Dr. Amit Raj, R12161,Dr. Sanchi Vohra, Dr. Ravi Kant Bamotra, Dr.Sudesh Kumar Arya Dr. Amit Raj,Dr. Ravi Kant Bamotra,Dr.Sanchi Vohra and Dr. Sudesh Kumar Arya
INTRODUCTION
Hemifacial spasm as an entity was first described by Gowers in 1884. It represents a segmental myoclonus of muscles innervated by the facial nerve. Hemifacial spasm (HFS) generally begins with brief clonic movements of the orbicularis oculi and spreads over years to other facial muscles (corrugator, frontalis, orbicularis oris, platysma, zygomaticus).(1) These abnormal movements can be attributed to movement disorders such as myoclonus, dystonia, and other movement disorders restricted to the facial musculature. However, these could be due to compressive lesions around the facial nerve nucleus or it’s the proximal nerve segment such as vascular compression, it is now believed that most the previously considered “idiopathic “ HFS might well be attributed to aberrant blood vessels compressing the facial nerve within the cerebellopontine angle.(2) Other compressive lesions could include brainstem lesions such as stroke or multiple sclerosis plaques, mass lesions and secondary causes such as trauma or Bell’s Palsy.
Spontaneous hemifacial spasm manifests with facial spasms that represent myoclonic jerks and are analogous to segmental myoclonus, which may affect other body regions. Postparalytic hemifacial spasm, following facial nerve trauma such as Bells’s palsy, manifests as facial synkinesis and contracture.
Dry Eye and Hemifacial Spasm: The 2007 International Dry Eye WorkShop (DEWS) defined dry eye as a multifactorial disorder of the tear film and ocular surface that results in eye discomfort, visual disturbance and often ocular surface damage, characterized by hyperosmolarity of tear film and inflammation of ocular surface .(3)
The co-existence of HFS with Dry eyes has often been noticed clinically, however the “cause-effect” relationship continues to baffle. Dry eyes in patients of HFS is a challenging problem requiring adequate look-out for signs and symptoms. In this five year retrospective study of patients with Hemifacial spasm, we try to elucidate the clinical profile of these patients as well as the association with dry eyes.
Methods: Retrospective analysis of patients diagnosed with hemifacial spasm who reported to a tertiary eye care center in north India between January 2011 and December 2015 was carried out. Dry eyes, as well as all other local factors that can cause hemifacial spasm, were ruled out at intial presentation. Systemic evaluation was done to rule out any neurological disorder. MRI of the brain was done in all cases for any facial nerve compression or tumor involving posterior fossa. Botulinum A injections were given as and when required according to the guidelines.
Results: A data of 20 patients was reviewed, of which 7 were males and 13 were females; ratio being 1:1.8(Fig.2). The mean age of the patients was 49.5(±12.2) years and mean duration of symptoms before presentation was 8.9 (±6.3) months. MRI of one patient showed age related atrophic changes and another showed compression on the trigeminal nerve. Nine (45%) patients(Fig.3) were having associated dry eyes which were treated according to guidelines by DEWS 2007. Twelve patients required botulinim A injections which showed improvement in symptoms and disability score. 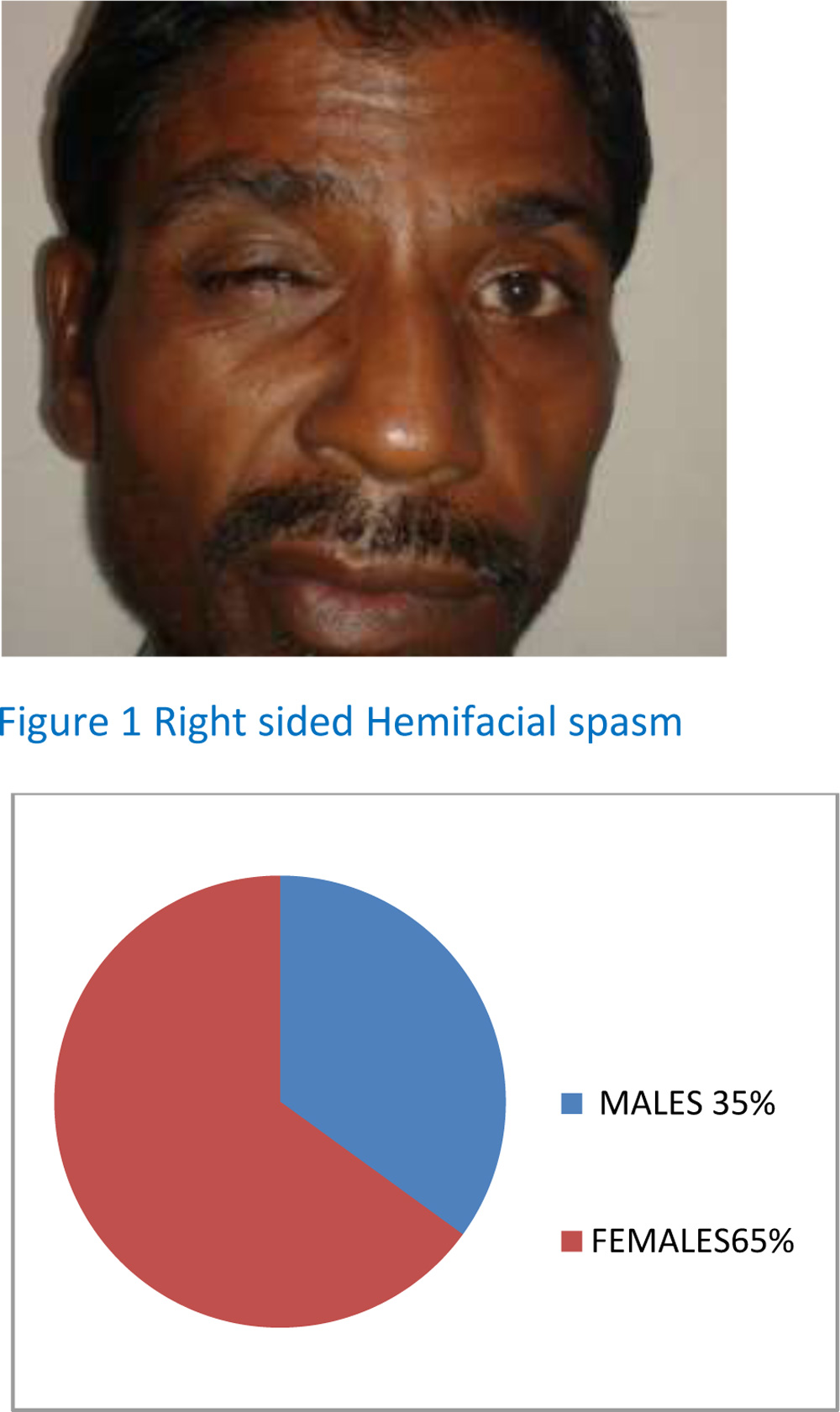
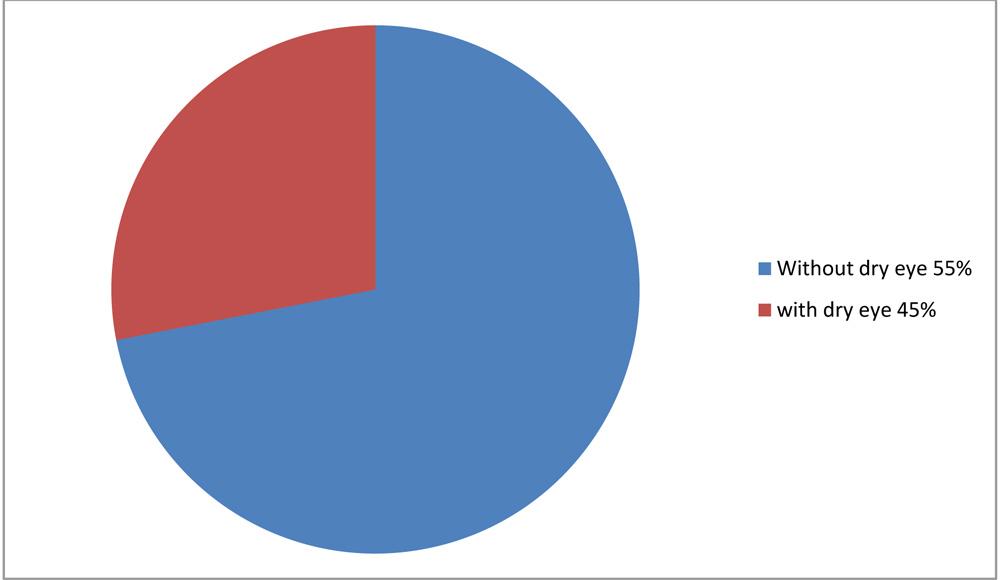 Figure 3 Pie chart showing percentage of patients with dry eyes
Figure 3 Pie chart showing percentage of patients with dry eyes
Discussion:
HFS characterized by involuntary unilateral contractions of the muscles innervated by the ipsilateral facial nerve, usually starting around the eyes before progressing inferiorly to the cheek, mouth, and neck has a prevalence of 9.8 per 100,000 persons.(4) The average age of onset for HFS is 44 years. Women and Asian populations have an increased susceptibility to HFS though valid prevalence data is scarce .(5,6) This issue is due to HFS underdiagnosis, misdiagnosis, and absence of population-based data. Worldwide estimates for the prevalence of HFS are 14.5 per 100,000 women and 7.4 per 100,000 men,in our analysis also 65% were females.
The etiology of HFS can be divided into two types: primary and secondary. Primary HFS is defined by vascular compression of the facial nerve root entry zone in the posterior fossa. Implicated arteries include the anterior inferior cerebellar artery (AICA), posterior inferior cerebellar artery (PICA), and vertebral artery (VA). Anatomic variations in vasculature such as lateral deviation of one or both vertebral arteries occurred on the ipsilateral side of HFS in 86.4% cases, making these variations a HFS risk factor.(7)
Secondary HFS occurs with damage anywhere along the facial nerve from the internal auditory canal to the stylomastoid foramen. Cases of secondary HFS have been linked to cerebellopontine angle (CPA) tumors and vascular malformations with other case linked to facial nerve trauma, demyelinating lesions.
Magnetic resonance imaging is the imaging study of choice to look for an underlying compressive lesion, whereas, Cerebral angiography or magnetic resonance angiography is performed prior to a vascular decompression surgical procedure.(8)
In Idiopathic or non-compressive HFS the treatment of choice is injection of botulinum toxin under electromyographic (EMG) guidance. The chemodenervation produced effectively treats most patients, spasm is relieved in 3-5 days after injection and lasts approximately for 6 months. Side effects of botulinum toxin injection (eg, facial asymmetry, ptosis, facial weakness) usually are transient.(9) Microvascular decompression surgery may be effective for those patients who do not respond to botulinum toxin.
Having been used since the early 1980s, Botulinum Toxin provides low-risk symptomatic relief in 85% of HFS patients, making it the treatment of choice. BoNT’s mechanism of action is to block calcium-mediated release of acetylcholine at the synaptic junction. Two serotypes are available: BoNT-A and BoNT-B, as well as four different commercial formulations: abobotulinumtoxinA, onabotulinumtoxinA, incobotulinumtoxinA, and rimabotulinumtoxinB.(10) After injection, BoNT is cleaved by trypsin into heavy and light chain components .At this point, the BoNT toxin is internalized into presynaptic nerve terminals, where the heavy chain binds synaptic vesicle protein 2, trisialoganglioside 1b, and synaptotagmin-1. The light chain then binds to the SNARE complex and cleaves target proteins such as synaptosomal-associated proteins of 25 kDa (SNAP-25) and synpatobrevin-2 to prevent exocytosis of neurotransmitters from the presynaptic terminal, leading to muscle paralysis.(11)
BoNT-A is the primary serotype used for HFS treatment.
Objective findings of Dry eye are usually less than the frequency of complaints in patients of HFS. Symptoms of ocular irritation, foreign body sensation, and blurred vision can be associated with dry eyes. Signs can include, ocular redness, reduced Tear film height, reduced Tear Break-up time (TBUT), tear film debris, etc. A comparative study comparing tear production in patients of Essential Blepharospam and HFS with control group suggested a significantly lower tear production in patients of Essential blepharospasm, However HFS patients did not have significantly different tear production than controls. (12). Another study evaluating the changes in corneal parameters following Botulinum toxin injection in patients of both Essential blepharospasm and HFS showed significant improvement in Tear film break up time(TBUT) following medical treatment.(13) So the unanswered question remains that what could be affecting the tear film and not the tear production in patients of HFS?
Considering that Blink characteristics determine the tear film stability, increasing blink amplitude thickens the lipid layer that overlies the aqueous layer of the tear film. This thickening reduces evaporation of the aqueous layer. Because each blink reforms the tear film, increasing blink frequency reduces tear film break-up time. Limited studies on animal models elucidate the same hypothesis, However human studies are yet awaited.
Conclusion:
Dry eye disease should be evaluated in all cases of hemifacial spasm and should be appropriately treated along with definitive treatment of spasms with botulinum toxin A.
1. Yaltho TC, Jankovic J. The many faces of hemifacial spasm: differential diagnosis of unilateral facial spasms.Mov Disord. 2011 Aug 1. 26(9):1582-92.
2.Campos-Benitez M, Kaufmann AM. Neurovascular compression findings in hemifacial spasm. J Neurosurg. 2008 Sep. 109(3):416-20.
3.Research in dry eye report of the Research Subcommittee of the International Dry Eye WorkShop (2007). Ocular Surf 2007; 5(2):179-93.
4.B. Nilsen, K.-D. Le, and E. Dietrichs, “Prevalence of hemifacial spasm in Oslo, Norway,” Neurology, vol. 63, no. 8, pp. 1532–1533, 2004.
5.Jankovic, “Peripherally induced movement disorders,”Neurologic Clinics, vol. 27, no. 3, pp. 821–832, 2009.
6.Wu, A. L. Davidson, T. Pan, and J. Jankovic, “Asian over-representation among patients with hemifacial spasm compared to patients with cranial-cervical dystonia,”Journal of the Neurological Sciences, vol. 298, no. 1-2, pp. 61–63, 2010.
7.-X. Guan, J. Zhu, and J. Zhong, “Correlation between idiopathic hemifacial spasm and the MRI characteristics of the vertebral artery,”Journal of Clinical Neuroscience, vol. 18, no. 4, pp. 528–530, 2011.
8.Rosenstengel, M. Matthes, J. Baldauf, S. Fleck, and H. Schroeder, “Hemifacial spasm: conservative and surgical treatment options,”Deutsches Ärzteblatt International, vol. 109, no. 41, pp. 667–673, 2012.
9.Rosenstengel, M. Matthes, J. Baldauf, S. Fleck, and H. Schroeder, “Hemifacial spasm: conservative and surgical treatment options,”Deutsches Ärzteblatt International, vol. 109, no. 41, pp. 667–673, 2012.
10.Esquenazi, A. Albanese, M. B. Chancellor et al., “Evidence-based review and assessment of botulinum neurotoxin for the treatment of adult spasticity in the upper motor neuron syndrome,”Toxicon, vol. 67, pp. 115–128, 2013.
11.R. Aoki, L. A. Smith, and M. Z. Atassi, “Mode of action of botulinum neurotoxins: current vaccination strategies and molecular immune recognition,”Critical Reviews in Immunology, vol. 30, no. 2, pp. 167–187, 2010.
12.Prince, Jane D.O.B.A.; O’Day, Justin F.R.A.C.O A. Comparative Study of Tear Secretion in Blepharospasm and Hemifacial Spasm Patients Treated with Botulinum Toxin. Journal of Clinical Neuro-Ophthalmology:March 1993.
13.Gunes, Alime; Demirci, Seden; Koyuncuoglu, Hasan Rifat; Tok, Levent; Tok, Ozlem. Corneal and Tear Film Changes After Botulinum Toxin-A in Blepharospasm or Hemifacial Spasm Cornea:August 2015 – Volume 34 – Issue 8 – p 906–910.

