![]()
AIOS – S Natarajan Award
Dr. Vanee Sheth,S15462, Dr. George J Manayath, Dr. Saravanan V R, Dr. Narendran V
Authors:
Vanee Sheth Shah MS, FICO
George J Manayath FRCS
Veerappan R Saravanan FRCS
Venkatapathy Narendran DNB
Parag K Shah, Vishma Prabhu, Smita S Karandikar, Ratnesh Ranjan, Venkatapathy Narendran, Narendran Kalpana, Pediatric Retina and Ocular Oncology Department, Aravind Eye Hospital and Postgraduate Institute of Ophthalmology, Coimbatore 641014, Tamilnadu, India
Department of Vitreoretina services, Aravind Eye Hospital, Coimbatore, Tamil Nadu, India
ABSTRACT:
Purpose: To study cases with features of polypoidal choroidal vasculopathy (PCV) and central serous retinopathy (CSR) simultaneously, on multimodal imaging.
Method: A review of 181cases of suspected PCV which underwent fundus fluorescein angiography(FFA) and indocyanine green angiography (ICGA) from Aug’15 to May’16 was done, to look for cases with features of CSR in the same eye. History, Best corrected visual acuity (BCVA), optical coherence tomography (OCT), FFA and ICG findings were evaluated.
Results: 5cases (2.8%); 3 males & 2 females with mean age 63.2yrs were included. OCT showed notched pigment epithelial detachment (PED) in 3 and irregular PED in 2 cases with subretinal fluid/subretinal haemorrhage over a thick choroid with dilated outer choroidal PACHYVESSELS. FFA showed stippled hyperfluorescence at polyp area and a separate CSR leak -Ink blot in 2 and Smoke stack in 3 cases. On ICGA, multiple polyps were noted suggestive of PCV.
Conclusion: The coexistence of PCV, CSR and thick choroid in same eye at the same time strongly suggest that these diseases have association and perhaps originate from predisposed thick choroid (PACHYCHOROID SPECTRUM).
Key Words
Central serous chorioretinopathy,pachychoroid, polypoidal choroidal vasculopathy
INTRODUCTION
Polypoidal choroidal vasculopathy (PCV) was described by Yannuzzi et al1 as a condition consisting of peculiar subretinal polypoidal vascular lesions associated with serous and hemorrhagic pigment epithelial detachments. Central serous chorioretinopathy (CSR)2 is characterized by idiopathic detachment of neurosensory retina. Usually a self limiting condition, but chronic cases are associated with atrophic changes in the RPE. Choroidal neovascularisation(CNVM)3 may also develop in some cases.
There are reports in literature linking PCV and CSR. The history of CSR is seen more commonly in eyes with PCV4. In a case series by Park et al5, changes of chronic CSR have been demonstrated in eyes with PCV. PCV can also masquerade as persistent or recurrent CSR6. Although the pathogenesis of these diseases is not clear, choroidal circulation abnormalities leading to congestion and hyperpermeability with thickened choroids are associated with both these clinical entities7,8,9.
However, till date there is no direct evidence yet connecting these clinical entities and with the advancement in imaging techniques, the visualization of choroid has become better. Hence, we studied the multimodal imaging features in cases of suspected PCV, looking for simultaneous presence of features of CSR.
METHODS
The clinical and imaging data of 181 cases of suspected PCV which underwent simultaneous fundus fluorescein angiography (FFA) and indocyanine green angiography (ICGA) from a period of August 2015 to May 2016 at Department of Vitreoretina services, Aravind Eye Hospital were reviewed to look for features of CSR. The relevant history, best corrected visual acuity(BCVA) and clinical features were noted. The spectral domain optical coherence tomography (OCT) and enhanced depth imaging (EDI)-OCT images (Spectralis Heidelberg Engineering and/or DRI OCT Triton plus, Topcon) as well as FFA and ICGA (Spectralis; Heidelberg Engineering) were reviewed.
PCV was suspected on the basis of presence of tall, peaked pigment epithelial detachment (PED) with a notch or irregular pigment epithelial detachment (PED) with subretinal fluid (SRF) or subretinal haemorrhage (SRH) on OCT and the diagnosis of PCV was confirmed on ICGA if focal hyperfluorescence suggestive of polyp was seen within the first 6 minutes on ICGA. Active CSR was identified by presence of characteristic pattern of leak on FFA- smoke-stack or ink blot, along with presence of SRF with or without serous PED on OCT. The subfoveal choroidal thickness was measured manually using calipers.
RESULTS
Of the 181 cases reviewed, 5 eyes of 5 patients (2.8%), 3 males and 2 females, were identified with features of active PCV as well as active CSR simultaneously in the same eye. The mean age was 63.2years (52 to 70 years). 3 patients had history of systemic hypertension. The BCVA in the affected eye was 6/18 in three cases, 6/12 in one case and 6/9 in one case. On OCT, notched PED was seen in 2 cases and irregular peaked PEDs were seen in 4 cases. SRF was noted in all cases at the site of polyp or CSR. The mean subfoveal choroidal thickness was 371.2µm. On FFA, the ink blot pattern of CSR leak was noted in 2 cases and 3 cases showed smoke-stack leak. A separate area of stippled hyperfluorescence was noted on FFA, which corresponded to the area of polyps on ICGA. Interestingly, in cases with the smoke-stack leak, a small micro retinal pigment epithelium (RPE) rip was identified on OCT. ICGA showed a cluster of polyps in all cases. The location of polyps was macular in 2 cases and extra macular in 3 cases. Case presentations of all the cases are detailed below.
Case 1:
A 52 year old female presented with the complaint of defective vision in Right eye (RE) for the past 6 months. She had history of injury in the left eye (LE) in childhood following which the eye had become phthisical. On examination, her BCVA in RE was 6/18. Anterior segment was within normal limits. Fundus examination revealed large amount of SRF almost extending from superior to inferior arcade vessels with a speck of SRH (Figure 1 A). OCT revealed a large, tall and notched PED; with a polyp beneath the undersurface of RPE and SRF. Enhanced depth imaging (EDI) revealed a thick choroid with subfoveal choroidal thickness (CT) of 406µm with dilated outer choroidal vessels (Figure 1 B). On FFA, smoke stack leak was noted superonasal to fovea(Figure 1 C,D) and ICGA revealed bunch of polyps inferior to fovea along with choroidal hyperpermeability in late phase corresponding to site of CSR leak (Figure 1 E,F). On reviewing the OCT, a micro RPE rip was also noted in the region corresponding to the CSR leak on FFA (Figure 1 G).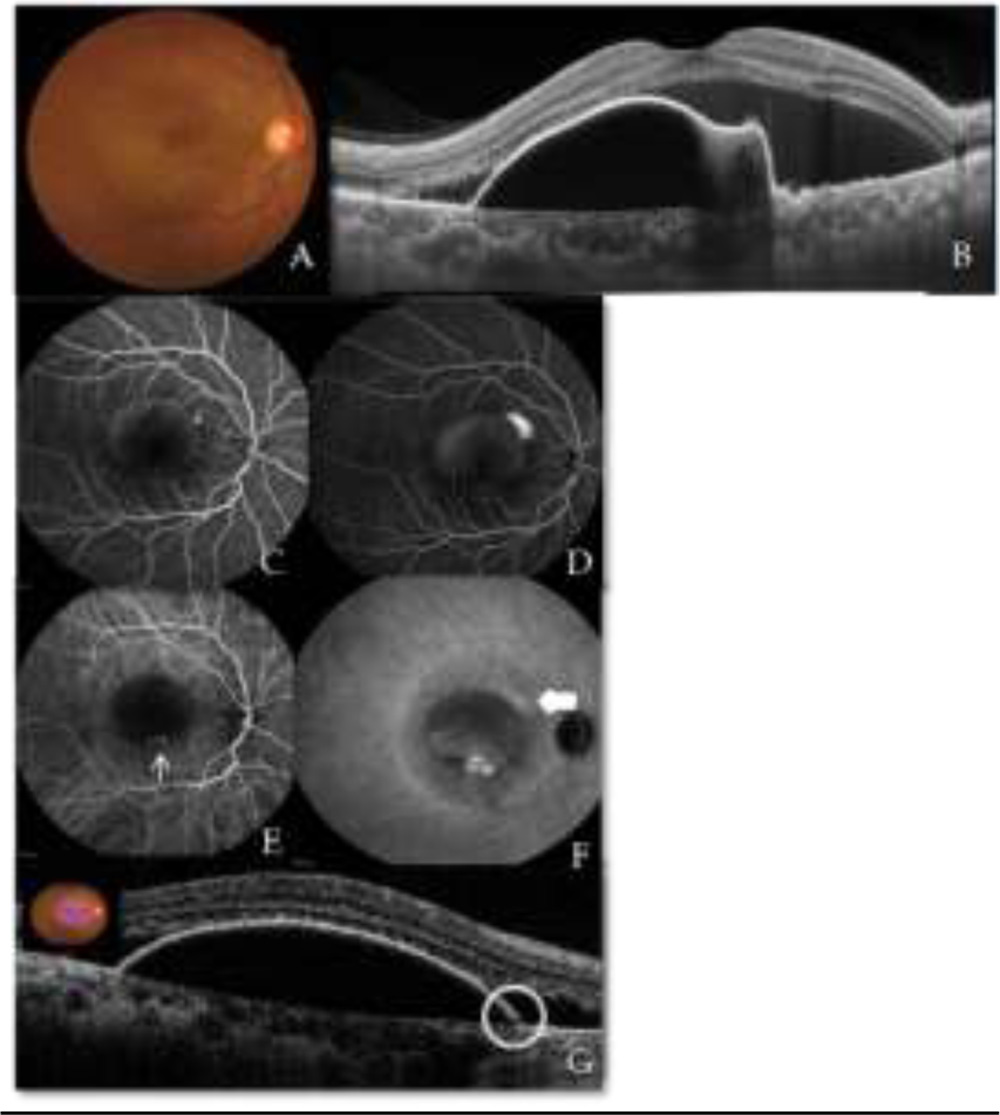 Figure 1 Case 1 A) Colour fundus photograph of RE showing subretinal fluid(SRF) with subretinal haemorrhage(SRH) B) EDI OCT image showing notched PED with SRF with subfoveal choroidal thickness(CT) of 406µm C) FFA early stage and D) FFA late stage showing a smoke-stack leak superonasal to fovea with gradual filling of dye in PED and a stippled hyperfluorescence inferior part of the PED E) ICGA early phase and F) ICGA late phase showing a bunch of polyps(thin arrow) in area of stippled hyperfluorescence and choroidal hyperpermeability(thick arrow) in late phase at the site of CSCR leak G) Micro RPE rip(circle) in area corresponding to smoke-stack leak
Figure 1 Case 1 A) Colour fundus photograph of RE showing subretinal fluid(SRF) with subretinal haemorrhage(SRH) B) EDI OCT image showing notched PED with SRF with subfoveal choroidal thickness(CT) of 406µm C) FFA early stage and D) FFA late stage showing a smoke-stack leak superonasal to fovea with gradual filling of dye in PED and a stippled hyperfluorescence inferior part of the PED E) ICGA early phase and F) ICGA late phase showing a bunch of polyps(thin arrow) in area of stippled hyperfluorescence and choroidal hyperpermeability(thick arrow) in late phase at the site of CSCR leak G) Micro RPE rip(circle) in area corresponding to smoke-stack leak
Case 2:
A 70 year old male presented with defective vision in the LE for past 2 months. He had been diagnosed as having occult CNVM in the LE and had received a single dose of IV Ranibizumab 1 month back. He had a history of vitreous haemorrhage in the RE and had undergone vitrectomy for the same 2 years back, but did not recover vision in that eye. He was on treatment for hypertension and ischemic heart disease. His BCVA was hand movements in RE and 6/12 in LE. Anterior segment examination showed immature cataract in both eyes. Fundus examination revealed a subfoveal scar in RE and a serous PED with SRH in the LE. OCT of RE showed foveal thinning with subretinal scar and dilated outer choroidal vessels with subfoveal CT of 401µ(Figure 2 A) and LE showed multiple irregular PED with a tall peaked PED with SRF and minimal SRH with dilated outer choroidal vessels and subfoveal CT of 464µm (Figure 2 B,C). FFA and ICGA of RE showed staining of the scar tissue whereas in the left eye, an extrafoveal ink blot leak was noted superotemporal to the fovea on FFA (Figure 2 D,E) along with multiple polyps at the fovea with late choroidal hyperpermeability at site of ink blot leak on ICGA (Figure 2 F,G).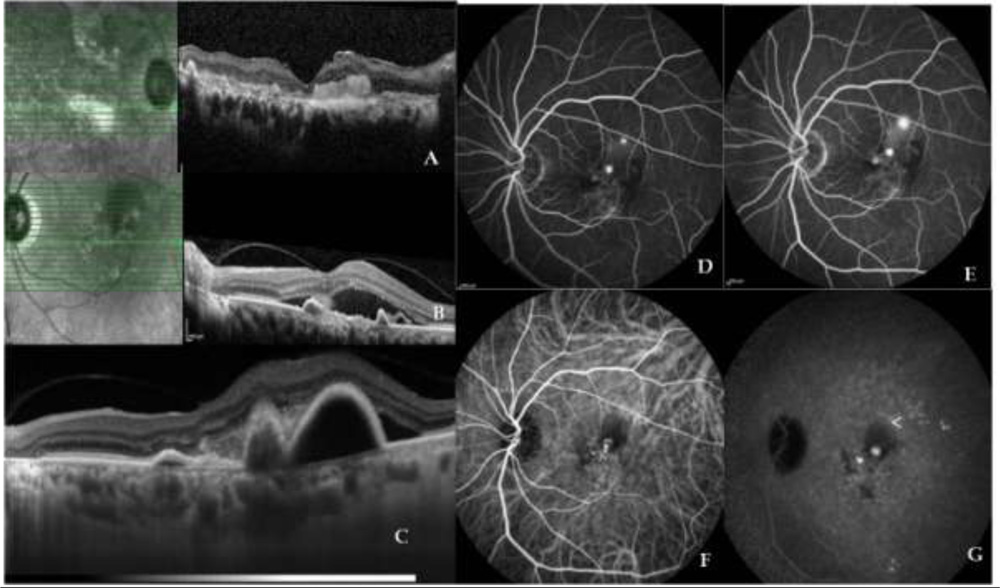 Figure 2 Case 2 A) RE OCT shows foveal thinning with subretinal scar and dilated outer choroidal vessels and subfoveal CT of 401µ B) LE OCT showing multiple irregular peaked PED and SRF with minimal SRH C) EDI OCT showing dilated outer choroidal vessels and subfoveal CT of 464µm D) FFA early phase and E) FFA late phase showing stippled hyperfluorescence with late leakage in foveal region and an extrafoveal ink blot leak superotemporal to fovea with blocked fluorescence temporal to fovea F) ICGA early phase G) ICGA late phase shows a bunch of polyps at the fovea and choroidal hyperpermeability(arrow head) at the site of ink blot leak
Figure 2 Case 2 A) RE OCT shows foveal thinning with subretinal scar and dilated outer choroidal vessels and subfoveal CT of 401µ B) LE OCT showing multiple irregular peaked PED and SRF with minimal SRH C) EDI OCT showing dilated outer choroidal vessels and subfoveal CT of 464µm D) FFA early phase and E) FFA late phase showing stippled hyperfluorescence with late leakage in foveal region and an extrafoveal ink blot leak superotemporal to fovea with blocked fluorescence temporal to fovea F) ICGA early phase G) ICGA late phase shows a bunch of polyps at the fovea and choroidal hyperpermeability(arrow head) at the site of ink blot leak
Case 3:
A 65 year old male had been diagnosed as having occult CNVM in the LE and was treated with PDT with bevacizumab in 2011. He had ischaemic heart disease. His BCVA was 6/9 in RE and 6/18 in LE. Anterior segment showed immature cataract in both eyes. On fundus examination, RE showed a serous PED while in the LE, SRH with SRF and minimal subretinal scarring was seen.OCT of RE showed a normal foveal contour with a serous PED and in LE, irregular PEDs with SRH was seen in the macular region and a large amount of SRF with a tall PED along inferior arcade was seen. In both eyes the choroid was thickened with CT being 300µm in RE and 357µm in LE. On FFA, some window defects with pooling of dye in PED were noted in RE, ICGA was normal. In the LE, a smoke stack leak was noted inferotemporal to macula and a RPE rip was seen still temporal to that with profuse leak on FFA and a cluster of polyps at the fovea on ICGA. OCT done through the smoke-stack leak area, showed a micro RPE rip corresponding to the leakage.
Case 4:
A 67 year old male presented with the complaint of defective vision in the LE since past 14 days. He was on treatment for diabetes mellitus and hypertension. His BCVA was 6/6 in RE and 6/18 in LE. Anterior segment examination was within normal limits in both eyes. Fundus examination revealed RPE changes at the fovea in the RE and PED with subretinal hard exudates in LE. OCT of RE showed RPE irregularity while LE showed SRF with hard exudates and a PED inferonasal to fovea along with fibrin .Double layer sign was present on OCT. The subfoveal CT in LE was 360µm. FFA and ICGA of RE were within normal limits. In LE, FFA showed an inkblot leak inferonasal to fovea and multiple polyps were noted superior to fovea on ICGA.
Case 5:
A 62 year old female presented with the complaint of defective vision in both eyes since 6 months. She was on treatment for systemic hypertension. On examination, her BCVA was 6/9 in both the eyes. She had immature cataracts in both the eyes. The retinal evaluation of RE showed a very large serous PED extending between the arcade vessels with subretinal deposits at edge of PED with SRF. LE showed some subretinal deposits temporal to macula and a large PED with subretinal deposits nasal to disc. OCT of RE showed a large PED with SRF with vitelliform deposits at the posterior pole and a large irregular PED with SRF superior to the disc with double layer sign. LE OCT showed RPE changes with irregular PEDs. OCT taken nasal to disc showed a micro RPE rip. In the left eye, although the subfoveal CT was 269µm, the sublesional CT was 423µm. FFA and ICGA of RE showed branched vascular network with polyps superior to disc where as in LE, along with branched vascular network and polyps superior to disc, a smoke stack leak was noted nasal to disc corresponding to the micro rip on FFA.
DISCUSSION
Pachychoroid10 is defined as abnormal and permanent increase in choroidal thickness which can be focal or diffuse and is generally associated with dilated outer choroidal vessels. The absolute choroidal thickness may be less in case the choriocapillaries and Sattler layer vessels are attenuated. Pachychoroid is associated with choroidal hyperpermeability on ICGA. Warrow et al11 described “Pachychoroid Pigment Epitheliopathy”(PPE) as retinal pigment epithelium (RPE) changes with reduced fundus tessellation, overlying a thick choroid but without any history of subretinal fluid. C. Pang and K. Fruend12 have described a type I choroidal neovascularization developing over focal areas of choroidal thickening and termed it as “Pachychoroid Neovasculopathy”(PNV).
The pathophysiologic mechanisms in PCV and CSR are poorly understood. Arteriosclerotic changes along with engorged vortex veins due to chronic choroidal venous congestion have been noted in PCV13. Choroidal hyperpermeability along with loss of RPE function secondary to choroidal hydrostatic pressure has been observed in CSR14. Pachychoroid features have been noted in both these entities as well.
Hence, the pathophysiology of pachychoroid spectrum of diseases can be explained as follows. In eyes with a pachychoroid, if the RPE is able to overcome the fluid overload, there might just be subtle pigmentary changes or small PED due to the stress on the RPE- called as Pachychoroid pigment epitheliopathy. If RPE is unable to overcome the fluid overload, a serous macular detachment results, in which case we see a CSR. In chronic stages due to the excess stress on RPE, micro rips may occur in the Bruch’s membrane. If these eyes develop a Type 1 choroidal neovascular membrane, a Pachychoroid neovasculopathy can be seen. And a long standing type1 CNVM can develop polyps or the dilated choroidal vessels may themselves develop polyps at the terminal ends to result in Polypoidal choroidal Vasculopathy11.(Figure 3)
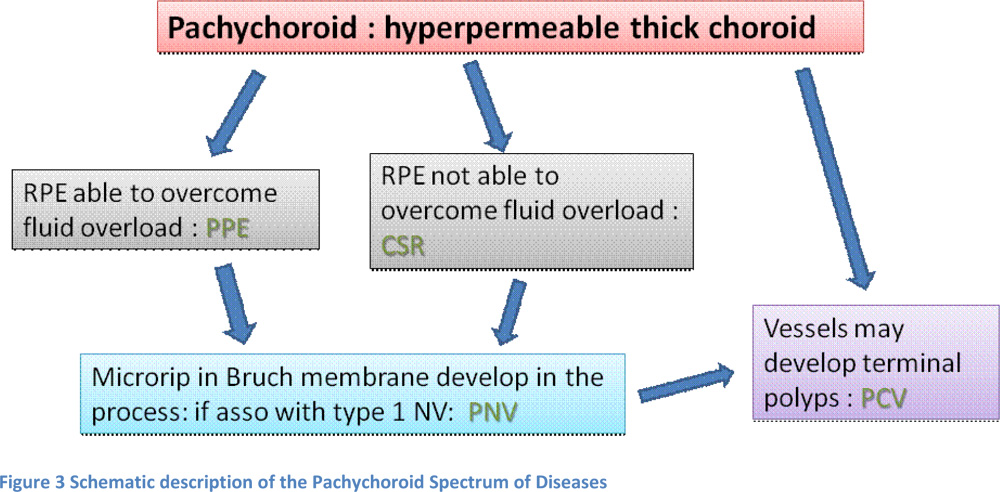 Here, we report 5 cases with features of active PCV along with active CSR in the same eye simultaneously along with a thickened choroid and dilated outer choroidal vessels. Findings suggestive of PPE associated with a pachyvessel on ICGA were also noted in one eye. Also, the findings are seen all over the fundus and not just confined to the posterior pole indicating an abnormality involving the entire choroid
Here, we report 5 cases with features of active PCV along with active CSR in the same eye simultaneously along with a thickened choroid and dilated outer choroidal vessels. Findings suggestive of PPE associated with a pachyvessel on ICGA were also noted in one eye. Also, the findings are seen all over the fundus and not just confined to the posterior pole indicating an abnormality involving the entire choroid
CONCLUSION
To the best of our knowledge, the coexistence of PCV, CSR and pachychoroid features has been unreported till now. This case series may provide direct evidence to the hypothesis that these diseases are associated and form a part of spectrum. However, larger longitudinal studies with longer follow-up are required to shed further light into the pathogenesis as well as progression of the entities belonging to this spectrum of diseases and may therefore help in planning better treatment protocols in the future.
References
- Yannuzzi L a, Sorenson J, Spaide RF, Lipson B. Idiopathic polypoidal choroidal vasculopathy (IPCV). Retina. 1990;10(1):1-8. doi:10.1097/00006982-199001010-00001.
- Gemenetzi M, De Salvo G, Lotery a J. Central serous chorioretinopathy: an update on pathogenesis and treatment. Eye (Lond). 2010;24(12):1743-1756. doi:10.1038/eye.2010.130.
- Fung AT, Yannuzzi L a., Freund KB. Type 1 (Sub-Retinal Pigment Epithelial) Neovascularization in Central Serous Chorioretinopathy Masquerading As Neovascular Age-Related Macular Degeneration. Retina. 2012;1(9):1. doi:10.1097/IAE.0b013e3182680a66.
- Toyama T, Ohtomo K, Noda Y, Ueta T. Polypoidal choroidal vasculopathy and history of central serous chorioretinopathy. Eye (Lond). 2014;28(8):992-997. doi:10.1038/eye.2014.132.
- Park HS, Kim IT. Clinical characteristics of polypoidal choroidal vasculopathy associated with chronic central serous chorioretionopathy. Korean J Ophthalmol. 2012;26(1):15-20. doi:10.3341/kjo.2012.26.1.15.
- Yannuzzi LA, Freund KB, Goldbaum M, et al. Polypoidal choroidal vasculopathy masquerading as central serous chorioretinopathy. Ophthalmology. 2000;107(4):767-777. doi:10.1016/S0161-6420(99)00173-6.
- Okushiba U, Takeda M. [Study of choroidal vascular lesions in central serous chorioretinopathy using indocyanine green angiography]. Nihon Ganka Gakkai Zasshi. 1997;101(1):74-82. http://www.ncbi.nlm.nih.gov/pubmed/9028111. Accessed July 10, 2016.
- Spaide RF, Hall L, Haas A, et al. Indocyanine green videoangiography of older patients with central serous chorioretinopathy. Retina. 1996;16(3):203-213. http://www.ncbi.nlm.nih.gov/pubmed/8789858. Accessed July 10, 2016.
- Sasahara M, Tsujikawa A, Musashi K, et al. Polypoidal Choroidal Vasculopathy with Choroidal Vascular Hyperpermeability. Am J Ophthalmol. 2006;142(4):601-607.e1. doi:10.1016/j.ajo.2006.05.051.
- Balaratnasingam C, Lee W-K, Koizumi H, Dansingani K, Inoue M, Freund KB. Polypoidal Choroidal Vasculopathy: A Distinct Disease or Manifestation of Many? Retina. 2016;36(1):1-8. doi:10.1097/IAE.0000000000000774.
- Warrow DJ, Hoang Q V, Freund KB. Pachychoroid Pigment Epitheliopathy. Retina. 2013;33(8):1659-1672. <Go to ISI>://WOS:000330233200022.
- Pang CE, Freund KB. Pachychoroid neovasculopathy. Retina. 2015;35(1):1-9. doi:10.1097/IAE.0000000000000331.
- Nakashizuka H, Mitsumata M, Okisaka S, et al. Clinicopathologic findings in polypoidal choroidal vasculopathy. Investig Ophthalmol Vis Sci. 2008;49(11):4729-4737. doi:10.1167/iovs.08-2134.
- Nicholson B, Noble J, Forooghian F, Meyerle C. Central Serous Chorioretinopathy: Update on Pathophysiology and Treatment. Surv Opthalmol. 2013;58(2):103-126. doi:10.1016/j.biotechadv.2011.08.021.Secreted.


 FP476 : PCV AND CSR: DISEASES OF THE SAME SPECTRUM?
FP476 : PCV AND CSR: DISEASES OF THE SAME SPECTRUM?