Dr. Sowmya S. Bhat, S15458, Dr. Sri Ganesh, Dr. Sheetal Brar
INTRODUCTION
The term “presbyopia” derives from Greek for “old eyes”1 and refers to the age-related loss of natural accommodation and resulting reduction of baseline near vision around the age of 40 years.As the world population becomes older, presbyopia prevalence will increase.The world population aged 40 years or over is expected to increase by up to 41%, by 20302-3.The effective treatment of presbyopia combined with any refractive error has proven to be a significant challenge for refractive surgeons. The principles used for monovision contact lenses have been applied to corneal refractive surgery using Laser in-situ Keratomileusis4,5 (LASIK) or Photo Refractive Keratectomy (PRK) techniques6.Similar limitations as with monovision contact lenseswere found to occur inmonovision induced by refractive surgery, including loss of fusion and stereoacuity7. Multifocal corneal ablation profiles have also been tried; Though an improvement in visual acuity for both near and distance vision were recorded, the efficacy remained relatively low8-10and safety and quality of vision were compromised11,12.There are also other techniques for the correction of presbyopia: multifocal lens, accommodating lens, femtosecond laser13or intracorneal implant14,15. A better solution offering improved visual results and greater tolerance is still needed.
Laser Blended Vision (LBV) is a procedure whichinvolves controlled induction of spherical aberration to increase the depth of field and Micro-monovisionto treat presbyopia in emmetropic, myopic and hyperopic patients. It is known that one way of increasing the depth of field is to increase the amount of corneal spherical aberration, which could compromise the visual quality and contrast sensitivity16,17,18.However, it was discovered that the depth of field of the cornea can be safely increased to 1.50 D for any starting refractive error.The pupil constriction during accommodation also contributes to increase in depth of field.The concept of micro-monovision is applied,where the near (non-dominant)eye was set up to be slightly myopic (anisometropia of 1.50 diopters) which enables to see in the near range and upto intermediate, while the predominantly distance (dominant) eye was able to see at distance down to intermediate by virtue of the depth of field.This creates a Blend zone to enable continuous distance to intermediate to near vision between the two eyes.Retinal and cortical processing increases the contrast of the retinal image.Finally the central cortical processing including neuronal gating and blur-suppression makes the image to be perceived as a clear image with well defined edges.The optimization is based on the patient age, refraction, preoperative spherical aberration, tolerance for anisometropia, and treatment centered on the corneal vertex.
We present a prospective single center study to evaluate distance and near visual outcomes and the functionalquality of vision, and patient satisfaction following Laser blended vision correction for presbyopia.
METHODS
In this single center, prospective study, we present the outcomes of Laser Blended Vision in myopic, hyperopic and emmetropic populations. A series of 50 eyes from 25 presbyopic patients were treated with LASIK between September 1, 2015 and March 10, 2016 at the NethradhamaSuperspeciality Eye hospital. All treatments were performed as bilateral simultaneous LASIK by a single surgeon.This study was approved by the Hospital ethics committee of NethradhamaSuperspeciality Eye hospital.
Patient selection
Inclusion criteria were medically suitable for LASIK, presbyopic withUncorrected near visual acuity (UNVA) of N10 or worse in both eyes, corrected distance visual acuity (CDVA) of0 LogMar or better in either eye, minimum follow-up of 3 months, and tolerance of at least –1.50 D anisometropia. Myopic patients with spherical equivalent refraction ≤–8.50 D in both eyes and hyperopic patients with spherical equivalent refraction ≤+4.00 D in both eyes and astigmatism of ≤+ 3.00D were included. Informed consent and permission to use their data for analysis and publication were obtained from each patient.The descriptive statistics of the patients including number of patients, gender, age, preoperative spherical equivalent refraction, preoperative cylinder and preoperative CDVA were obtained.
Preoperative assessment
A full ophthalmologic examination was performed on all patients prior to surgery including manifest refraction, cycloplegic refraction, slit-lamp microscopy of the anterior segment, dilated funduscopy, and intraocular pressure measurement. The pre-operative examination also included topography andkeratometry (Orbscan II-Bausch & Lomb, Oculus Pentacam and Pentacam HR), abberometry (Tracey iTrace Visual Function Analyzer) and Dry eye evaluation.Preoperative evaluation was done to rule out cataract and corneal pathologies.Uncorrected distance (UDVA)and near visual acuities (UNCA), Corrected distance (CDVA) and near visual acuities (CNVA)were assessed with the logMAR acuity charts.
Ocular dominance testing
Ocular dominance was assessed using two methods:
- The hole test involved the patient binocularly aligning a distant object through a 1inch diameter hole in a white A4 sheet of paper, held at arm’s length in landscape format with each hand holding either end. The eyes were alternately covered while looking through the hole. The eye with which the object appeared most centered through the hole was labeled to be the dom- inant eye.
- Pointing test involved the patient pointing at a spot light source 6 meters from the patient so that their finger and the light source were visually aligned. Each eye was then occluded and the eye in which the separation between their finger and the spot was the smallest was labeled to be the dominant eye.
Dominance was confirmed if the result was the same for both the tests. If both the tests were inconclusive, the micro-monovision assessment (see below) was repeated with each eye in turn as the dominant eye and the dominance was decided according to which setup felt more natural for the patient when asked after experiencing both setups.
Micro-monovision assessment
The standard micro-monovision protocol corrected the dominant eye to plano and the non-dominant eye to –1.50 D irrespective of age. Patients were tested for tolerance with the intended refraction in place using trial lens and the amount of cross-blurring reported by the patient during simulation was evaluated. Patients who reported minimal cross-blurring and was happy with the vision at distance and near were said to be tolerant of a –1.50 D add.Patients were counselled to expect an adaptation period of up to 3 months.
Surgical procedure
All patients underwent LASIK using the MEL90 excimer laser (Carl Zeiss Meditec, Jena, Germany) operating with a Advanced Ablation Algorithm (Triple-A) profile, basically a combination of the 2 profiles used with the MEL 80, aspheric optimization (ASA) and tissue-saving (TSA) profiles. It also includes a component called cone for controlled atmosphere (CCA) which maintains the atmospheric conditions by automatically and intelligently adapting airflow to either 250 or 500 Hz operation modes. The effective ablation spot size is 0.7 mm diameter (flying spot laser) at a pulse-rate frequency of 250-500 Hz(Flexiquence). The CRS-Master software platform was used to generate the ablation profiles. The surgeon selected the option in the Laser Blended Vision CRS-Master software to indicate the patient tolerated the 1.50 D test, which set the target refraction to –1.50 D in the non- dominant eye.
The ablation profiles used for all eyes (both distance and near) incorporate a pre-compensation factor for the induction of spherical aberration; the profiles were intended to reduce (not eliminate) the induction of spherical aberration so that postoperative spherical aberration is within a range that provides an increased depth of field, but without affecting contrast sensitivity and quality of vision. This range was based on studies to understand the spherical aberration levels needed to increase depth of field19,20, and the 0.56 μm spherical aberration limit above which quality of vision might be subjectively affected as previously reported by Reinstein DZ et al21.
Optical treatment zone diameters used for the study population were in the range of 6.00 mm to 7.00mm. The CRS-Master generated a file on a USB mass storage device, which was transferred to the laser and imported for treatment. The VisuMax femtosecond laser22 was used in all eyes of the study population.Both the flap and the ablation profile were centered on the corneal vertex for all patients, which closely approximates the visual axis23. The coaxially sighted corneal vertex was determined under the operating microscope of the laser by viewing through the surgeon’s contralateral eye. Centration on the corneal vertex was used so that the spherical aberration induction was symmetrical around the visual axis rather than the line of sight.
Postoperative evaluation
Follow up was conducted at day 1, 2 weeks and 3months. On all visits evaluation of binocular distance and near visual acuity using logMAR charts were conducted. The functional quality of vision was evaluated by assessing:
- The reading acuity (logMAR) and reading speeds (words per minute) at 70, 80 and 90 cm using Salzburg reading desk,
- Mesopic contrast sensitivity (F.A.C.T. STEREO OPTICAL CO., INC. Chicago)with distance correction was measured in all the treated eyes at 1.5,3, 6, 12, and 18 cycles per degree (cpd),
- Higher order abberations (Tracey iTrace Visual Function Analyzer)
Finally the subjective assessment of dysphotopsia, spectacle independence for all activities and overall patient satisfaction was carried out using a questionnaire.
STATISTICAL ANALYSIS
The outcomes were analyzed for final results after all treatments. Microsoft Excel 2003 (Microsoft Corporation, Seattle, WA, USA) was used for data entry. The Statistical analysis was done using SPSS software (version 15, Illinois, Chicago). Descriptive analysis was performed and independent variables were analysed using appropriate tests at 5% level of significance.
RESULTS
A total of 50 eyes of 25 patients were operated. Average age was 47.72 ± 4.06 years. There were 14 women (56%) and 11 men (44%). Out of 25 patients 13(52%) were myopes, 10(40%) were hyperopes and 2(8%) were emmetropes.The mean preoperative spherical equivalent (SE) in the hyperopic population was +1.26± 0.66 D, while that of the myopic population was −3.5 ± 1.59D(Table 1)
Table 1. Demographic characteristics and preoperative data
| Parameters | Mean ± SD | |
| Mean age | 47.72 ± 4.06 years | |
| Males : females | 11: 14 | |
| Mean near add | +1.58D ± 0.43 | |
| Hyperopes | Myopes | |
| Mean Spherical equivalent | +1.26D± 0.66 | -3.5D± 1.59 |
The mean binocular uncorrected distance acuity (UCDA) was 0.15 logMAR at pre-op evaluation, 0.008 logMAR(p=0.00) at post op day1, -0.016 logMAR at 2 weeks post op and -0.02 logMAR at 3 months post op. The mean binocular uncorrected near acuity (UCNA) was 0.25 logMAR at pre-op evaluation, -0.04 logMAR(p=0.01) at post op day1, -0.22logMAR at 2 weeks post op and -0.24logMAR at 3 months post op.
23 (92%) patients had cumulative binocular UCDA of 0 or better logMAR(≥6/6) and 25 (100%) pateints had cumulative binocular UCNA of 0 or better logMAR (≥6/6) at 3 months post operatively.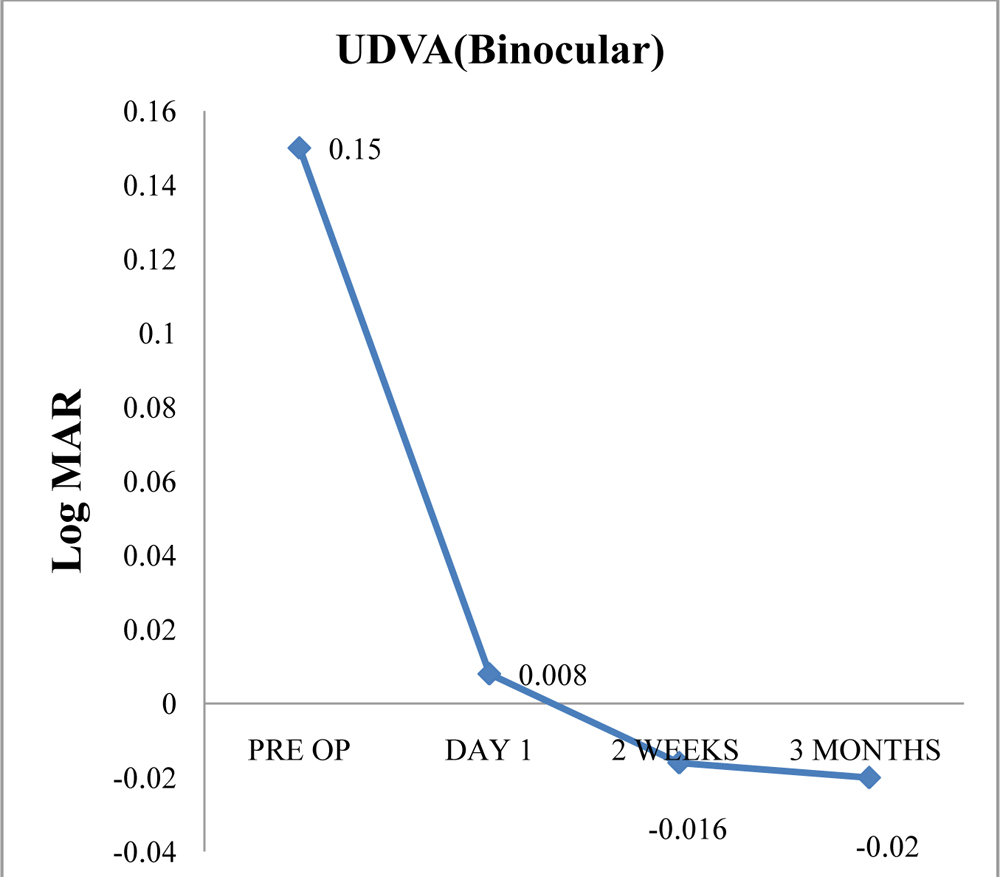
Figure 1:Binocular Uncorrected Distance visual acuity over time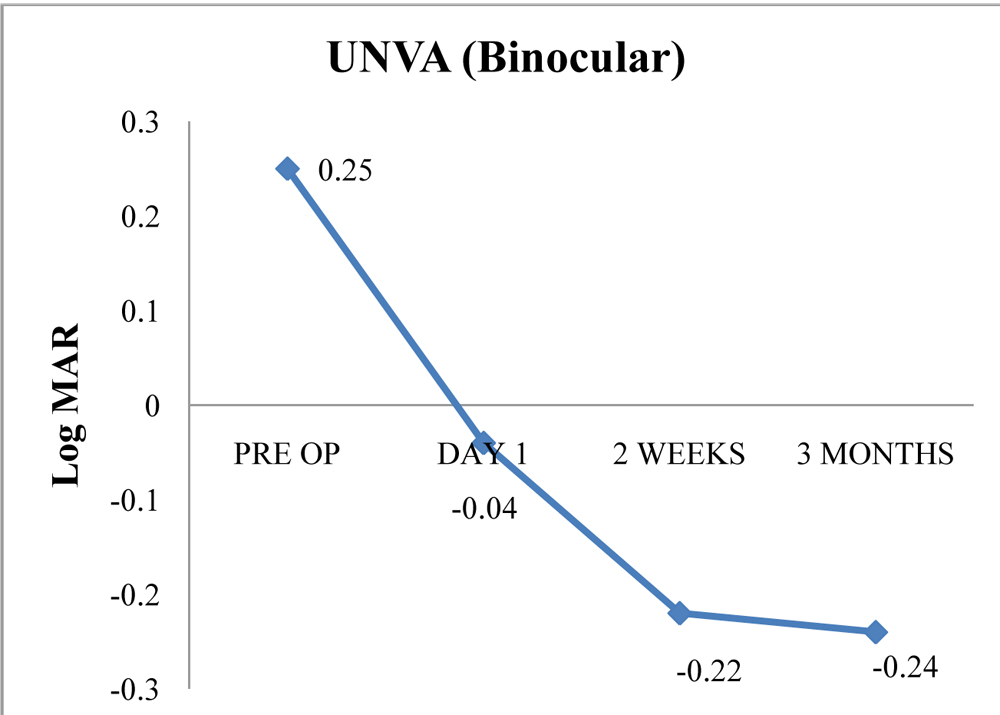 Figure 2:Binocular Uncorrected Near visual acuity over time.
Figure 2:Binocular Uncorrected Near visual acuity over time.
All 50(100%) eyes had a cumulative reading acuity of better than or equal to 0.4 LogMar (≥6/15) (Figure 3) and had reading speed of better than or equal to 80 words per minute (wpm)(Figure 4) at all distances (70cm, 80cm and 90cm) when subjected to Salzburg reading desk test.
All patients had good stereopsis and no significant change in contrast sensitivity at post operative period of 3 months as shown in the Figure 5.
At 3 months the Higher order aberrations (HO total µ) in myopic eyes reduced from 0.1433 and 0.1330 to 0.1008 and 0.0892 in the dominant and non-dominant eye respectively(Figure 6). 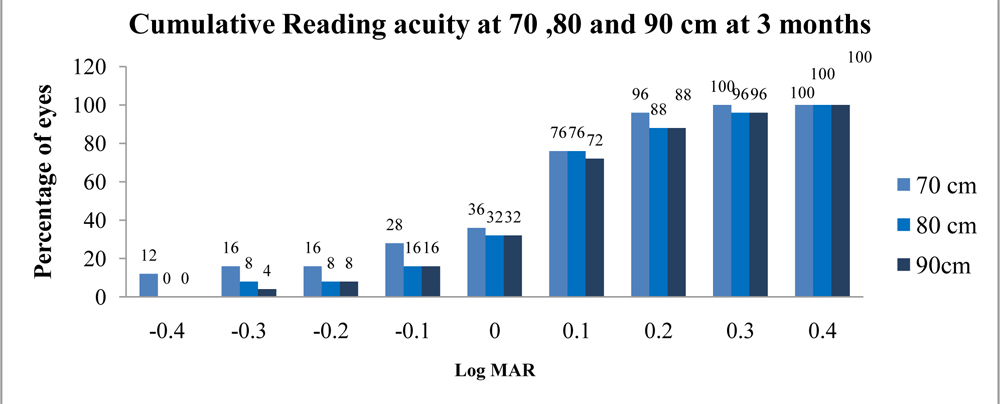
Figure 3: Cumulative Reading acuity at 70 ,80 and 90 cm at 3 months
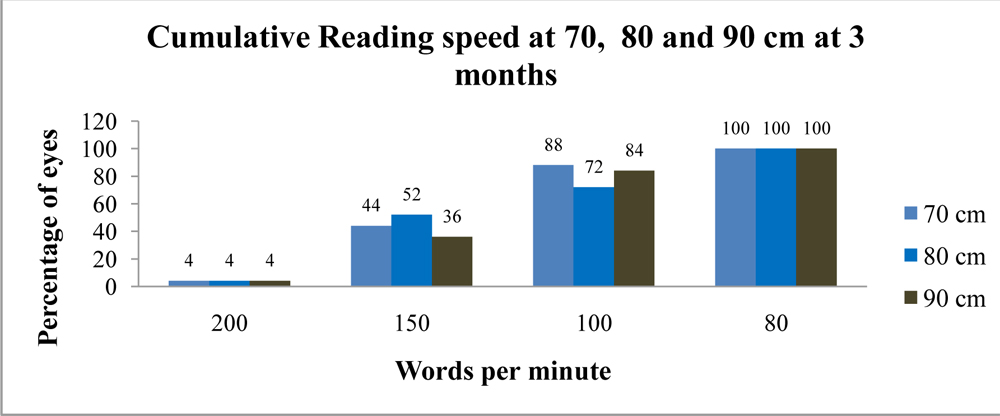
Figure 4: Cumulative Reading speed at 70 ,80 and 90 cm at 3 month
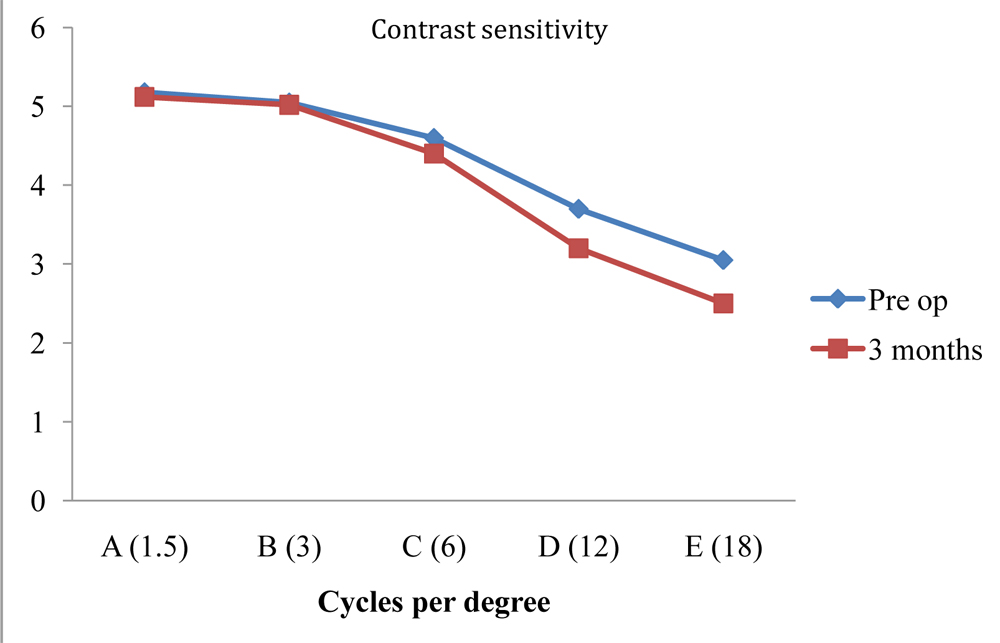 Figure 5: Showing the comparison of contrast sensitivity in the pre-operative and 3rdpost operative month.
Figure 5: Showing the comparison of contrast sensitivity in the pre-operative and 3rdpost operative month.
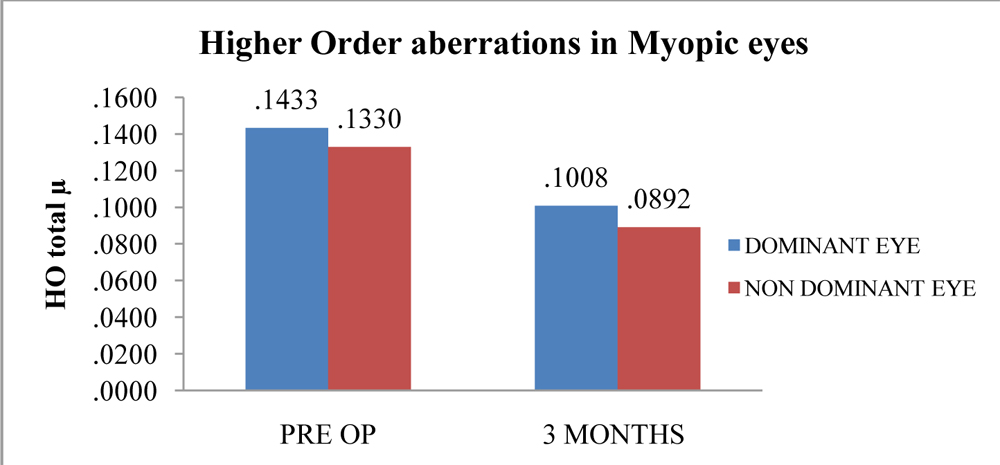 Figure 6: Showing the comparison of higher order aberrations in myopic eyes in the pre-operative and 3rdpost operative month.
Figure 6: Showing the comparison of higher order aberrations in myopic eyes in the pre-operative and 3rdpost operative month.
None of the operated eyes required a second intervention to modify the refractive
results obtained. There were no complications during or after any of the surgeries performed. None of the study patients reported presence of glare or halos in the subsequent post operative periods. All of the patients (100%) achieved complete independence from spectacles for all activities while only 2 patients (8%) had subjective complaints of dryness.
DISCUSSION
Laser Blended Vision was a well tolerated, safe and effective procedure for the treatment of presbyopia in myopia up to –8.50 D, hyperopia up to +5.75 D and emmetropia24,25.
All eyes showed significant improvement in the post operativebinocular near and distant vision at day1 (p values <0.05) which was sustained at subsequent visits with no significant change thereafter. The binocular UDVA was found to be better than the UDVA of the distance eyes alone. This shows that the near eye still appears to contribute positively to binocular distance vision despite a blurred image. This finding is in contrast to the traditional contact lens monovision where binocular distance vision is often slightly reduced, particularly for near addition of +2.00 D or greater26-29.
The near efficacy outcomes were also better than might be expected given the low degree of anisometropia. The near vision reported in the myopic and hyperopic populations was comparable with the near vision reported in previous LASIK monovision studies (94-100% of patients could read N54,6,30-32). However, these other LASIK monovision studies had used an age related nomogram for anisometropia. Therefore, an equivalent level of near vision was achieved in this study despite the lower degree of anisometropia employed in the Laser Blended Vision protocol.
In summary, the factors described above appear to demonstrate evidence of an increased depth of field induced by the Laser Blended Vision procedure in that the non-dominant eye behaves as though it has more ‘add’ at near and less ‘add’ at distance than might be expected given the nominal spherical equivalent refraction. This improvement in depth of field might be explained by the induction of spherical aberration.
The safety of the procedure was found to be very high with no eye in the study populations losing two lines of CDVA. The safety, in terms of the contrast sensitivity was also found to be high, with no reduction in average contrast sensitivity at each frequency level. Satisfactory intermediate reading acuity and reading speeds were recorded in our study objectively with the Salsburg Reading Desk.
The patient acceptance and tolerance of the Laser Blended Vision protocol was high, demonstrating an improvement over other presbyopic treatment modalities, such as the reported tolerance for contact lens monovision of 59-67%29.
In conclusion, the results obtained in our study demonstrate that Laser blended vision appears to be a promising modality for treatment of presbyopia with favorable distance, intermediate and near visual outcomes and good functional quality of vision, thus offering patients greater independence from glasses in their daily lives.
REFERENCES
1.Dictionary.comUnabridged,“presbyopia,”RandomHouse,Inc, http://dictionary.reference.com/browse/presbyopia.
2.Census data and global changes in demographics. Available at: http://www.census.gov/cgi-bin/broker. Accessed March 15, 2012.
3.Patel I, West S. Presbyopia: prevalence, impact and interventions. Community Eye Health J. 2007; 20:40-1.
4.Goldberg DB. Laser in situ keratomileusismonovision. J Cataract Refract Surg. 2001; 27(9): 1449-1455.
5.Miranda D, Krueger RR. Monovision laser in situ ker- atomileusis for pre-presbyopic and presbyopic patients. J Refract Surg. 2004; 20(4): 325-328.
6.Wright KW, Guemes A, Kapadia MS, Wilson SE. Binocular function and patient satisfaction after monovision induced by myopic photorefractive keratectomy. J Cataract Refract Surg. 1999; 25(2): 177-182.
7.Fawcett SL, Herman WK, Alfieri CD, Castleberry KA, Parks MM, Birch EE. Stereoacuity and foveal fusion in adults with long-standing surgical monovision. J Aapos. 2001; 5(6): 342-347.
8.El Danasoury AM, Gamaly TO, Hantera M. Multizone LASIK with peripheral near zone for correction of presbyopia in myopic and hyperopic eyes: 1-year results. J Refract Surg. 2009; 25(3): 296-305.
9.Uy E, Go R. Pseudoaccommodative cornea treatment using the NIDEK EC-5000 CXIII excimer laser in myopic and hyperopic presbyopes. J Refract Surg. 2009; 25(1 Suppl): S148-155.
10.Jung SW, Kim MJ, Park SH, Joo CK. Multifocal corneal abla- tion for hyperopic presbyopes. J Refract Surg. 2008; 24(9): 903-910.
11.Alio JL, Chaubard JJ, Caliz A, Sala E, Patel S. Correction of presbyopia by technovision central multifocal LASIK (presbyLASIK). J Refract Surg. 2006; 22(5): 453-460.
12.Pinelli R, Ortiz D, Simonetto A, Bacchi C, Sala E, Alio JL. Correction of presbyopia in hyperopia with a center-distance, paracentral-near technique using the Technolas 217z platform. J Refract Surg. 2008; 24(5): 494-500.
13.Cosar CB, Sener AB. Supracor hyperopia and presbyopia correction: 6-month results. Eur J Ophthalmol.2014; 24:325-9.
14.Tomita M, Kanamori T, Waring GO, 4th, Nakamura T, Yukawa S. Small-aperture corneal inlay implantation to treat presbyopia after laser in situ keratomileusis. J Cataract Refract Surg. 2013; 39:898-905.
15.Limnopoulou AN, Bouzoukis DI, Kymionis GD et al. Visual outcomes and safety of a refractive corneal inlay for presbyopia using femtosecond laser. J Refract Surg. 2013; 29:12-8.
16.Cantu R, Rosales MA, Tepichin E, Curioca A, Montes V, Ramirez-Zavaleta JG. Objective quality of vision in presbyopic and non-presbyopic patients after pseudoaccommodative advanced surface ablation. J Refract Surg. 2005; 21(5 Suppl): S603-605.
17.Oliver KM, O’Brart DP, Stephenson CG, Hemenger RP, Applegate RA, Tomlinson A, Marshall J. Anterior corneal opti- cal aberrations induced by photorefractive keratectomy for hyperopia. J Refract Surg. 2001; 17(4): 406-413.
18.Artola A, Patel S, Schimchak P, Ayala MJ, Ruiz-Moreno JM, Alio JL. Evidence for delayed presbyopia after photorefractive keratectomy for myopia. Ophthalmology. 2006; 113(5): 735- 741 e731.
19.Marcos S, Moreno E, Navarro R. The depth-of-field of the human eye from objective and subjective measurements. Vision Res. 1999; 39(12): 2039-2049.
20.Marcos S, Barbero S, Jimenez-Alfaro I. Optical quality and depth-of-field of eyes implanted with spherical and aspheric intraocular lenses. J Refract Surg. 2005; 21(3): 223-235.
21.Reinstein DZ, Archer TJ, Couch D, Schroeder E, Wottke M. A new night vision disturbances parameter and contrast sensi- tivity as indicators of success in wavefront-guided enhance- ment. J Refract Surg. 2005; 21(5): S535-540.
22.Reinstein DZ, Archer TJ, Gobbe M, Johnson N. Accuracy and Reproducibility of Artemis Central Flap Thickness and Visual Outcomes of LASIK With the Carl Zeiss MeditecVisuMax Femtosecond Laser and MEL 80 Excimer Laser Platforms. J Refract Surg. 2010; 26(2): 107-119.
23.Pande M, Hillman JS. Optical zone centration in keratorefrac- tive surgery. Entrance pupil center, visual axis, coaxially sight- ed corneal reflex, or geometric corneal center? Ophthalmology. 1993; 100(8): 1230-1237.
24.Reinstein DZ, Couch DG, Archer TJ. LASIK for Hyperopic Astigmatism and Presbyopia Using Micro-monovision With the Carl Zeiss Meditec MEL80.J Refract Surg. 2009; 25(1): 37-58.
25.Reinstein DZ, Archer TJ, Gobbe M. LASIK for Myopic Astigmatism and Presbyopia Using Non-Linear Aspheric Micro-Monovision with the Carl Zeiss Meditec MEL 80 Platform. J Refract Surg. 2011; 27(1): 23-37.
26.Erickson P. Potential range of clear vision in monovision. J Am Optom Assoc. 1988; 59(3): 203-205.
27.Erickson P, McGill EC. Role of visual acuity, stereoacuity, and ocular dominance in monovision patient success.Optom Vis Sci. 1992; 69(10): 761-764.
28.Durrie DS. The effect of different monovision contact lens powers on the visual function of emmetropicpresbyopic patients (an American Ophthalmological Society thesis). Trans Am Ophthalmol Soc. 2006; 104): 366-401.
29.Evans BJ. Monovision: a review. Ophthalmic Physiol Opt. 2007; 27(5): 417-439.
30.Goldberg DB. Comparison of myopes and hyperopes after laser in situ keratomileusismonovision. J Cataract Refract Surg. 2003; 29(9): 1695-1701.
31.Reilly CD, Lee WB, Alvarenga L, Caspar J, Garcia-Ferrer F, Mannis MJ. Surgical monovision and monovision reversal in LASIK. Cornea. 2006; 25(2): 136-138.
32.Braun EH, Lee J, Steinert RF. Monovision in LASIK. Ophthalmology. 2008; 115(7): 1196-1202.

