Dr. Umesh Chandra Behera, Dr Monica Budhwani, B18990
Authors:
Umesh Chandra Behera, MS
Monica Budhwani MBBS (presenting author)
Manas Ranjan Barik
Author’s institutional affiliation:
L V Prasad eye institute, Patia, Bhubaneswar, Odisha, India 751024;
Address correspondence:
Umesh Chandra Behera, L V Prasad eye institute, Patia, Bhubaneswar, Odisha, India 751024. Phone: 91-674-3987100; Fax: 91-0674-398 7130.
Email: umeshcbehera@gmail.com
Financial support: Hyderabad Eye Research Foundation
Conflict of interest: No conflicting relationship exists for any author
Abstract:
Title: Early vitrectomy in fungal endophthalmitis does not alter the visual and structural outcome
Purpose – whether early pars plana vitrectomy while sampling vitreous for culture alter the visual and structural outcome.
Methods – Retrospective chart review included 66 consecutive culture positive fungal endophthalmitis patients who were grouped either into group A (n=31) or group B (n=35) based on whether they underwent aggressive early vitrectomy or had diagnostic vitrectomy alone respectively.
Results– Log Mar vision improved in group A (p=0.0278) but post treatment visual outcomes in both groups showed no significant difference (p=0.322).Phthisis was seen in 25.80% in group A and 25.71% in B (p=0.993).
Conclusions– Early vitrectomy doesn’t significantly alter the functional and anatomical outcomes.
Introduction:
Fungal aetiology in endophthalmitis is relatively uncommon but potentially devastating both in terms of visual and structural outcome. Treatment is met with unique challenges, principally because the diagnosis is often challenging, choices of therapies available are limited and the outcomes are frequently unfavourable. As fungal agents like Aspergillus and Candida are most commonly isolated in fungal endophthalmitis and are known to run a fulminant course, early vitrectomy is often advocated.1-3 Butthe timing of vitrectomy is still unclear because of lack of evidence from randomised controlled trials.We aimed at looking at our 10year surgical outcome data and analysed retrospectively the visual and structural outcome when fungal endophthalmitis was treated promptly by vitrectomy at the time of vitreous sampling for microbiological identification.
Materials and methods
A retrospective review of the microbiological records with positive fungal culture from vitreous samples of suspected endophthalmitis cases at L V Prasad Eye Institute Bhubaneswar over 10years (2006-2015) was carried out. The demographic details, predisposing factors, clinical presentation, mode of management and treatment outcome were analyzed from the medical records. To compare the treatment outcome, the data was divided into two groups based on prompt or deferred vitrectomy during the vitreous sampling for pathogen identification. The study was approved by the institutional review board (IEC code: 2016-65-IM-13) and followed the tenets of the Declaration of Helsinki.
The study included fungus culture positive endophthalmitis cases. Follow up of less than 2 months and the patients whose charts were incomplete and/or lost to follow up were excluded. The patients were divided into two groups: Group A included patients who received a near complete vitrectomy and Group B included the patients who received only a diagnostic vitrectomy (undiluted vitreous collected for microbiological study using a vitreous cutter).
The primary objective was to study if prompt vitrectomy helped restore vision or maintain anatomical contour of eye. The secondary objective was to review the spectrum of fungal affection in endophthalmitis in the given cohort.
Endophthalmitis management:
Each of the clinically suspected endophthalmitis case based on the history and clinical presentation was categorized into one of the endophthalmitis types – post-operative, traumatic or endogenous. The decision for vitrectomy in endophthalmitis was based on the presence of B-scan documented vitreous opacities or membranes with a reasonably clear cornea. The cases with poor corneal clarity and the ones with milder vitritis received only vitreous biopsy and injection of anti-microbials within 12hrs of presentation without any additional vitrectomy. Moreover, if meticulous clearing of anterior segment exudates through a corneal paracentesis did not improve posterior segment visualization, the surgery was limited to vitreous biopsy and injection of anti-microbials alone. The ones with good view of posterior segment had vitrectomy in addition to vitreous sampling and intravitreal antibiotic injections.
Vitreous biopsy was performed by a standard 20G or 23G vitreous cutter. Approximately 0.2 to 0.3ml of vitreous sample was collected for microbiological culture and smear. Empirical intravitreal antibiotic combination was – vancomycin 1gm, ceftazidime 1.25gm and dexamethasone 0.4gm each in 0.1ml. Subjects who had a known history of vegetable matter trauma and/or had progression to endophthalmitis from traumatic fungal keratitis received intravitreal amphotericin B 0.05mg or voriconazole 0.1mg along with the empirical intravitreal antibiotics; intravitreal dexamethasone was withheld. The choice of the anti-fungal agent was at the discretion of the operating surgeon. Subjects receiving intravitreal anti-fungals were additionally treated with systemic anti-fungals (table 1) soon after vitreous sampling. The ones who grew fungus on culture received systemic antifungals only after microbiological confirmation. Systemic antifungals were continued for a minimum of 2 months from the day of initial vitreo-retinal intervention with periodic monitoring of liver function.
Fungal identification:
The smear for fungus detection was done with a wet mount of 10% potassium hydroxide (KOH) along with 1% calcofluor white and was examined under a fluorescent microscope. At least two different media were chosen for pathogen identification. The selective media chosen was sabouraud’s dextrose agar (SDA) and the non-selective media were chocolate, blood agar and brain heart infusion broth. They were incubated in room temperature between 250 and 370 Celsius. The specimen was incubated for at least 4 weeks before reporting it negative. The yeasts and the moulds were distinguished from each other under direct microscopy. Mycological diagnosis was considered only when both smear and the culture were positive for fungus. The specimen was labeled as culture positive if the same fungus had grown on two or more solid media at the inoculation site or had grown on one solid media and was positive on smear stained with gram, giemsa or Gomorimethenamine silver stain.
The cultured media were observed daily for the growth of fungi. The colony morphology was recorded. Fungal growths on media other than SDA were usually sub-cultured on SDA for the formation of typical fungal growth and sporulation.
Filamentous fungal species were identified based on rate of growth, colony morphology (color, texture, consistency) and pattern of sporulation. The types of spores were studied by examining a lactophenol cotton blue preparation of the colony. Prolonged incubation (up to one month) was sometimes required to obtain appropriate spores for identification. Slide culture was done to study the pattern of sporulation.
Statistical analysis:
Statistical analysis was done using the InStat statistical software version 3.0 (GraphPad software Inc, CA, USA). Data were expressed as mean± standard deviation (SD) for continuous variables, were compared by means of Student paired t-test and frequencies for categorical variables were compared through chi-square test. Statistical significance was set at 0.05 for all comparisons.
Results:
During the 10yrs of study period 970 subjects suspected of endophthalmitis were routinely subjected to bacterial and fungal culture. Only 7.1% (n=69) of them had a positive fungus growth on culture of vitreous fluid (66 met the inclusion and exclusion criteria). Among the 3 that were excluded, 2 did not have required follow up and one had information missing in the records. There were 31 and 35 subjects in group A and B respectively based on whether they received early near complete vitrectomy or a diagnostic vitrectomy alone as first line
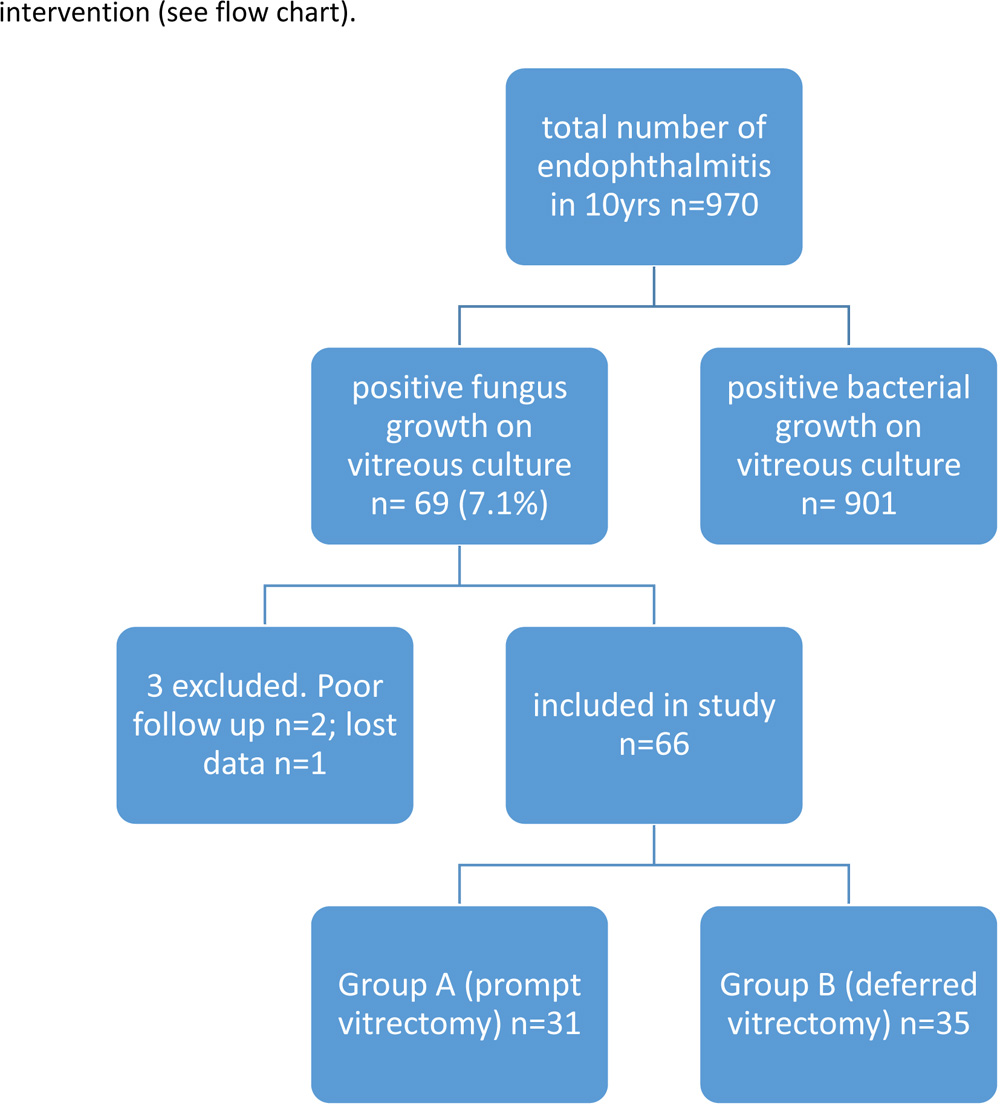 The median age was 56yrs (range6-85 yrs) in group A and52yrs in group B (range 8-82 yrs). The mean duration of symptoms at presentation was significantly longer (p = 0.0338, unpaired t-test)in early vitrectomy group (53.48 days) than in the deferred vitrectomy group (24.46 days).The incidence of diabetes in group A was higher and likewise was the associated systemic illness at presentation(table 1).
The median age was 56yrs (range6-85 yrs) in group A and52yrs in group B (range 8-82 yrs). The mean duration of symptoms at presentation was significantly longer (p = 0.0338, unpaired t-test)in early vitrectomy group (53.48 days) than in the deferred vitrectomy group (24.46 days).The incidence of diabetes in group A was higher and likewise was the associated systemic illness at presentation(table 1).
The ocular symptoms and signs in group A Vs B – Pain (54.83% Vs 60%), lid edema (48.38% Vs 48.57%), conjunctival congestion (87.09% Vs 94.28%) and corneal involvement (87.09% Vs 80%) – were comparable.
The proportion of post operative (54.83% Vs 51.42%), traumatic (29.03% Vs 34.28%) and endogenous (16.12% Vs 14.28%) endophthalmitis in each group were similar. Clinical and ultrasonographic evidence of vitreous hemorrhage in traumatic fungal endophthalmitis was seen in 3.22% of group A against 2.85% in group B. Slit-lamp biomicroscopic documentation and in doubtful instances the ultrasonographic evidence of the lens status showed a proportionate distribution of eyes with an intraocular lens at presentation (table 1). Media haze that required B-scan ultrasonography was noted in 87.09% of patients in group A and in 88.5% of patients in group B. Patients in both the groups divided on the basis of the clinical diagnosis showed a similar pattern of distribution (table 1). While all the subjects in group A received near complete vitrectomy at presentation, only 14.28% in group B received the same and the mean delay in vitrectomy from presentation was 18.8 days (range 7-30days). The mean follow up in group A (11.61months) was longer than group B (9.37months) but were comparable (p=0.06000).
The mean best corrected log MAR vision was 2.534 (range 20/125 to no light perception) in patients of group A and 2.336 (range 20/25 to NPL) in group B at presentation and was comparable in both the groups (p=0.189, unpaired t-test). The mean best corrected log MAR vision at last visit was 2.153 (range 20/30 to NPL) in group A and 2.367 (range 20/20 to NPL) in group B. Prompt vitrectomy showed significant improvement in vision (p=0.027, paired t-test) but when compared with the final visual outcome in the deferred vitrectomy group it was not superior (p=0.322, unpaired t-test).
Complications such as retinal detachment and phthisis (table 1) were noted in comparable number of cases in both groups (p=0.993, Chi-square test with yates correction). Proportionately more number of subjects had painful unsalvageable eyes that required evisceration in the deferred vitrectomy group.
Table 1: Comparative demographics, clinical and surgical data
| Parameters | Group A | Group B | p value/total |
| Age (median) | 56yrs | 52yrs | |
| Symptom duration (mean) | 53.48days | 24.46days | P=0.0338 |
| ocular signs and symptoms | |||
| Lid edema | 48.38% | 48.57% | P=0.9881 |
| Conjunctival congestion | 87.09% | 94.28% | P=0.5586 |
| Corneal involvement | 87.09% | 80% | P=0.6882 |
| pain | 54.83% | 60% | P=0.8619 |
| Systemic illness | |||
| Diabetes Mellitus | 12.9% | 2.85% | |
| Hypertension | 6.45% | 2.85% | |
| DM+HTN | 6.45% | 0% | |
| others | 3.22% | 5.71% | |
| none | 70.96% | 88.57% | |
| Lens status | |||
| Phakic | 41.91% | 34.28% | |
| Pseudophakic | 51.61% | 48.57% | |
| Aphakic | 6.45% | 11.42% | |
| No view | 0% | 5.71% | |
| Type of endophthalmitis | |||
| Post-operative | 54.83% | 51.42% | 53.03% |
| traumatic | 29.03% | 34.28% | 31.82% |
| endogenous | 16.12% | 14.28% | 15.15% |
| Mean LogMAR visual acuity | |||
| Pre-operative | 2.534 | 2.336 | P = 0.189 |
| Last follow up | 2.153 | 2.367 | P = 0.322 |
| Visual acuity in snellens’ better than or equal to 20/200 | |||
| First visit | n=1 | n=4 | 0.4290 |
| Final visit | 5 | 5 | 0.8349 |
| Treatment | |||
| Intravitreal antifungal after obtaining culture positivity | |||
| Ampho B | 12 | 13 | 25 |
| Voriconazole | 14 | 12 | 26 |
| None | 5 | 10 | 15 |
| Systemic antifungal | |||
| fluconazole | 2 | 3 | |
| ketoconazole | 13 | 6 | |
| voriconazole | 4 | 3 | |
| Itraconazole | 7 | 10 | |
| None | 3 | 13 | |
| Pars plana vitrectomy | |||
| PPV | 100% | 14.28% | |
| Structural Outcome | |||
| Phthisis | 25.8% | 23.71% | P=0.9932 |
| Evisceration | 3.22% | 11.42% | p=0.4290 |
| Retinal detachment | 3.22% | 2.85% | P=0.9144 |
The spectrum of mycotic agents that caused endophthalmitis is listed in the table 2 and the distribution in various types of endophthalmitis is listed in table 3. Aspergillus species was the most common mycotic pathogen isolated in this series; distribution was comparable in both groups. It also dominated in the post-operative group. Unidentified dematiaceous fungi were most commonly isolated in traumatic fungal endophthalmitis.
Table 2: Distribution of mycotic spectrum
| Fungal Agents | Patient Groups | ||
| Hyaline filamentous fungi | Group A | Group B | Total |
| Aspergillus terreus | 5 (16.12%) | 4 (11.42%) | 9(13.64%) |
| Aspergillus flavus | 3 (9.67%) | 7 (20%) | 10(15.15%) |
| Aspergillus niger | 1 (3.22%) | 1 (2.85%) | 2(3.03%) |
| Aspergillus fumigatus | 4(12.90%) | 3 (8.57%) | 7(10.60%) |
| Fusarium solani | 1(3.22%) | 0 | 1(1.51%) |
| Fusarium spp. | 2 (6.45%) | 3 (8.57%) | 5(7.57%) |
| Acremonium spp. | 2 (6.45%) | 5 (14.28%) | 7 (10.60%) |
| Unidentified hyaline fungus | 2 (6.45%) | 3 (8.57%) | 5(7.58%) |
| Dematiaceous filamentous fungi | |||
| Cladosporium spp. | 2 (6.45%) | 1 (2.85%) | 3(4.55%) |
| Drechslera spp. | 0 | 1 (2.85%) | 1 (1.52%) |
| Unidentified dematiaceous fungus | 4 (12.90%) | 5 (14.28%) | 9 (13.64%) |
| Yeast and yeast like fungi | |||
| Candida albicans | 2 (6.45%) | 1(2.85%) | 3 (4.55%) |
| Candida spp. | 1 (3.22%) | 1 (2.85%) | 2 (3.03%) |
| Others | |||
| Scedosporiumapiospermum | 1 (3.22%) | 0 | 1 (1.52%) |
| Sporotrichumspp. | 1 (3.22%) | 0 | 1 (1.52%) |
Table 3: Fungal etiology in various types of endophthalmitis
| Fungal Agents | Post-operative | Trauma | Endogenous |
| Hyaline filamentous fungi | |||
| Aspergillus terreus | 7 | 2 | 0 |
| Aspergillus flavus | 5 | 2 | 3 |
| Aspergillus niger | 2 | 0 | 0 |
| Aspergillus fumigatus | 3 | 3 | 1 |
| Fusarium solani | 1 | 0 | 0 |
| Fusarium spp. | 3 | 1 | 1 |
| Acremonium spp. | 3 | 3 | 1 |
| Unidentified hyaline fungus | 5 | 0 | |
| Dematiaceous filamentous fungi | |||
| Cladosporium spp. | 1 | 1 | 1 |
| Drechslera spp. | 0 | 1 | 0 |
| Unidentified dematiaceous fungus | 2 | 6 | 1 |
| Yeast and yeast like fungi | |||
| Candida albicans | 2 | 0 | 1 |
| Candida spp. | 0 | 1 | 1 |
| Others | |||
| Scedosporiumapiospermum | 0 | 1 | 0 |
| Sporotrichumspp. | 1 | 0 | 0 |
| TOTAL | 35 | 21 | 10 |
A positive history of trauma with vegetable matter was found in 85.71% (n=18) of cases of traumatic endophthalmitis. Likewise, 60% (n=6) of endogenous endophthalmitis had a history of febrile illness for which hospitalization was required prior to the onset of ocular symptoms. Two out of 6 had grown E. Coli on culture from the aspirates – one had abscess appendix and the other had duodenal fistula and intrahepatic stone.
Discussion:
Despite availability of safer and better antifungal agents4,5 and improvements in vitrectomy procedures6 the standard treatment for fungal endophthalmitis is still elusive, although a combination of vitrectomy and antifungal agents appear to be the best therapy. The combined procedure result in better visual and anatomical outcomes, but the timing of vitrectomy is not optimised yet. Furthermore, the institution of antifungal treatment is often delayed until microbiological reports are available, as it is difficult to clinically distinguish fungal from bacterial endophthalmitis. Only a small case series with an early and aggressive approach with vitrectomy in recent times had shown vision recovery in all their cases of fungal endophthalmitis.7 Rest of the studies that have stated a better visual recovery with vitrectomy either have not mentioned the timing of vitrectomy or had performed it as in the course of management. The ones with a favourable visual outcome (better than 20/200) with vitrectomy are listed in table 4.
Table 4: favorable visual outcomes reported in various studies7-16
| Article
|
type of cases included
|
case number
|
vision better than 20/200
|
| Yang YS 20095
|
post operative fungal endophthalmitis
|
7 | 100% |
| Narang S 20016
|
post operative fungal endophthalmitis
|
27 | 29.6% |
| Pettit TH 19807
|
post operative fungal endophthalmitis
|
13 | 15.4% |
| Majji AB 19998
|
post operative fungal endophthalmitis
|
11 | 27% |
| Mc Guire TW 19919
|
endogenous fungal endophthalmitis
|
17 | 17.6% |
| Zhang YQ 200510
|
endogenous fungal endophthalmitis
|
20 | 40% |
| Birnbaum FA 201611
|
endogenous fungal endophthalmitis
|
5 | 80% |
| Takebayashi H 200612
|
endogenous fungal endophthalmitis
|
29 | 44% |
| Shen X 200913
|
endogenous fungal endophthalmitis
|
29 | 38% |
| Lingappan A 201214
|
endogenous fungal endophthalmitis
|
65 | 50.76% |
| Majji AB 19998 | post trauma fungal endophthalmitis
|
9 | 56% |
| Our study | Post-operative(17), endogenous(5), traumatic(9) fungal endophthalmitis | 31 | 16.1% |
Our results of early vitrectomy were not as encouraging as the reported successes in previous studies.7-16 Also when compared with the results of deferred vitrectomy we did not get a statistically significant difference even when the parameters in both groups were alike.
Vitrectomy is often delayed due to various factors like corneal clarity and the delay in obtaining a positive fungal culture. Thus, the decision for early vitrectomy at the time of vitreous sampling, if at all, is driven by the severity of vitreous involvement and vision, not on the
microbiological affection. This study cohort followed the same cues of severe vitreous involvement for early vitrectomy. The treatment aim in general was to debulk the vitreous cavity at the earliest opportunity. The Endophthalmitis Vitrectomy Study treatment protocol was followed while treating post operative endophthalmitis,17 whereas a primary vitrectomy during the vitreous sampling was attempted in endogenous and traumatic ones. Media clarity that precluded a good fundus view for vitrectomy and a milder inflammation of vitreous at presentation were the reasons for deferred vitrectomy. Our study showed corneal involvement (a potential cause of delayed vitrectomy) in comparable number of cases in both groups. The distribution of post operative endophthalmitis in both groups was also analogous. Hence the decision for timing the vitrectomy may not be implicated as a confounder for treatment outcome in our cohort. The other risk factors that determine the final visual outcome in fungal endophthalmitis include trauma and infection by Aspergillus species.18 Our series had comparable proportion of traumatic endophthalmitis ( group A 29.03%; group B 34.28%) and Aspergillus spp affection in both groups – quite supportive of the fact that they did not influence the study outcome.
As reported by one of the widely quoted series from India, vitrectomy in the setting of post-operative fungal endophthalmitis result in good visual outcome over no vitrectomy but are not statistically significant.8 Our results also showed similar outcome of significant improvement in mean visual acuity with prompt vitrectomy but did not reach statistical significance when compared with deferred vitrectomy (table 5). In contrast to the other study the spectrum of fungal involvement in our series encompassed all forms of fungal endophthalmitis (postoperative, endogenous and traumatic), and vitrectomy was done as a primary procedure for management. A prompt vitrectomy did reduce the risk of evisceration in our series though it was not statistically significant. Progression to phthisis in both groups was comparable.
Table 5: comparison of visual acuity outcome
| GROUP A prompt vitrectomy
n =31 |
GROUP B deferred vitrectomy
n= 35 |
||
| pre op vision (mean VA) | 2.534 | 2.336 | P = 0.189 |
| post-op vision (mean VA) | 2.153 | 2.367 | P = 0.322 |
| P = 0.027 | P = 0.844 |
The results reaffirm the notion that fungal inoculation in eye although ominous is slow in progression and a delay in vitrectomy may not alter the final outcome. As fungus culture is time consuming, vitrectomies may thus be deferred if poor visibilities of posterior segment of eye preclude a safe surgery. Significant reduction in risk of retinal detachment (RD) with early vitrectomy has been shown in one series5 but our series did not find any such advantage. The post vitrectomy RD in group A was in post-operative endophthalmitis and in group B it was in traumatic endophthalmitis. Based on the findings from this study vitrectomy could be delayed until the surgical procedure is deemed safe.
Since the major limitation of this study was its’ retrospective, nonrandomized and uncontrolled design the conclusions drawn from this study could not be generalized. As a prospective study is rather not possible in fungal endophthalmitis given its’ rarity, the inference drawn from this study could be used as a guide to fungal endophthalmitis treatment and thus avoid the complications related to vitrectomy in endophthalmitis.
The other objective of this study was to look at the prevalence and the spectrum of fungal affection at our center, one of the referral eye hospitals in eastern India. To the best of our knowledge this is the largest reported case series of fungal endophthalmitis from India based on the culture positivity from vitreous samples. The other large series published from north India18 had cases included based on clinical presentation, positive smear with/without culture positivity. The overall incidence of fungal endophthalmitis at our center (7.1%) was within the reported range of incidences in previous studies from India (Chakrabarti A 4.1%; Gupta A 21.5%; Kunimoto DY 16.7%; Sharma S 12.1%;).18-21 Intra-ocular surgery was the commonest cause (53.03%) of fungal endophthalmitis followed by traumatic and endogenous. Only one in this series was our in-house complication following cataract surgery. All rest was referred from diverse surgical settings ranging from private clinics to major ophthalmic institutes with no incidences of cluster infection. Lapse in stringent surgical practices could be ascribed to such an occurrence.
Our series explored the fungal infection spectrum in endophthalmitis specific to eastern India that spanned over a decade and confirmed prior reports of Aspergillus species predominance in fungal endophthalmitis.8,18Aspergillusflavuswas the most common isolated species in our series (15.15%). In this series Aspergillus and Fusariumwere isolated in endogenous endophthalmitis notCandida spp.as reported by others.22,23A similar trend of the predominance of molds over yeasts was also noted in traumatic and endogenous endophthalmitis. The geographical location and tropical climate may have been responsible for such a pattern of fungal involvement in endophthalmitis.
References:
- Essman TF, Flynn HW Jr, Smiddy WE, Brod RD, Murray TG, Davis JL, Rubsamen PE. Treatment outcomes in a 10-yr old study of endogenous fungal endophthalmitis. Ophthalmic surg lasers 1997;28(3):185-194
- Gupta A, Gupta V, Dogra MR, Chakrabarti A, Ray P, Ram J, Patnaik B. Fungal endophthalmitis after a single intravenous administration of presumably contaminated dextrose infusion fluid. Retina 2000;20(3):262-268
- Kim DY, Moon HI, Joe SG, Kim JG, Yoon YH, Lee JY. Recent clinical manifestation and prognosis of fungal endophthalmitis: A 7yr experience at a tertiary referral center in Korea. J Korean Med Sci 2015;30(7):960-964
- Hariprasad SM. Mieler WF, Lin TK, Sponsel WE, Graybill JR. Voriconazole in the treatment of fungal eye infections: a review of current literature. Br J Ophthalmol 2008;92:871-878
- Riddell 4th J, Comer GM, Kauffman CA. Treatment of endogenous fungal endophthalmitis: focus on new antifungal agents. Clin Infect Dis 2011;52:648–53
- Androudi S, Praidau A, Symeonidis C, Tsironi E, Iaccheri B, Fiore T, Tsinopoulos I, Brazitikos P. Safety and efficacy of small incision, sutureless pars plana vitrectomy for patients with posterior segment complications secondary to uveitis. Acta Ophthalmol 2012;90(5):e409-e410
- Yang YS. Results of extensive surgical treatment of seven consecutive cases of postoperative fungal endophthalmitis. Korean J Ophthalmol. 2009;23(3):159-63
- Narang S, Gupta A, Gupta V, et al. Fungal endophthalmitis following cataract surgery: clinical presentation, microbial spectrum and outcome. Am J Ophthalmol 2001;132:609–617
- Pettit TH, Olson RJ, foos RY, Martin WJ. Fungal endophthalmitis following intraocular lens. Arch Ophthalmol 1980;98:1025-39
- Majji AB, Jalali S, Das T, Gopinathan U. Role of intravitreal dexamethasone in exogenous fungal endophthalmitis. Eye 1999;13:60-5
- Mc Guire TW, Bullock JD, Bullock JD Jr, Elder BL, Funkhouser JW. Fungal endophthalmitis. An experimental study with a review of 17 human ocular cases. Arch Ophthalmol 1991;109(9):1289-96
- Zhang YQ, Wang WJ. Treatment outcomes after pars plana vitrectomy for endogenous endophthalmitis. Retina 2005;25(6):746-50
- Birnbaum FA, Gupta G. The role of early vitrectomy in the treatment of fungal endogenous endophthalmitis. Retin Cases Brief Rep 2016;10(3):232-5
- Takebayashi H, Mizota A, Tanaka M. Relation between stage of endogenous fungal endophthalmitis and prognosis. Graefes Arch Clin Exp Ophthalmol 2006;244(7):816-20
- Shen X, Xu G. Vitrectomy for endogenous fungal endophthalmitis. OculImmunolInflam 2009;17(3):148-52
- Lingappan A, Wykoff CC, Albini TA, Miller D, Pathengay A, Davis JL, Flynn HW Jr. Endogenous fungal endophthalmitis: causative organisms, management strategies and visual acuity outcomes. Am J Ophthalmol 2012;153(1):162-6
- Results of Endophthalmitis Vitrectomy Study. A randomized trial of immediate vitrectomy and of intravenous antibiotics for the treatment of postoperative bacterial endophthalmitis. Endophthalmitis Vitrectomy Study Group. Arch Ophthalmol 1995;113:1479-96
- Chakrabarti A, Shivaprakash MR, Singh R, Tarai B, George VK, Fomda BA, Gupta A. Fungal endophthalmitis: fourteen years’ experience from a center in India. Retina 2008; 28: 1400-7
- Gupta A, Gupta V, Gupta A, Dogra MR, Pandav SS, Ray P, Chakraborty A. Specrum and clinical profile of post cataract surgery endophthalmitis in north India. Indian J Ophthalmol 2003;51(2):139-45
- Kunimoto DY, Das T, Sharma S, et al. Microbiologic spectrum and susceptibility of isolates. Part I-postoperative endophthalmitis. Endophthalmitis Research Group. Am J Ophthalmol 1999;128(2):240-242
- Sharma S, Padhi TR, Basu S, Kar S, Roy A, Das T. Endophthalmitis patients seen in a tertiary eye care centre in Odisha: a clinic-microbiological analysis. Indian J Med Res 2014;139(1):91-98
- Schiedler V, Scott IU, Flynn HW Jr, Davis JL, Benz MS, Miller D. Culture proven endogenous endophthalmitis: clinical features and visual acuity outcomes. Am J Ophthalmol 2004;137(4):725-31
- Lingappan A, Wykoff CC, Albini TA, Miller D, Pathengay A, Davis JL, Flynn HW Jr. Endogenous fungal endophthalmitis: causative organisms, management strategies and visual acuity outcomes. Am J Ophthalmol 2012;153(1):162-6

