Dr. Tushar Grover, G15580, Dr. Rohit Shetty, Dr. Rushad Shroff, Dr. Abhijit Sinha Roy
Introduction
Laser In Situ Keratomileusius (LASIK) is the most commonly performed procedure for correction of refractive error. Its safety and efficacy has been established with numerous studies. Femtosecond laser has improved flap creation and allowed customisation which was not previously available with conventional LASIK.
Small Incision Lenticule Extraction (SMILE) is a newly developed novel procedure that works by the creation of an intrastromal lenticule that is removed by a small corneal incision. This has eliminated the need for a corneal flap and is an all in one procedure
Theoretically, SMILE being a minimally invasive procedure is likely to be more biomechanically stable,lead to better visual outcomes and cause reduced dry eye. There is however no clear understanding of the mechanistic pathways responsible for these differences. This study aims to compare the two procedures in contralateral eyes with Corneal Biomechanics, Corneal Tomography, Imaging from a high definition Spectral Domain OCT and proteomic analysis from tears.
Materials and Methods
Eighty eyes of forty patientsmatched for corneal biomechanics and refractive error were randomly assigned to undergo SMILE in one eye and FS LASIK in the other eye in this prospective, interventional, study. The study was conducted in accordance with the tenets of the Declaration of Helsinki and was approved by the institutional research and ethics committee of Narayana Nethralaya Eye Hospital, India. A written informed consent was obtained from all patients before the procedure.
Study Population
Patients between 18 to 50 years of age with stable myopia for a minimum period of one year (a change of 0.25D or less), a corrected distance visual acuity (CDVA) of 20/32 or better, a spherical equivalent refraction less than −10D and a refractive astigmatism less than -3D were included in the study. Exclusion criteria included a central corneal thickness (CCT) less than 480 micrometer, a calculated residual stromal bed thickness of less than 250 micrometer after the surgery, patients with a history of keratoconus, diabetes, collagen vascular disease, pregnancy, breastfeeding and any prior ocular surgery.
Study design:
Eligible patients underwent a detailed ocular examination, which included visual acuity and manifest refraction assessment, and fundoscopy. Aberration measurements and corneal topography were performed using the Pentacam (Oculus Optikgerate GmBH, Wetzlar, Germany). Corneal biomechanics was measured with Corvis ST(Oculus Optikgerate GmBH, Wetzlar, Germany ), imaging with a high resolution anterior segment Spectral Domain Optical Coherence Tomography (SD – OCT) by Bioptigen andquality of vision with Optical Quality Analysis System(Visiomerics, Terrasa, Spain). All parameters were assessed preoperatively and at 1 month, 3 month and 6 months postoperatively. Tears from schirmer strips were also collected from both eyes for proteomic analysis at the same intervals
Study treatments and procedures:
All surgeries were performed by a single experienced surgeon under aseptic precautions and topical anaesthesia after instilling 0.5% proparacaine hydrochloride (Paracain, Sunways Pvt. Ltd., India). One eye of each patient was randomly assigned to the SMILEgroup and the fellow eye to the LASIK group.
Assessment of corneal Biomechanics
The corneal biomechanical outcome of the surgery was measured in three different ways using analytical model, inverse finite element method (FEM) model and bowman’s imaging
Analytical model
The analytical model of corneal and extra-ocular tissue was realized using spring dashpot based mechanical equivalent of cornea. The model was constructed based on the known rapid response of cornea and, rapid deformation and slow reformation of the extra-ocular tissue. From the 32ms Corvis-ST (OCULUS Optikgerate Gmbh, Germany) deformation video, extra-ocular tissue deformation can be quantified based on the peripheral movement of the cornea (represented as u2,
fig 1), which has reluctance in reforming after the air-puff pressure goes to zero.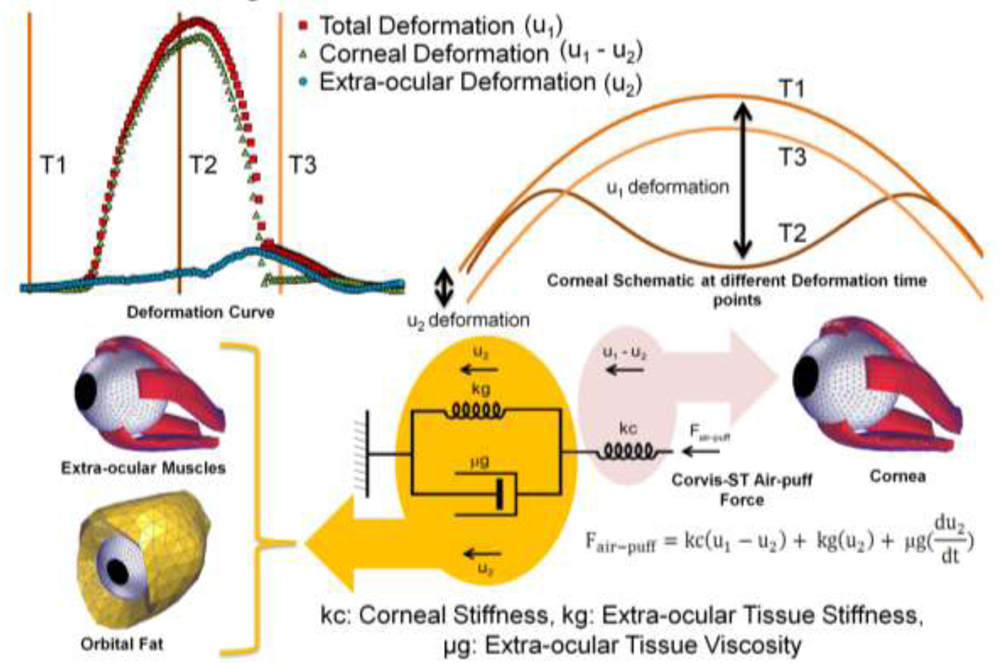 Fig 1: The analytical biomechanics model of corneal and extra-ocular tissue. T1 (before air-puff), T2(peak air-puff pressure) and T3 (zero air-puff pressure) time points of anterior corneal schematic and its corresponding deformation curve. The dashpot based mechanical equivalent of cornea and its mathematical representation. The material property indices (kc, kg and µg) representing the corneal and extra-ocular tissue properties.
Fig 1: The analytical biomechanics model of corneal and extra-ocular tissue. T1 (before air-puff), T2(peak air-puff pressure) and T3 (zero air-puff pressure) time points of anterior corneal schematic and its corresponding deformation curve. The dashpot based mechanical equivalent of cornea and its mathematical representation. The material property indices (kc, kg and µg) representing the corneal and extra-ocular tissue properties.
Inverse FEM Model
The inverse FEM uses structural information from Pentacam HR (OCULUS Optikgerate Gmbh, Germany) and functional information from Corvis-ST. The structural information was converted to mesh grid and the inverse FEM model was solved using the Abaqus/CAE 6.12 (Dassault Systèmes, France).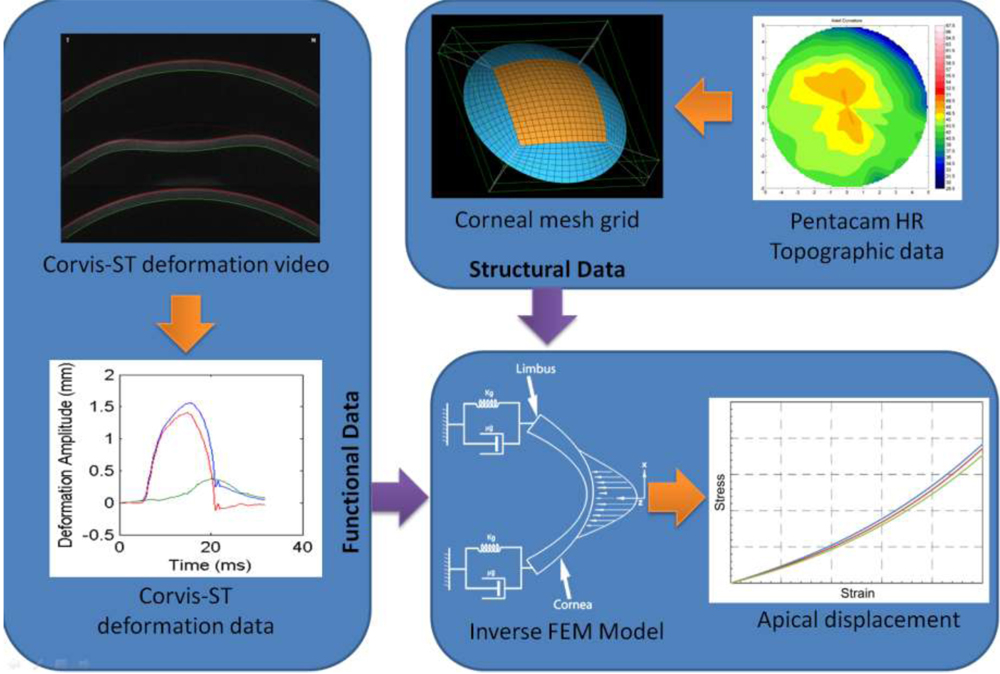 Fig 3: Inverse FEM calculation flow with the model used for the calculation
Fig 3: Inverse FEM calculation flow with the model used for the calculation
Bowman’s imaging
Corneal high resolution OCT images show micro distortions in the bowman’s layer which change post refractive surgery. The bowman’s roughness index (BRI) was defined as a quantitative term to describe these distortions. Once the images were acquired from the OCT they were further processed using graph theory based algorithm (MathWorks Inc., Natick, MA). After the bowman’s layer was detected a third order polynomial was made to fit on the edge data to get the hypothetical edge which doesn’t have micro distortions. The difference in the area between the actual bowman’s edge and the smooth fit will give the area of the micro distortions. The arithmetic sum of the obtained area provides the BRI. These distortions which where quantified before and after surgery, to get the change induced due to the procedure on the bowman’s layer.
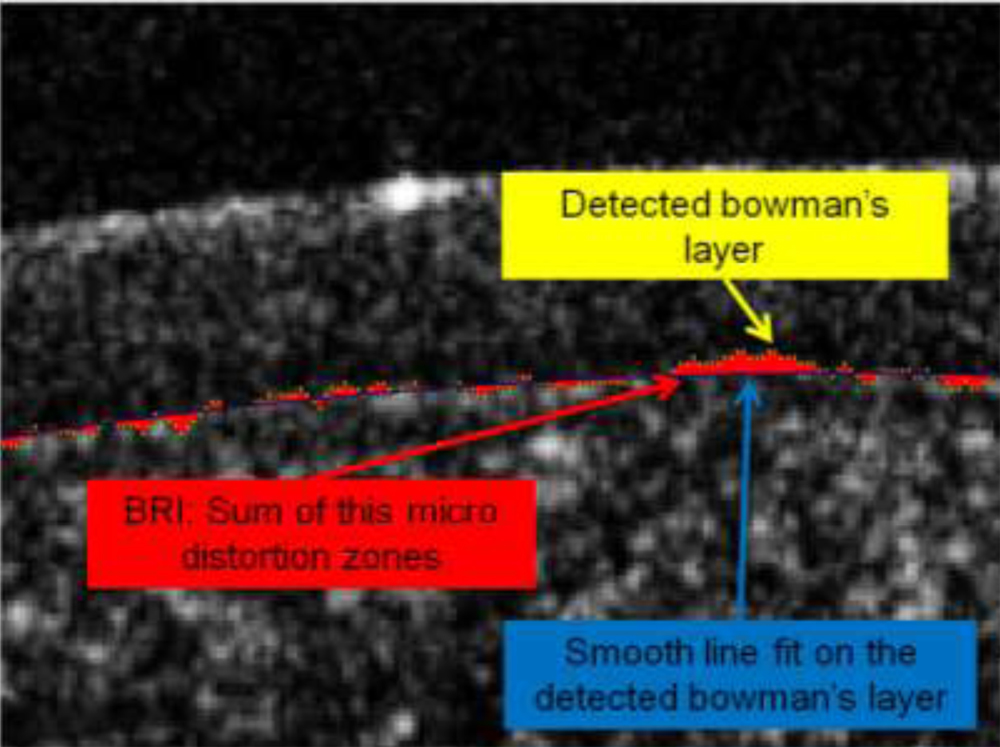 Fig 3: Overlay of the bowman’s edge detected (yellow) and the smooth line fit (blue), bowman’s roughness represented in red area.
Fig 3: Overlay of the bowman’s edge detected (yellow) and the smooth line fit (blue), bowman’s roughness represented in red area.
Tear Proteomics
Tear fluid was collected using Schirmer’s strip from contralateral eyes of patients prior to LASIK/SMILE surgery (Pre-Op) and from longitudinal follow-ups of 1 month and 3 months post surgery. The tear proteins from the Schirmer’s strips were extracted using a suitable buffer and the protein concentration from each sample was estimated by Standard Bradford assay.
Results
Visual
Efficacy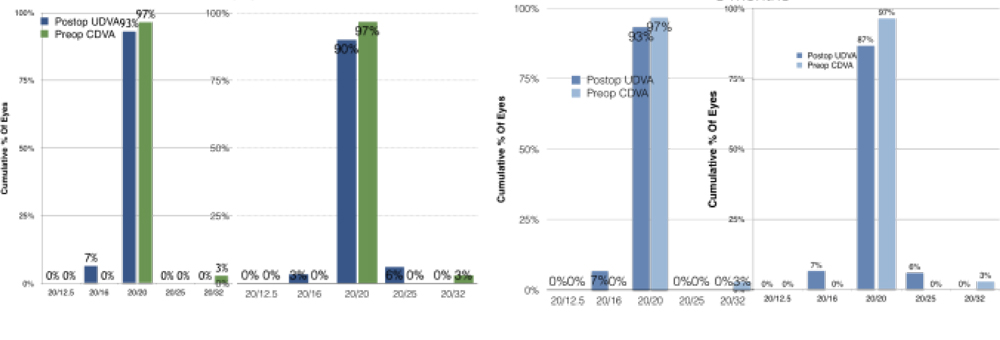 Postoperative UDVA was comparable between the two groups. Both FS and SMILE had UDVA of 20/20 in 97% of subjects at 3 months postoperatively.
Postoperative UDVA was comparable between the two groups. Both FS and SMILE had UDVA of 20/20 in 97% of subjects at 3 months postoperatively.
Accuracy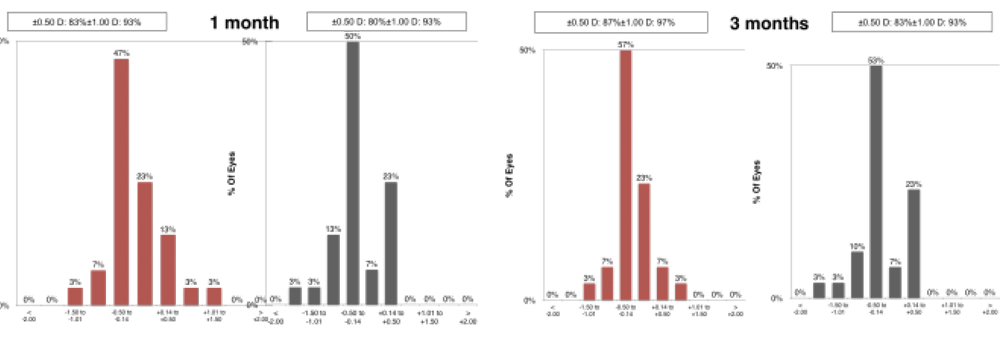 At 1 month, the postoperative Manifest Refraction Spherical Equivalent(MRSE) was ±0.50 D in 83% of eyes and ±1.00 D in 93% in the LASIK group. The MRSE was±0.50 D in 80% and ±1.00 D in 93% subjects in the SMILE group at 1 month. The same at 3 months was ±0.50 D in 87% and 83 % and ±1.00 D in 97% and 93% in the LASIK Group and SMILE group respectively. These results were not statistically significant
At 1 month, the postoperative Manifest Refraction Spherical Equivalent(MRSE) was ±0.50 D in 83% of eyes and ±1.00 D in 93% in the LASIK group. The MRSE was±0.50 D in 80% and ±1.00 D in 93% subjects in the SMILE group at 1 month. The same at 3 months was ±0.50 D in 87% and 83 % and ±1.00 D in 97% and 93% in the LASIK Group and SMILE group respectively. These results were not statistically significant
Biomechanics
The analytical model based data indicates that SMILE has a better biomechanical outcome at third month compared to FS-LASIK, fig 4 left. Similar trend was also observed in the case of inverse FEM analysis (randomly selected six from the above 12 patients cohort) indicating the FS-LASIK instability at third month, fig 5. BRI value was decreased and stabilized in the first month itself in case of SMILE, indicating possible tissue healing, which was better than FS-LASIK, fig 4 right.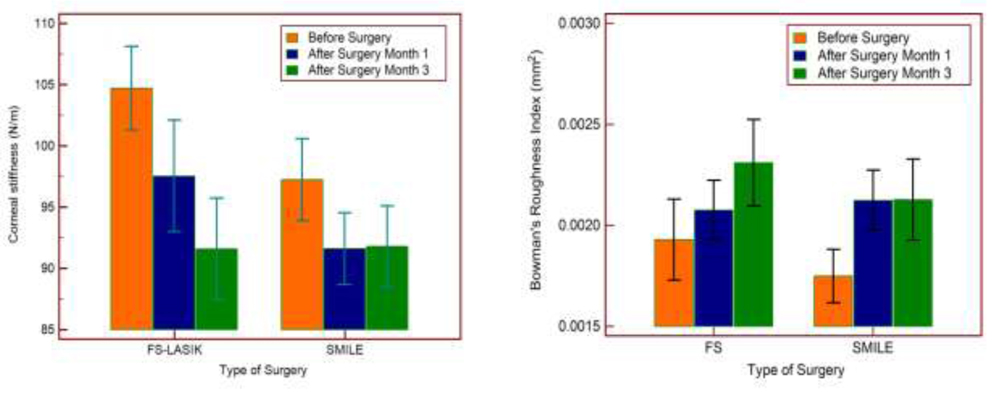 Fig 4: Left figure show the analytical model results and right figure show the bowman’s roughness index result
Fig 4: Left figure show the analytical model results and right figure show the bowman’s roughness index result 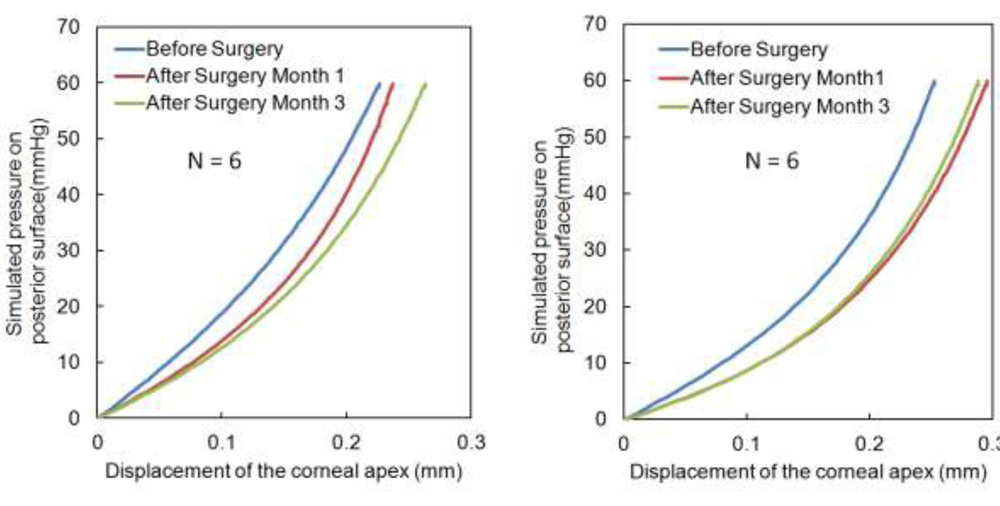 Fig 5: The inverse FEM parameters based IOP simulation (0-70 mmHg) results of the cornea (6 patients)
Fig 5: The inverse FEM parameters based IOP simulation (0-70 mmHg) results of the cornea (6 patients)
The analysis of BRI data comparing SMILE and FS-LASIK shows high value of the BRI in SMILE, fig 6.
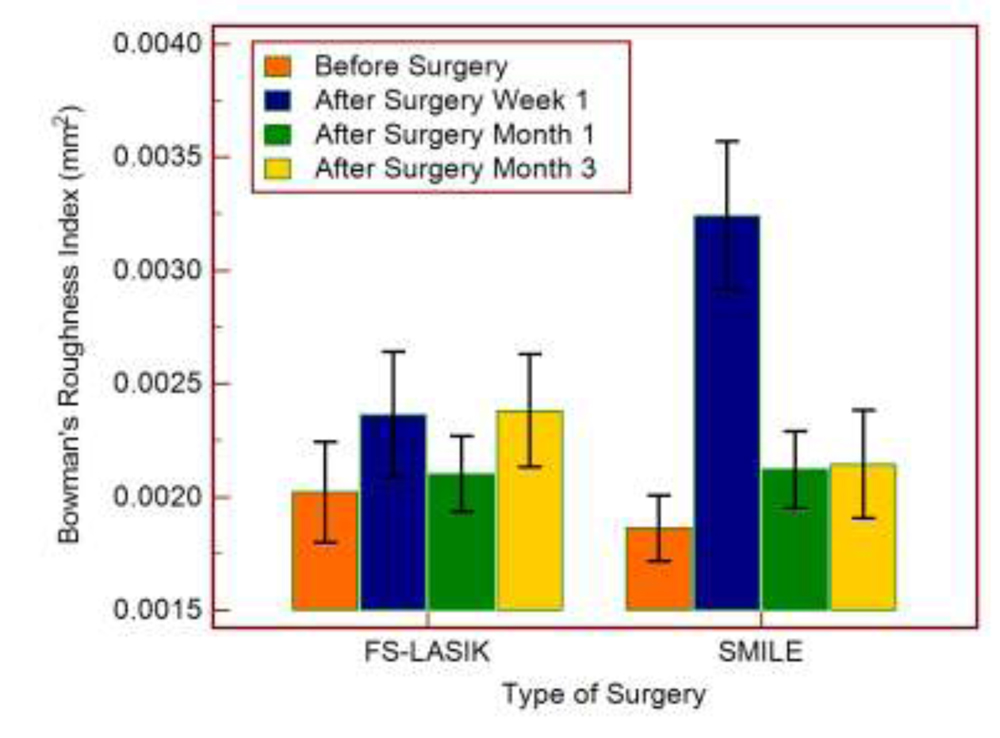 Fig 6: BRI result for time points including one week SMILE vs LASIK
Fig 6: BRI result for time points including one week SMILE vs LASIK
All these methods show SMILE to stabilize biomechanically at first month and FS-LASIK to be unstable even at third month.
Tear Proteomics
Functional analysis performed showed proteins related to wound healing/inflammatory response, oxidative stress and cell homeostasis to have altered among the LASIK and SMILE surgeries. Proteins involved in inflammation such as lysozyme C (LysC), interleukin 1 receptor antagonist (IL-1RA) alpha 1 antichymotrypsin (SERPINA3) were observed at higher levels in LASIK 3 month post-op whereas their levelsreduced in SMILE eyes 3 months post-op compared to LASIK. Fetuin A (FETUA) involved in wound healing response was identified at higher levels in SMILE compared to LASIK indicating aggressive wound healing. (Fig 7).
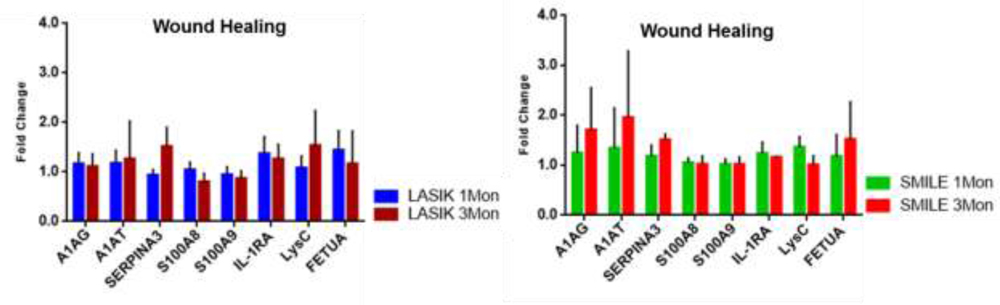 Fig 7: Modulated inflammatory response in SMILE compared to LASIK 3 Mon post-
Fig 7: Modulated inflammatory response in SMILE compared to LASIK 3 Mon post-
Discussion
Refrative surgery has undergone a paradigm shift in the last decade. Though Femtosecond LASIK has proved to provide excellent outcomes, the introduction of SMILE has shifted the spotlight and there has been increasing interest in comparison of the two procedures.
In this study, we found that visual outcomes were comparable between the two groups. Both SMILE and LASIK showed high efficacy and accuracy. These results were similar to other studies thatshow comparable efficacy between the two groups.1,2
Biomechanics analysed from Analytical and Finite Element Model showed better biomechanical outcome at 3 months in the LASIK group with greater biomechanical instability in LASIK.LASIK has been found to have reduced Corneal Hysteresis(CH) and Corneal Resistance Factor(CRF).3This study focuses on contralateral eyes, hence the outcome is more representative of the actual surgery and is less likely to be affected by intra patient variability.Bowman Roughness Index(BRI) was higher in the SMILE group at 1 week post op. This could be a possible indicator of corneal tissue disturbance during the surgical procedureThere was however rapid restoration to normal levels indicating rapid recovery of corneal biomechanics in SMILE.
SMILE has been found to induce less keratocyte apoptosis, proliferation and inflammation compared with femtosecond LASIK in animal studies4. In this study, proteomic analysis from tears collected revealed reduced inflammatory mediators in SMILE, while proteins associated with wound healing were found to be increased in the SMILE group, indicating reduced inflammation and rapid healing response in SMILE
Conclusion
Our study found that despite the presence of comparable visual outcomes, there is increasing evidence towards SMILE being a more stable procedure in the long term. This is indicated by faster recovery of Bowman’s layer, stronger biomecanics, less inflammation and better wound healing in SMILE.
References
1) Liu M, Chen Y, Wang D, Zhou Y, Zhang X, He J, Zhang T, Sun Y, Liu Q. ClinicalOutcomes After SMILE and Femtosecond Laser-Assisted LASIK for Myopia and MyopicAstigmatism: A Prospective Randomized Comparative Study. Cornea. 2016Feb;35(2):210-6. doi: 10.1097/ICO.0000000000000707.
2) Lin F, Xu Y, Yang Y. Comparison of the visual results after SMILE andfemtosecond laser-assisted LASIK for myopia. J Refract Surg. 2014Apr;30(4):248-54. doi: 10.3928/1081597X-20140320-03. Erratum in: J Refract Surg.2014 Sep;30(9):582.
3) Wang D, Liu M, Chen Y, Zhang X, Xu Y, Wang J, To CH, Liu Q. Differences in thecorneal biomechanical changes after SMILE and LASIK. J Refract Surg. 2014Oct;30(10):702-7. doi: 10.3928/1081597X-20140903-09.
4) Dong Z, Zhou X, Wu J, Zhang Z, Li T, Zhou Z, Zhang S, Li G. Small incision lenticule extraction (SMILE) and femtosecond laser LASIK: comparison of corneal wound healing and inflammation. Br J Ophthalmol. 2014 Feb;98(2):263-9. doi:10.1136/bjophthalmol-2013-303415

