Dr. Sanitha Sathyan, S12939, Dr. Meena Chembil Kakkitampara, Dr. Elizabeth Joseph
Sanitha Sathyan, Meena C.K, Elizabeth Joseph K
Little Flower Hospital and Research Centre, Angamaly
ABSTRACT:
AIM:
To compare the circumpapillary retinal nerve fibre layer thickness (RNFLT), optic nerve head parameters, macular parameters, and choroidal thickness between amblyopic and fellow eyes in patients between 5-15 years of age with unilateral strabismic amblyopia using stratus domain-optical coherence tomography (SD-OCT) with enhanced depth imaging (EDI).MATERIALS AND METHODS: This was a cross sectional observational study which included 48 unilateral strabismic amblyopes between 5-15 years using Optical Coherence Tomography.
RESULTS:
48 eyes of 48 patients were enrolled There was a statistically significant difference in the optic nerve head rim area (p=0.00, unpaired t-test), disc area (p=0.001, unpaired t-test) between the amblyopic and fellow eyes. There was no significant difference in the average RNFL thickness (p=0.34, unpaired t-test), average cup-disc ratio (p=0.45, unpaired t-test), cup volume (p=0.271, unpaired t-test), choroidal thickness (p=0.35, unpaired t-test) between the eyes. However, when analyzed separately for the type of strabismus, there was significant difference of RNFLT in all quadrants in the esotropia group, whereas no such significant difference was seen in the exotropia group. There was no significant difference in any of the macular parameters between the amblyopic eyes and fellow eyes.
CONCLUSION:
There were significant changes in optic disc area and rim area between the amblyopic eyes and the fellow eyes of children with strabismic amblyopia when analyzed using SD-OCT. Esotropic amblyopes demonstrated a significant change in circumpapillary retinal nerve fibre thickness in all quadrants. No change could be demonstrated in the macular and choroidal parameters.
BACKGROUND:
The neural sites that are affected by visual deprivation in amblyopia are still under investigation. The deleterious effect of amblyopia on the cell growth of the lateral geniculate body and damage on the ocular dominance columns in the striate cortex during the critical period of visual development have been established by histological studies in several animal species and in humans1,2. Most of the studies indicate that the lateral geniculate nucleus and the visual cortex are the structures primarily affected in amblyopia.
Whether histological changes occur in clinically normal retina/ optic nerve in strabismic amblyopes is still unanswered by the existing literature. Animal studies have failed to demonstrate significant structural changes in amblyopic retina and optic nerve as such3, 4. Prior electrophysiological studies demonstrate that the latencies of the event related visual evoked potentials were more variable in the amblyopic eye when compared to the fellow eye.5 Also, the pattern electroretinograms were found to be significantly reduced in various types of amblyopias.6 These indirect electrophysiological studies were suggestive of possible retinal and optic nerve changes in amblyopic eyes.
A study conducted by Ikeda etal7 in 1979, had documented retinal abnormalities in amblyopia patients, but a few other reports have rejected this opinion8, 9. However, in 2004, reinvestigations conducted by Lempert etal10, 11 using fundus photographs have suggested the presence of organic changes in the optic nerve head and retina of amblyopic eyes, which had previously been assumed to be normal. These controversies indicate that the current literature regarding the retinal and optic nerve involvement in amblyopia is rather inconclusive and the initial neural sites of the visual deficit in these eyes are still under investigation. However, it is logical to assume that retinal and optic nerve head changes can occur in amblyopic eyes as a result of retrograde degeneration or due to disuse. Since retina and optic nerve are the primary sites of visual input processing, these sites could be the initial sites where the neurophysiological changes occur in amblyopic eyes.
The development of spectral-domain OCT (SD-OCT) has allowed a more detailed assessment of pathologic changes of the retina, simulating histological sections. There were a few recent studies on the amblyopic retinal thickness using OCT.12-19Some of them report no differences in the retinal thickness between amblyopic and fellow eyes; while in some others, the amblyopic eyes were found to be either thicker or thinner than the fellow eyes. Most of these studies were marred with inadequate study designs, generalized analysis of various types of amblyopia together, inadequate sample size for subgroup analysis and confounding factors like high refractive errors, thereby making clinical interpretations difficult. Also, some of the studies were conducted using time-domain OCTs or older generation of stratus domain OCTS, leading to a less accurate measurement of macular and retinal nerve fibre layer abnormalities. Therefore, a consensus has not yet been obtained even on OCT based studies, and the degree of retinal and optic nerve involvement in amblyopia remains elusive. There were no SD-OCT studies exclusively done on strabismic amblyopes; and no studies comprehensively evaluating the macular, optic nerve head, RNFL and choroidal changes in strabismic amblyopia.
Hence this study was done to evaluate and quantify the retinal nerve fibre layer, optic nerve head, macular and choroidal parameters in strabismic amblyopes between 5-15 years of age using spectral domain OCT with enhanced depth imaging.
AIM:
To compare the circumpapillary retinal nerve fibre layer thickness (RNFLT), optic nerve head parameters, macular parameters, and choroidal thickness between amblyopic and fellow eyes in patients between 5-15 years of age with unilateral strabismic amblyopia using stratus domain-optical coherence tomography (SD-OCT) with enhanced depth imaging (EDI).
MATERIALS AND METHODS:
This was a cross sectional observational study which included 48 unilateral strabismic amblyopes between 5-15 years, enrolled through simple random sampling, who presented to the Pediatric Ophthalmology unit of a tertiary care eye facility between June, 2015 and June, 2016. Institutional review board approval and informed written consent from the parents of the patients was obtained prior to the study.
Strabismic amblyopia was defined as reduction in best corrected visual acuity of at least 2 lines in the Snellen chart, which was not, associated with refractive error or sensory deprivation, with horizontal strabismus more than 12 prism diopters on prism bar cover testing.
Inclusion criteria: Unilateral strabismic amblyopic patients between 5-15 years of age enrolled through simple random sampling. Exclusion criteria: Patients with neurological diseases or ocular diseases such as refractive error, glaucoma, nystagmus, retinal pathology, patients who were too young to cooperate, and patients in whom pupillary dilation was not sufficient to obtain accurate OCT recordings were excluded from the study. Isolated vertical strabismus, incomitant strabismus and microtropias were also excluded.
All the patients underwent baseline examinations including visual acuity (Snellen visual acuity chart), subjective refraction, sensory tests for strabismus, ocular motility test, prism bar cover test, slit lamp examination, cycloplegic refraction, and dilated fundus examination.
To measure the parameters of the macula, choroid, optic nerve head and retinal nerve fiber layer, Optical Coherence Tomography from Zeiss (Cirrus HD-OCT-Model:5000 OCT version 6.5.0.772, Zeiss Humphrey, Dublin, USA) was used. This machine has a scanning speed of 27,000 to 68,0003 A-scans/sec, axial resolution of 5µm and fundus image field of view of 36 x 30 degrees.
The measurements were conducted by a single experienced examiner (third author), who was masked to the patients clinical details, under the manufacturer prescribed conditions, on dilated pupils.
To measure the macular thickness, the examiner focused a scanning beam on the fovea by inspecting the fundus with an infrared sensing camera. Subsequently, the images were obtained by performing a macula scan. The macula scan consisted of 6 radial scans that had the maximal 1,024 pixels focused on the fovea, and each scan was rotated by a 30 degree interval. Using the images obtained by the 6 scans, the macular thickness map equivalent to a central 20 degrees of vision (a 6-mm diameter map) or to a central 10 degrees of vision (a 3.45-mm diameter map) was then prepared for each eye. The thick areas of the retina appeared as bright colors (white and red) and the thin areas appeared as dark colors (blue and black). The map of the macular thickness composed of three concentric circles: a central circle, an inner ring and an outer ring. In the case of the 6-mm diameter map, the diameters of the concentric circles were 1 mm, 3 mm, and 6 mm, and in the case of the 3.45-mm diameter map, the diameters of the concentric circles were 1 mm, 2.22 mm and 3.45 mm. In addition, each concentric circle was divided again into 4 quadrants and this is shown as 9 sections. The location of the vitreoretinal interface and the retinal pigment epithelium defined the inner and outer boundaries. These two boundaries were generated by the high contrast in the optical reflectivity between the relatively non reflective vitreous and the reflective neurosensory retina and between the minimally reflective photoreceptor outer segments and the highly reflectively retinal pigment epithelium. The distance between photoreceptor outer segments and retinal pigment epithelium was defined as the thickness of retina.
RNFL scan was performed with three peripapillary scans with the protocol of having diameter of 3.4 mm centered on the optic disc. Choroidal thickness was measured as the distance between the outer limit of the hyper-reflective band representing the retinal pigment epithelium (RPE) and Bruch’s membrane complex according to the guidelines given by Boonarpha et al20. All children were measured three times, and their average values were obtained.
Statistical analysis:
SPSS software version 21.0 (SPSS, Inc., Chicago, IL) was used for statistical analyses. The Shapiro-Wilk test was used to examine the distribution of the numerical data. Statistical analysis for the comparison of the macular, optic nerve head, retinal nerve fiber layer and choroidal parameters between the amblyopic eye and fellow eye was performed using unpaired t- test. Effect size was measured using Cohen’s d- test. A p-value less than 0.05 was considered to be statistically significant. Snellen visual acuity measurements were converted into logarithm of the minimum angle of resolution (log MAR) units for the statistical analysis.
RESULTS:
48 eyes of 48 patients between 5-15 years with unilateral strabismic amblyopia were enrolled in this study between June, 2015 and June, 2016. The normal fellow eyes of the same patients were taken as control. Mean age of the patients was 9.63 ± 3.00 years; 28 were females, and 20 were males. 35 had esotropia, 13 had exotropia. The mean best corrected visual acuity of the amblyopic eye was 0.6±0.2 log MAR units. All the normal fellow eyes had log MAR visual acuity of zero. Demographic characteristics of the population are listed in Table 1.
Table:1 Demographic characteristics of the population:
| PARAMETER | FREQUENCY |
| Age | 9.63 ± 3.0 years
Range: 5-15 years |
| Sex distribution
Females: Males: |
28 (58.33%) 20 (41.67%) |
| Laterality
Right eye: Left eye: |
18 (37.5%) 30 (62.5%) |
| Esotropia | 35 (72.91%) |
| Exotropias | 13 (27.08%) |
| BCVA of the amblyopic eye | 0.6±0.2 log MAR
Range: 0.4-2 logMAR |
In our study, there was a statistically significant difference in the optic nerve head rim area (p=0.00, unpaired t-test) and disc area (p=0.001, unpaired t-test) between the amblyopic and fellow eyes. There was no significant difference in the average cup-disc ratio (p=0.45, unpaired t-test) and cup volume (p=0.271, unpaired t-test) between the eyes. There was no significant difference between the two groups in terms of choroidal thickness (p=0.35, unpaired t-test). Table: 2 shows the ONH parameters, RNFL parameters and choroidal thickness between the amblyopic and fellow eyes.
Table-2: Comparison of ONH, RNFL parameters and choroidal thickness between amblyopic and fellow eyes.
| Parameter | Amblyopic eye | Fellow eye | T -value | p-value | Cohens d |
| AVERAGE RNFL THICKNESS (µm) | 95.67±12.37 | 96.41±11.11 | 0.42 | 0.34
Not significant |
– |
| SYMMETRY PERCENTAGE | 82±11.28
|
||||
| RIM AREA (mm2) | 1.35±0.33 | 1.55±0.31 | 3.19 | 0.000
significant |
0.03 |
| DISC AREA (mm2) | 1.76±0.37 | 1.96±0.37 | 3.02 | 0.001
significant |
0.11
|
| AVERAGE CD RATIO | 0.41±0.19 | 0.38±0.18 | 0.05 | 0.45
Not significant |
– |
| CUP VOLUME (mm3) | 0.10±0.12 | 0.09±0.01 | -0.61 | 0.271
Not significant |
– |
| CHOROIDAL THICKNESS (µm) | 301±62.22 | 298±72.01 | -0.38 | 0.35
Not significant |
– |
Fig-1: Comparison of optic disc parametres between the amblyopic eye and the fellow eye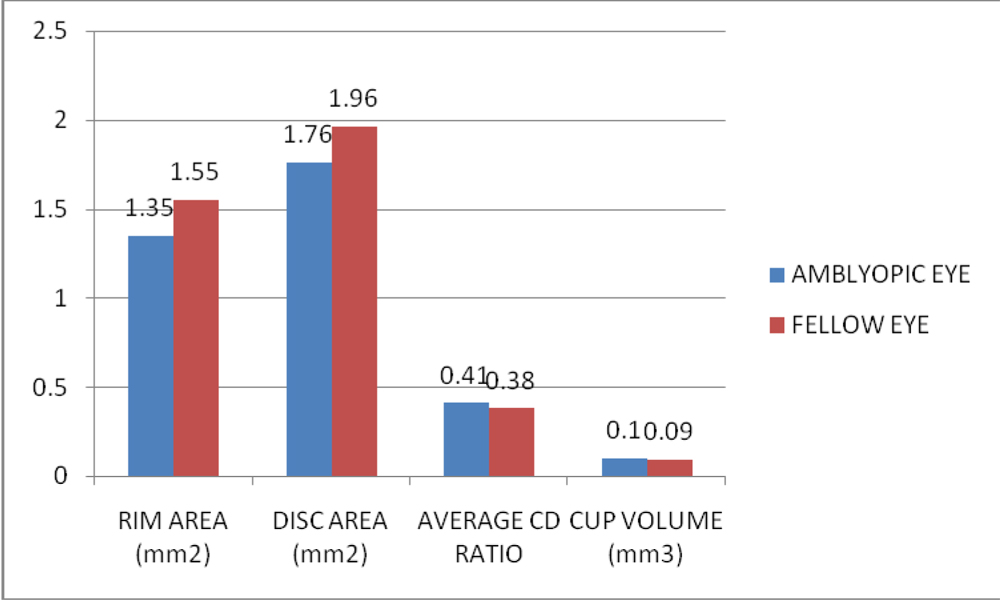 Quadrant-wise comparison of RNFL did not show a significant difference between the amblyopic eye and the fellow eye in any quadrant. However, there was no significant difference in the average RNFL thickness all four quadrants when taken together (p=0.34, unpaired t-test).
Quadrant-wise comparison of RNFL did not show a significant difference between the amblyopic eye and the fellow eye in any quadrant. However, there was no significant difference in the average RNFL thickness all four quadrants when taken together (p=0.34, unpaired t-test).
Fig-2:Comparison of circumpapillary quadrant-wise RNFL thickness between the amblyopic eye and the fellow eye
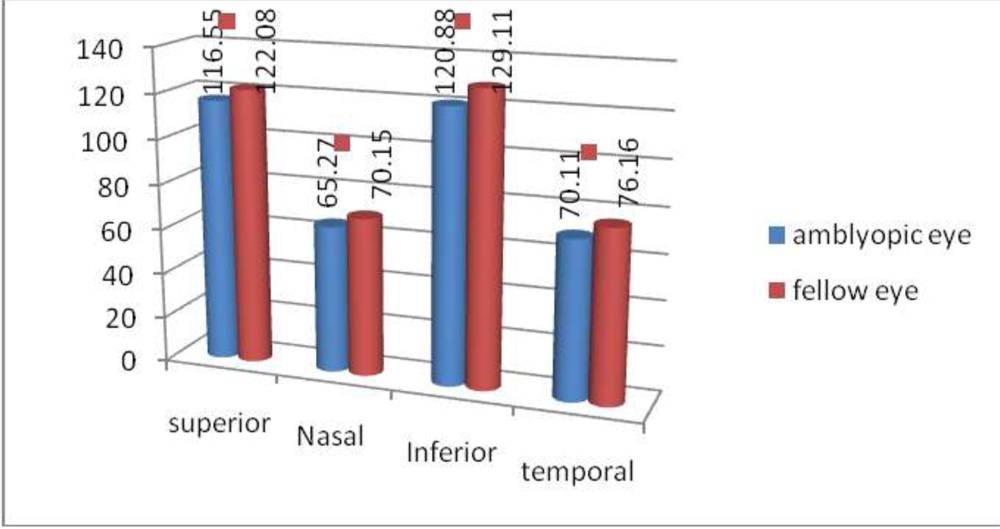
However, when analyzed separately for the type of strabismus, there was significant difference of RNFLT in all quadrants in the esotropia group, whereas no such significant difference was seen in the exotropia group.
Table:3 Comparisons of RNFLT between amblyopic and fellow eyes in exotropes and esotropes.
Exotropic amblyopes:
| RNFL quadrant | Amblyopic eye | Fellow eye | t-value | p-value | Significance level | |
| Superior |
|
119.08±17.70 | 0.44 | 0.33 | Not significant | |
| Nasal | 75.67±15.31 | 71.75±14.36 | 1.31 | 0.09 | Not significant | |
| Inferior | 128.88±22.43 | 127.61±22.43 | 0.82 | 0.20 | Not significant | |
| Temporal | 72.62±16.09 | 70.36±16.20 | 0.69 | 0.25 | Not significant |
Esotropic amblyopes:
| RNFL quadrant | Amblyopic eye | Fellow eye | t-value | p-value | Cohens-d | Significance level | |
| Superior |
|
122.08±15.72 | 1.64 | 0.03 | 0.04 | significant | |
| Nasal | 65.27±13.81 | 70.15±12.35 | 1.71 | 0.05 | 0.03 | significant | |
| Inferior | 120.88±12.40 | 129.11±20.49 | 1.80 | 0.04 | 0.03 | significant | |
| Temporal | 70.11±12.04 | 76.16±14.98 | 1.93 | 0.02 | 0.02 | significant |
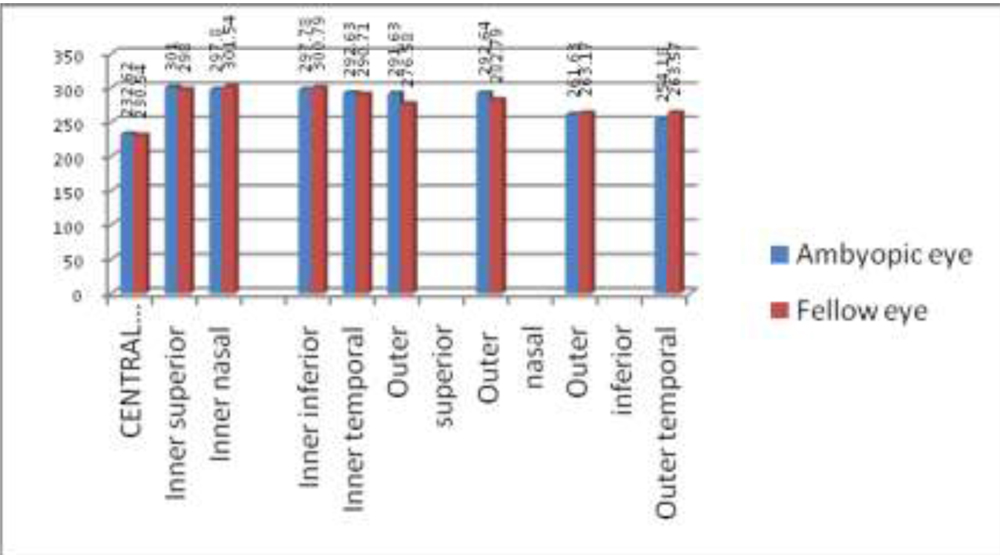
There was no significant difference in any of the macular parameters between the amblyopic eyes and fellow eyes. Following table represents the comparison of macular parameters between the groups.
Table-4: Comparison of Macular Parameters between the Amblyopic Eye and the Fellow Eye
| PARAMETER | AMBYOPIC EYE | FELLOW EYE | t-VALUE | p-VALUE | Level of significance |
| CENTRAL THICKNESS | 232.62±29.80 | 230.54±24.81 | 0.04 | 0.48 | Not significant |
| Volume cube | 9.67±0.70 | 9.82±0.69 | -1.72 | 0.44 | Not significant |
| Average thickness | 268.33±19.21 | 273.167±19.20 | -1.25 | 0.11 | Not significant |
| Inner superior | 301.00±26.20 | 298±24.81 | 1.67 | 0.44
|
Not significant |
| Inner nasal | 297.80±23.03 | 301.54±20.99 | -1.27 | 0.11 | Not significant |
| Inner inferior | 297.78±28.29 | 300.79±18.20 | -0.6 | 0.27 | Not significant |
| Inner temporal | 292.63±23.26 | 290.71±21.13 | 1.41 | 0.03
|
Not significant |
| Outer
superior |
291.63±24.61 | 276.58±24.03 | -0.11 | 0.46 | Not significant |
| Outer
Nasal |
292.64±27.33 | 282.79±26.63 | 1.62 | 0.54 | Not significant |
| Outer
Inferior |
261.63±21.39 | 263.17±17.20 | -0.41 | 0.34 | Not significant |
| Outer temporal | 254.18±24.36 | 263.57±31.09 | -1.51 | 0.67 | Not significant |
Fig-3: Bar chart comparing macular parametres between the amblyopic eye and the fellow eye
 DISCUSSION:
DISCUSSION:
In our study, the parameters which differed significantly in the amblyopic and fellow eyes were optic nerve head rim area (p=0.00) and disc area (p=0.001). These observations point towards a possible optic nerve pathology in amblyopic eye of patients with strabismic amblyopia. This is in concordance with the reports by Lempert etal10,11 who noted changes in optic nerve morphology in amblyopes from fundus photographs. Previous histological evidences given by Von Noorden etal1 have demonstrated changes in the lateral geniculate body of monkeys with amblyopia and with strabismic amblyopia in particular2. However psychophysical studies done by Chaw etal3 failed to observe such changes in monkeys reared in dark. But there were no previous SD-OCT based studies demonstrating changes optic nerve head in strabismic amblyopes. Our results are in agreement with the data of Araki etal16, who observed significantly larger rim area and smaller cup-disc ratios in amblyopic eyes. But their study population included strabismic, anisometropic, and mixed type amblyopic eyes and there was inadequate sample size for subgroup analysis.
Table:5 Comparison of Previous Studies on Amblyopia Using Optical Coherence Tomography
| NO | STUDY | YEAR | DESIGN | TYPE OF AMBLYOPIA | SAMPLE SIZE | OCT USED | PARAMETERS ANALYSED | CONCLUSIONS | COMMENTS |
| 1 | Yalcin et al19 | 2014 | Cross sectional, comparative
Amblyopes versus emmetropes |
Hypermetropic anisometropic amblyopia | 30 patients with hypermetropic anisometropic amblyopia versus 30 emmetropes | Time-domain OCT (OCT 3000, version A 3.0; Carl Zeiss, Inc, Dublin, CA, USA) | RNFL, Fovea | RNFL thickness not significant; mean foveal thickness thicker in amblyopic eyes | Time domain OCT used,
Cannot be extrapolated to strabismic amblyopia |
| 2 | Andalib et al18 | 2013 | Cross sectional, comparative
Amblyopic versus fellow eyes |
Anisometropic and strabismic | 30 adults | NIDEK RS-3000 SLO spectral domain OCT | RNFL
Macular |
No difference in macular peripapillary RNFL, or optic disc structures | No comparison group,
Cannot be extrapolated to children |
| 4 | Araki
et al17 |
2014 | Cross sectional, comparative
Amblyopic versus fellow eyes |
strabismic, anisometropic or combined | 21 | RTVue-100 | RNFL
ONH Macular |
RNFL thicker, macular outer retinal: thicker macular inner retinal thickness, ONH: not different | Small sample size
No subgroup analysis for different types of amblyopia |
| 5 | Firat
et al15
|
2012 | Cross sectional, comparative
Amblyopes v/s emmetropes |
strabismic, anisometropic or combined | 32 | RS-3000 OCT Retina scan, Nidek | RNFL
Macular |
No difference | Heterogenous
groups were compared |
| 6 | Dickmann et al14
|
2012 | Cross sectional, comparative
|
Strabismic, anisometropic or combined | 38 | time-domain OCT device, and a custom-built OCT image analysis software (OCTRIMA) | Macular | Subtle changes in amblyopic eyes affecting the outer nuclear layer of the fovea suggesting the possible involvement of the photoreceptors. | Sample size inadequate
for subgroup analysis Not studied about RNFL or choroid Time domain OCT used |
| 7 | Park et al13
|
2011 | Pilot study
Cross sectional, comparative
|
Strabismic, anisometropic or combined
|
20 | Zeiss (SD-OCT version 4.0) | Ganglion cell layer, macula | Thickness of ganglion cell layer and inner plexiform layer different. | Small sample size
No subgroup analysis for different types of amblyopia |
| 8 | Al-Haddad et al12
|
2011 | Cross sectional, comparative | Strabismic, anisometropic or combined | 20 | Zeiss (SD-OCT version 4.0) | RNFL, macular | Central macular thickness was significantly increased in anisometropic amblyopia using SD-OCT. | Small sample size,
older version of OCT |
| 9 | Se-Young Kee et.al 23 | 2006 | Cross sectional, comparative
Amblyopes versus emmetropes |
anisometropia, strabismus, or combined | 26 amblyopes versus
42 normal children |
Zeiss (OCT version 3.0) | RNFL, Macular | no differences RNFL and fovea | Heterogenous groups,
no subgroup analysis older version of OCT |
| 10 | Repka et al22 | 2009 | Cross-sectional, comparative
Amblyopic versus fellow eyes |
strabismic, anisometropic or combined | 37 | Humphrey-Zeiss Stratus (OCT3) software 4.0.1 | RNFL | No difference between amblyopic and fellow eyes | Heterogenous groups,
no subgroup analysis older version of OCT |
| 11 | Abdullah G21 | 2009 | Cross sectional observational | strabismic, anisometropic or combined | 93 | Type of OCT used: not mentioned | Macular
RNFL |
Thicker RNFL in amblyopia, no difference in macular thickness | Heterogenous groups,
no subgroup analysis older version of OCT |
| 12 | Andrea Szigeti24 | 2014 | Cross sectional | Strabismic, anisometropic or combined | 38 | Stratus OCT device (Carl Zeiss Meditec, Dublin, CA), | RNFL
Macular |
subtle changes in amblyopic eyes affecting the outer nuclear layer of the fovea | Heterogenous groups,
no subgroup analysis older version of OCT |
| 13 | May-Yung Yen24 | 2004 | Cross sectional | Strabismic, anisometropic or combined | 38 | OCT model 2000 (Carl Zeiss version was 5.1 | RNFL | RNFL thicker in amblyopia | Heterogenous groups,
no subgroup analysis older version of OCT |
| 14 | Aygit ED25 | 2015 | Cross sectional, comparative | anisometropic | 40 | SD-OCT VERSION:4.1 | Choroidal thickness | S
ubfoveal choroid of eyes with anisometropic and strabismic amblyopia is significantly thicker than that of the fellow eye and the age-matched controls. |
RNFL, macular details:
not analyZed |
Quadrant wise analysis of circumpapillary RNFL thickness showed significant difference only in those strabismic amblyopes with esotropia and no significant difference could be demonstrated for exotropic patients. This may be because the exotropias tend to produce less severe degree of amblyopia than esotropias, which could not be detected by the SD-OCTs. Also, esotropias are known to cause amblyopia at a much earlier age than exotropias. It has been well acknowledged that visual deprivation and abnormal binocular interactions have the potential to cause more damage to the visual system during the earliest period of visual development. This could have been the cause for the isolated involvement in esotropic amblyopes in our study. Our findings indicate that peripapillary RNFL thickness is thinner in eyes with moderate to severe strabismic amblyopia in esotropes, compared with their normal fellow eyes. In an earlier study, Xu et al17, failed to find a significant difference in foveal or RNFL thickness in 21 children with unilateral esotropic amblyopia. Our findings differ from the above mentioned study.
Amblyopia occurs during the period when the neuronal network between the retina and the cerebral cortex is developing and maturing. Slowing down of normal post-natal reduction of neurons have been proposed as the probable mechanism for increased RNFL thickness found in some studies12, 13. However, we choose to differ from this observation as our sample also included strabismic amblyopes with late onset exotropias. It is unlikely that the post natal reduction was continuing to produce increased thickness of the RNFL even in such late onset strabismus amblyopia. We therefore propose that the mechanism of Wallerian degeneration may have left its trails even at the level of optic nerve head and retinal ganglion cell layer, resulting in reduced thinness of RNFL in strabismic amblyopic eyes when compared to the normal fellow eyes.
An indirect proof of this association comes from the works of Lembert etal10, 11, who rediscovered through photographic analysis that optic disc changes were present in amblyopic eyes in the form of reduced disc size and volume. In our study, we also found that the disc area and disc volume were significantly reduced in amblyopic eyes, providing supportive evidence to the loss of RNFL subsequent to a possible retrograde degeneration.
However no such differences were reflected on the macular and choroidal parameters. The absence of macular and choroidal changes indicate that optic nerve pathology alone is the basis for anterior changes in amblyopia and macula remains relatively unaffected even in severe strabismic amblyopia. Several studies conducted in anisometropic amblyopes12, 18 refute this and report a significant change in macular parameters as well. But the finding in those studies should be interpreted with caution. This is because refractive error alone can change the macular parameters as in high myopia or astigmatism associated with amblyopia. In a pilot study conducted in our centre, we also had observed a change in macular thickness in anisometropic amblyopias and ametropic amblyopias. But whether the change was brought about by amblyopia or refractive error per se could not be explained.
Merits of our study include a relatively comparable population in terms of age, comparison with normal fellow eye of the same patients, relatively good sample size, inclusion of moderate to severe amblyopia only and comparison with multiple parameters of macula, choroid and RNFL. We chose to study strabismic amblyopia alone as high anisometropic amblyopes are known to produce disc and peripapillary retinal changes like staphyloma, tilted disc which could interfere with accurate measurements of RNFL parameters using SD-OCT. Limitations of our study include lack of information about the exact age of onset of strabismus and inclusion of cases with previous occlusion therapy for amblyopia.
CONCLUSION:
There was significant reduction in optic disc area and rim area between the amblyopic eyes and the fellow eyes of children with strabismic amblyopia when analyzed using SD-OCT with EDI. Esotropic amblyopes demonstrated a significant reduction in circumpapillary retinal nerve fibre thickness in all quadrants also. However, no difference in RNFL thickness was seen in exotopic amblyopes. No change could be demonstrated in the macular and choroidal parameters in any of the groups. Our findings indicate that reduction in optic nerve area and thinning of circumpapillary RNFL was evident in strabismic amblyopic eyes when compared to the normal fellow eyes. Therefore, it can be concluded that in addition to the previous histological changes obtained from lateral geniculate body and visual cortex, optic nerve and peripapillary retinal nerve fibers are also selectively affected in neuronal damage due to amblyopia. The primary sites of injury and mechanism of axonal damage need to be ascertained through further histological and molecular level studies.
REFERENCES:
1.Von Noorden GK (1973) Histological studies of the visual system in monkeys with experimental amblyopia. Invest Ophthalmol 12: 727–738.
2.Von Noorden GK, Crawford ML (1992) The lateral geniculate nucleus in human strabismic amblyopia. Invest Ophthalmol Vis Sci 33: 2729–2732.
3.Chow KL (1955) Failure to demonstrate changes in the visual system of monkeys kept in darkness or in colored lights. J Comp Neurol 102: 597–606.
4.Rasch E, Swift H, Riesen AH, Chow KL (1961) Altered structure and composition of retinal cells in dark reared mammals. Exp Cell Res 25: 348–363.
5.Banko EM, Kortvelyes J, Nemeth J, Weiss B, Vidnyanszky Z (2013)Amblyopic deficits in the timing and strength of visual cortical responses to faces. Cortex49: 1013–1024.
6.Arden GB, Wooding SL (1985) Pattern ERG in amblyopia. Invest Ophthalmol Vis Sci 26: 88–96.
7.Ikeda H, Tremain KE. Amblyopia occurs in retinal ganglion cells in cats reared with convergent squint without alternating fixation. Exp Brain Res. 1979;35(3):559–582.
8.Von Noorden GK, Crawford ML, Middleditch PR. Effect of lid suture on retinal ganglion cells in Macaca mulatta. Brain Res. 1977;122(3):437–444.Hess RF. Amblyopia: site unseen. Clin Exp Optom. 2001;84(6):321–336.
9..Lempert P. Optic nerve hypoplasia and small eyes in presumed amblyopia. J AAPOS. 2000;4(5):258–266.
10.Lempert P. The axial length/disc area ratio in anisometropic hyperopic amblyopia: a hypothesis for decreased unilateral vision associated with hyperopic anisometropia. Ophthalmology. 2004;111(2):304–308.
11.Al-Haddad CE, El Mollayess GM, Cherfan CG, Jaafar DF, Bashshur ZF. Retinal nerve fiber layer and macular thickness in amblyopia as measured by spectral-domain optical coherence tomography. Br J Ophthalmol. 2011;95(12):1696–1699.
12.Park KA, Park DY, Oh SY. Analysis of spectral-domain optical coherence tomography measurements in amblyopia: a pilot study. Br J Ophthalmol. 2011;95(12):1700–1706.
13. Dickmann A, Petroni S, Perrotta V, et al. Measurement of retinal nerve fiber layer thickness, macular thickness, and foveal volume in amblyopic eyes using spectral-domain optical coherence tomography. 3 AAPOS. 2012;16(1):86–88.
14.Firat PG, Ozsoy E, Demirel S, Cumurcu T, Gunduz A. Evaluation of peripapillary retinal nerve fiber layer, macula and ganglion cell thickness in amblyopia using spectral optical coherence tomography. Int J Ophthalmol. 2013;6(1):90–94.
15.S. Araki, A. Miki, T. Yamashita et al., “A comparison between amblyopic and fellow eyes in unilateral amblyopia usingspectral-domain optical coherence tomography,” Clinical Ophthalmology,vol. 7, no. 8, pp. 2199–2207, 2014.
16.J. Xu, F. Lu,W. Liu, F. Zhang,W. Chen, and J. Chen, “Retinalnerve fibre layer thickness and macular thickness in patientswith esotropic amblyopia,” Clinical and Experimental Optometry,vol. 96, no. 3, pp. 267–271, 2013.
17.D.Andalib, A. Javadzadeh, R. Nabai, and Y. Amizadeh,“Macular and retinal nerve fiber layer thickness in unilateral anisometropic or strabismic amblyopia,” Journal of PediatricOphthalmology and Strabismus, vol. 50, no. 4, pp. 218–221, 2013.
18.E.Yalcin and O. Balci, “Peripapillary retinal nerve fiber layerand foveal thickness in hypermetropic anisometropic amblyopia,”Clinical Ophthalmology, vol. 12, no. 8, pp. 749–753, 2014.
19.Nattapon Boonarpha,Yalin Zheng,Alexandros N. Stangos, Huiqi Lu,Ankur Raj,Gabriela Czanner et al Standardization of choroidal thickness measurements using enhanced depth imaging optical coherence tomography. Int Journal Ophthalmology, 2015; 8(3): 484–491.
20.Abdullah G. Alotaibiand Badriah Al Enazi Unilateral amblyopia: Optical coherence tomography findings; Saudi J Ophthalmology 2011 Oct; 25(4): 405–409.
21.Michael X. Repka, Raymond T. Kraker, Susanna M. Tamkins, Donny W. Suh, Nicholas A. Sala, and Roy W. Beck, for the Pediatric Eye Disease Investigator Group Retinal Nerve Fiber Layer Thickness in Amblyopic EyesAm J Ophthalmol. 2009 Jul; 148(1): 143–147.
22.Se-Young Kee, Se-Youp Lee, and Young-Chun Lee, Thicknesses of the Fovea and Retinal Nerve Fiber Layer in Amblyopic and Normal Eyes in Children;2006 Sep; 20(3): 177–181.
25.Andrea Szigeti, Erika Tátrai, Anna Szamosi, Péter Vargha, Zoltán Zsolt 3N3gy. A Morphological Study of Retinal Changes in Unilateral Amblyopia Using Optical Coherence Tomography Image Segmentation PLoS ONE, 2014; 9(2): e88363.
26.May–Yung Yen; An-Guor Wang; Yi-Ling Yeh; An-Lo Lin, Retinal Nerve Fibre 4Thickness in Unilateral Amblyopia. Investigative Ophthalmology and Visual Sciences, 2004 45(7) 2224-2230.
25.Aygit ED, Yilmaz I, Ozkaya A, Alkin Z, Gokyigit B, Yazici AT, Demirok A. Choroidal thickness of children’s eyes with anisometropic and strabismic amblyopia.2015 Jun;19(3):237-41.

