Gaucher D, Haouchine B, Tadayoni R, Massin P, Erginay A, Benhamou N,et al.Long-term follow-up of high myopic foveoschisis: Natural course and surgical outcome. Am J Ophthalmol 2007
11.Shimada N, Ohno-Matsui K, Yoshida T, Futagami S, Tokoro T, Mochizuki M. Development of macular hole and macular retinoschisis in eyes with myopic choroidal neovascularization. Am J Ophthalmol 2008
12.Imamura Y, Iida T, Maruko I, Zweifel SA, Spaide RF. Enhanced depth imaging optical coherence tomography of the sclera in dome-shaped macula. Am J Ophthalmol 2011
13.Gaucher D, Erginay A, Lecleire-Collet A, Haouchine B, Puech M, Cohen SY, et al. Dome-shaped macula in eyes with myopic posterior staphyloma. Am J Ophthalmol 2008
14.Ikuno Y, Gomi F, Tano Y. Potent retinal arteriolar traction as a possible cause of myopic foveoschisis. Am J Ophthalmol 2005

