Dr. Archana Anand Pawar, Dr. Sri Ganesh, Dr. Sheetal Brar
Presenting author – DrArchana Pawar
Co-authors -Dr Sri Ganesh, Dr Sheetal Brar
FINANCIAL INTEREST – None of the authors have any financial interest in the products / materials discussed in the study.
Abstract
Purpose–
To compare results of T-ICL,Fs-LASIK and ReLex SMILE for low to moderate myopic astigmatism.
Methods–
30eyes in each group (Gp A-T-ICl,GpB-FS-LASIK,GpC-RELEX SMILE) with SE -3to -8D and cylinder -0.75to – 3D were included.At 1,6 and12months postop visual acuity,refraction, contrast sensitivity, abberometry, specular microscopy and dry eye evaluation was done.
Results-better UCVA was seen in GpC followed by GpA and B(p=0.115).Safety,efficacy indices were highest in Gp A(1.24 and 1.12 ).93% had postop cylinder within ±0.5D in GpC compared to 90% each in GpA and C.SE was within ±0.5D in 90%in GpA ,77% in GpB and 93% in GpC.contrast sensitivity scores were highest in GpA. while TBUT had reduced in GpB significantly. Aberrations increased in GrpB and C while reduced in GpC. specular count had non significantly reduced GrpA.
Conclusion–
refractive results with TICL were similar to ReLEx SMILE.With superior safety, efficacy and better contrast, aberrations and dry eye profile,T-ICL has an edge over corneal procedures for correction of low to moderate myopic astigmatism
KEYWORDS- Toric implantable collamer lens, femtosecond LASIK, ReLEx SMILE, myopic astigmatism
CTRI Registration number- CTRI/2014/04/004537
Introduction
Modern refractive surgery provides various modalities for the surgical correction of myopia and myopic astigmatism depending upon the grade of refractive error. Generally, an excimer based corneal procedure such as LASIK or PRK is preferred for low to moderate myopia, while an Implantable collamer lens (ICL) is indicated for higher degrees of myopia, outside the safety limit of corneal correction (1, 2). Although the latter is FDA approved to correct myopia ranging from -3.00 to -20.00 diopters (D), being an intraocular procedure, its use is generally restricted to patients with high refractive error beyond the range of corneal procedures to prevent the risk of corneal ectasia(3).However, the validity of use of ICL in lower degrees of myopia and myopic astigmatism as a primary treatment modality, has already been verified (4).
Both LASIK and Toric ICL have been shown to be safe and effective for the correction of myopic astigmatism in various studies(5-8). It was found that LASIK was effective for correction of astigmatism upto 4.5 D(6), while Toric –ICL provided safe and stable results with astigmatism upto 7D in the long term studies(8).
ReLEx SMILE has been introduced as a relatively new, all femtosecond laser procedure for correction of myopia and myopic astigmatism, in which an intrastromal lenticule is extracted through a small incision(2-4mm). Various studies have suggested that this modality is a safe and effective method to correct varying degrees of myopia, with the advantages of minimum post-operative discomfort and restrictions, less dry eye and probably better biomechanics compared to LASIK (9,10).However LASIK has shown better outcomes than ReLEx SMILE for correction of astigmatism, most likely due to non-availability of iris registration and eye tracker in the current version of the femtosecond laser used for the ReLEx SMILE procedure(11,12). However, similar comparison studies have not been reported between Toric ICL and ReLEx SMILE.
Astigmatism correction with refractive surgery still remains the most difficult part. Whether astigmatism should be corrected on the cornea or inside the eye with a Toric ICL is also a matter of ongoing debate, especially in eyes absolutely suitable for a corneal procedure.
In this study, three currently available and popular surgical modalities – Toric ICL, Femtosecond LASIK and ReLEx SMILE for the treatment of low to moderate myopic astigmatism were evaluated. We compared visual and refractive outcomes, safety, efficacy, predictability and stability of astigmatic correction in this one year prospective clinical trial.
MATERIAL AND METHODS
This was a prospective, interventional, non- randomized, single centre study which was approved by institutional ethics committee and was performed in accordance with the tenets of the Declaration of Helsinki. All study participants provided written informed consent and ensured a long term follow –up.
30 eyes from 30 patients undergoing bilateral surgery with either of the three procedures – Toric ICL(Group A) , Femto-LASIK(Group B) or ReLEx SMILE(Group C) were included in the study, for correction of low to moderate myopic astigmatism . To ensure uniformity and eliminate bias in the interpretation of results, one eye from each patient was included.
Inclusion criteria were age between 21- 40 years, myopic astigmatism within the range of -3 to -8 D spherical equivalent (SE) with a minimum astigmatism of -0.75 D, stable refraction (<0.5 D change in past 12 months), corrected distant visual acuity(CDVA) of 20/30 or better, healthy tear film and ocular surface, absence of corneal ectatic diseases, corneal scars , absence of any retinal pathology and assured follow-ups. Patients using soft and rigid contact lenses were instructed to discontinue their lenses at least one and 3 weeks respectively, prior to the topographic evaluation. For patients planned for T-ICL, a minimum anterior chamber depth of 2.8 mm and endothelial cell count of 1500 cells/mm2 was ensured.
Patients with unstable refraction, pregnancy or using medications such as hormonal preparations, anti-depressants, oral steroids and immuno-suppressants were excluded from the study.
Preoperative evaluation
Preoperative evaluation included anterior and posterior segment examination, assessment of uncorrected and corrected distance visual acuity, corneal topography using Pentacam HR (Oculus Optikgeräte GmbH, Wetzlar, Germany) ) and Orbscan II (Bausch & Lomb, Rochester, NY),contrast sensitivity using Functional Acuity Contrast Test.(F.A.C.T. Stereo Optical Co.Inc. Chicago), abberometry (iTrace; Tracey Technologies, Houston, TX ), specular microscopy ( Tomey, Japan) and dry eye evaluation (Schirmer 1 and tear film break up time).
Surgical technique
All surgeries were performed by a single experienced refractive surgeon(S.G.)under topical anesthesia.
Toric ICL (Group-A)
T-ICL power calculation was performed by the manufacturer (STAAR Surgical) using a modified vertex formula. The size of the ICL was selected on the basis of the horizontal corneal diameter and anterior chamber depth measured with scanning-slit topography (Orbscan IIz). All patients underwent implantation of the Visian Toric- ICL V4c model(STAAR Surgicals, Monrovia, USA) using the standard surgical technique(13). Preoperatively 0-180 degree axis was marked at the slit lamp. Following this, T-ICL was inserted through a temporal, 2.8 mm limbal incision and carefully positioned posterior to iris using a Vukich’s manipulator in the intended axis as per the rotation diagram provided by the manufacturer.
Femto- LASIK(Group B)
Visumax femtosecond laser (Carl Zeiss MeditecAG) was used to create superior hinged flaps with 100-120 microns thickness,flap diameter 7.5-8.0 mm, optical zone between 6-7mm and a transition zone of 8.2mm. The ablation was performed using an MEL 90 excimer laser (Carl ZeissMeditec AG) with iris-recognition software using the iris pattern image taken preoperatively with a Wavefront Aberration Supported Corneal Ablation analyzer (Carl Zeiss Meditec AG). A standard profile ablation (non-wavefront) was used to correct sphere and refractive cylinder with the aim of achieving emmetropia. An active eye tracker was used to ensure accurate ablation on the center of the pupil. Mean optical zone used was 6.3±0.21 mm.
ReLEx SMILE(Group-C)
The procedure was performed with the VisuMax femtosecond laser (Carl Zeiss Meditec, Jena, Germany), with a pulse repitition rate of 500 KHz, 6.5- to 7-mm optical zone, cap thickness of 120 µm and a 2 mm superior incision, using the standard surgical technique(14). The treatment was centered on the visual axis. Mean optical zone used was 6.5±0.23 mm.
Pre-operatively, the limbus was marked in the 0º to 180º axis with an infrared transmitting dye (Viscot surgical skin marker 1436; Viscot Medical) using either a marker pen or Ganesh bubble marker (Epsilon Surgicals; Figure 1A)in the upright position. The patient was then positioned under the VisuMax FS laser and instructed to look into the green flashing fixation light. Once proper centration was achieved, the eye was docked to the patient interface followed by application of suction. At this point, the extent of cyclotorsion was determined using the reticule in the eyepiece and any cyclotorsion was manually compensated by gently rotating the contact glass to align the horizontal marks on the eye to 0º to 180º axis of the reticule. Once both were aligned, the active laser process was started to create the refractive lenticule and surgery completed in routine manner.
No intraoperative complications were observed in any of the eyes operated in the three study groups.
Post-operative examinations were conducted at day 1, 1 week, 1 month, 6 months and 1 year. At each follow-up visit from 1 month onwards, assessment of uncorrected visual acuity (UCVA), manifest refraction, corrected distant visual acuity (CDVA), topography, contrast sensitivity, abberometry and dry eye evaluation with tear film break up time was performed for all study groups. Endothelial cell counts were repeated for all eyes at 1 year post-operative visit.
In the T-ICL group, in addition to these tests, spectral domain anterior segment OCT(OCT, Optovue(®) iVue, Fremont, USA), was also performed for measurement of the post-operative vault of the T-ICL on all visits.
Statistical and graphical analysis
Statistical analysis was performed using SPSS software (version 17). ANOVA test was carried out for intergroup comparison and paired t test was used for intragroup comparison of means. A P-value of 0.05 or less was considered statistically significant. JRS standard graphs were generated using Datagraph-med 5.20 software.
Results
The groups were appropriately matched with respect to age, SE, contrast sensitivity scores and tear film break up time(p >0.05). However, the mean cylinder was significantly higher in T-ICL group (-1.88D±0.88)compared to FS-LASIK(-1.42D±0.99) and ReLEx SMILE(-1.08D±0.38) (p<0.05).Also, the mean pre-operative CDVA was lower in the T-ICL group(0.032 ± .048LogMAR) compared to other two groups (p<0.05) (Table 1). This was due to the inclusion of two eyes with mild amblyopia (pre-op CDVA 20/30) in this group, while the other two groups had all eyes with pre-op CDVA of 20/20 or better. The higher order aberrations(HOAs) were also significantly high in the T-ICL group, compared to the other two groups, (Table 1).
Safety (Post-operative CDVA/ Preoperative CDVA)– At 1 year, in the FS LASIK group, 4 eyes showed loss of CDVA by one line. T-ICLgroup, had the highest percentage of eyes(60%) with gain in lines of CDVA by one or more lines, compared to FS-LASIK(40%) and ReLEx SMILE group (50%)respectively. Correspondingly, the safety indices were 1.24, 1.15 and 1.11 in group A, C and B respectively(Fig 1).
Efficacy (Post-operative UDVA/ Preoperative CDVA)– At 1 year, 97% eyes in group-C (ReLEx SMILE), 93% eyes in T-ICL and 90 % eyes in FS-LASIK group had UDVA of 20/20 or better(Fig 2). However, the efficacy index was highest in T-ICL group(1.12) compared to ReLEx SMILE(1.06) and FS- LASIK group(1.02).
Mean post op UDVA(LogMAR) was marginally better in ReLEx SMILE group compared to other two groups, however the differences were not significant (p<0.05 at all visits). All three groups showed an improvement in UDVA over time. Similar trends were observed in mean post op CDVA. (Table2)
Refractive outcomes
Comparable results were found with respect to SE correction in T-ICL and ReLEx SMILE group at 1 month post op. Mild undercorrection was observed in both Femto-LASIK and ReLEx SMILE groups, while the SE remained fairly stable in T- ICL group over time. However, differences between the groups were not significant at any follow-up visits(p<0.05)(Table 2). SE predictability was within ± 0.5D in 93% eyes in ReLEx SMILE group compared to 90% eyes in T-ICL and 77% eyes in Femto-LASIK group. All 3 groups showed SE predictability within ± 1D in100% eyes. (Fig 3).
Predictability of astigmatic correction was slightly better with FS-LASIK with 93% eyes being within ± 0.5D at 1 year post-op compared to T-ICL and ReLEx SMILE group (90% eyes within ± 0.5D for both groups). All eyes in all groups had cylinder predictability within ± 1D (Fig 4).All 3 groups showed a slight undercorrection of cylinder at 1 year, which was not statistically significant from 1 month values. (p<0.05)(Fig5)
Contrast sensitivity and aberrations
Mean photopic contrast sensitivity scores were significantly better at 1yr in T-ICL group among the three groups (P<0.05). The scores were in fact better than the preoperative values for spatial frequencies of 6 and 18cpds. On the other hand, the mean scores were significantly lower in LASIK group, however, they were comparable to pre-operative values in ReLEx SMILE group (Fig 6, Table 3).
A significant improvement in higher order aberrations(HOAs) in the T-ICL group was noted at 1 yr showed ,which had actually reduced(p=0.007) , whereas in the FS-LASIK group, the HOAs had significantly increased compared to pre-operative values(p=0.038). In the ReLEx SMILE group, HOAs marginally increased, but the difference was not statistically significant compared to pre-op values (p=0.68). (Table 3)
Endothelial cell count and dry eye assessment
There was no significant change in the endothelial cell count in either of the study groups compared to preoperative values at 1 year,(Table 3).
Dry eye evaluation revealed a significant decrease in TBUT scores in FS- LASIK group at 1 year(p=0.00), while they did not show a significant change in the T-ICL group(p=0.34), when compared to the pre-operative scores. Although, the mean TBUT scores in the SMILE groups showed a transient, non-significant reduction at one month, they improved over time and were comparable to pre-operative values at 1 year(p=0.06 ),(Table 3).
Patient satisfaction and Long term complications
Upto 60% patients in the T-ICL group reported symptoms of dysphotopsia in the immediate post-op period, which gradually reduction in the severity over time. In the SMILE group, most patients complained about slight foggy vision initially, their vision settled by one month. At the end of 1 year, patients in the T-ICL and SMILE group reported excellent satisfaction with their quality of vision, but LASIK patients reported lower satisfaction due to persistent dryness and need for frequent instillation of lubricant eye drops.
In the T-ICL group, 3 eyes required T-ICL exchange due to frequent rotation by > 30 degrees, and excessive high vault in the post-operative period .However, no eye in this group suffered from any sight threatening complication such as cataract, retinal detachment, endophthalmitis or glaucoma. In the FS-LASIK group, 2 eyes had loss of CDVA due to microwrinkles. However, no post-operative complications were observed in the ReLEx SMILE group. (Table2)
Discussion
Outcomes of myopic astigmatism with Toric ICL, Femto- LASIK and ReLEx SMILE have been previously reported(6,12,15). However, to the best of our knowledge, no study has compared these three modalities for astigmatism correction and long term safety, efficacy, visual quality and complications.
In the present study, all the three groups were comparable in terms of mean postoperative UCVA, DCVA, SE and cylinder correction, indicating that all the 3 modalities were effective with no statistically significant differences between the groups for these parameters at 1 year(Table1). However, the UCVA and CDVA was marginally better with ReLEx SMILE and T-ICL respectively in comparison with FS- LASIK. On the other hand, in terms of predictability of astigmatism correction, FS-LASIK scored slightly better compared to both T-ICL and SMILE groups.
These results were consistent with the study by Hasegawa et al, who reported a higher predictability of correction with FS-LASIK compared to toric pIOL in eyes with moderate refractive cylinder(1.50 to 2.75 D), compared to high cylinder >3D, where toric pIOL was found to have better predictability.The authors speculated that this difference was due to difference in the units of refractive cylinder correction since the refractive cylinder power of the Implantable Collamer Lens (T-ICL) comes in 0.50 D increments, while, the corrective refractive cylinders used in LASIK are in 0.25 D steps (16).
Accurate compensation of potential static and dynamic rotation of the eye during treatment by the iris registration and active eye tracker software present in the MEL-90 excimer laser may also have contributed to slightly better outcomes of cylinder correction with FS-LASIK(17). However, outcomes with T-ICL were shown to depend upon factors such as post-operative rotation, sizing issues and accuracy of pre-operative marking (18).In the event of significant(>20degrees) and recurrent rotation in the post-operative period, re – alignment and often an exchange of the T-ICL is warranted (19). This happened with two eyes in this series. However, the T-ICL maintained stable position after exchanging with one size bigger T-ICL, and did not rotate later in both cases.
Previous studies comparing astigmatism correction showed superior results with FS-LASIK compared to ReLEx SMILE(11). Jesper et al, have shown significant under correction of cylinder with SMILE over time(20). This was mainly attributed to non-compensation of errors of cyclotorsion and non-application of nomograms during the procedure(12). However, excellent results for astigmatism treatment were observed with ReLEx SMILE ,which were far better compared to the published literature on SMILE. Since we performed a manual compensation of cyclotorsion and applied a 10% nomogram (based on our experience and post-op results), the accuracy of astigmatism correction was good and comparable to the other two modalites.
Studies using the Optical Quality Analysis System have demonstrated that ICL with central hole appeared to be essentially equivalent in the optical quality variables to conventional ICL implantation, suggesting that the presence of the central artificial hole did not significantly affect the optical quality and the intraocular scattering after surgery(21). The contrast sensitivity following ICL as demonstrated by Igarashi et al, did not significantly change , while there was a definite reduction in contrast after FS- LASIK (22). However the contrast sensitivity was found to be better with SMILE compared to FS-LASIK, as earlier reported(14).Also, the induction of aberrations with ICL, was shown to be minimal and showed significant improvement in aberrations post-operatively(23). Both SMILE and FS-LASIK were reported to induce significant higher order aberrations (24,25), although SMILE was shown to induce less aberrations compared to FS-LASIK(25). Interestingly, in this series, we observed that the HOAs increased after SMILE, but they were not statistically significant at one year. This may be due to use of relatively larger optical zones, minimal transition zone and no loss of energy in periphery(cosine effect) in SMILE compared to LASIK.
Post-operative dry eye was least with T-ICL and was comparable to SMILE, however it was significantly more with FS-LASIK even at the end of one year. Since both the former procedures involve minimal disruption of the corneal nerve fibers, post-operative dryness was practically non-existent compared to FS-LASIK. These observations corroborate with the results of previous studies on FS-LASIK and post-operative dry eye(9,10,14).
Due to the above reasons, the optical quality with T-ICL was better than the other two corneal procedures, leading to highest patient satisfaction in the immediate post-operative period. However, at the end of 1 year, both T-ICL and SMILE had similar post-op patient satisfaction regarding the quality of vision. ReLEx SMILE scored over FS- LASIK in terms of visual quality due to centration of treatment on visual axis, less induction of aberrations and minimal post-operative dry eye(9,14,26).
In conclusion, all the three modalities were found to be effective for myopic astigmatism at the end of one year. However, quality of vision and patient satisfaction with T-ICL and ReLEx SMILE was similar, both being better than FS-LASIK. Slight chances of post-op rotation and exchange exist, which warrant thorough pre-operative evaluation. Selecting the size of ICL based on sulcus to sulcus in comparison to the traditional white to white measurements my prevent exchanges due to sizing issues.
Figures with Legends:
Figure 1. Safety (gain /loss in lines of CDVA) of the three study groups at 1 year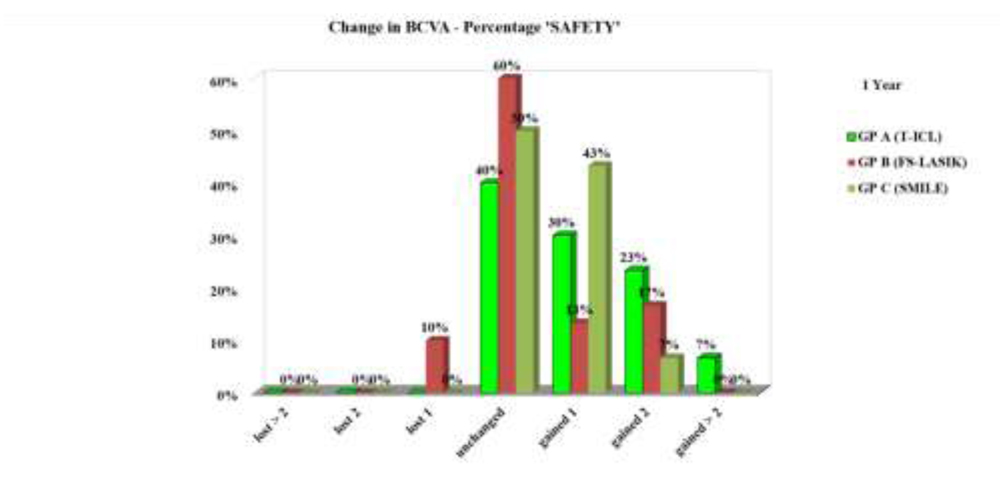
Figure 2.Cumulative UDVA (% of eyes 20/20 or better) in the three study groups at 1 year
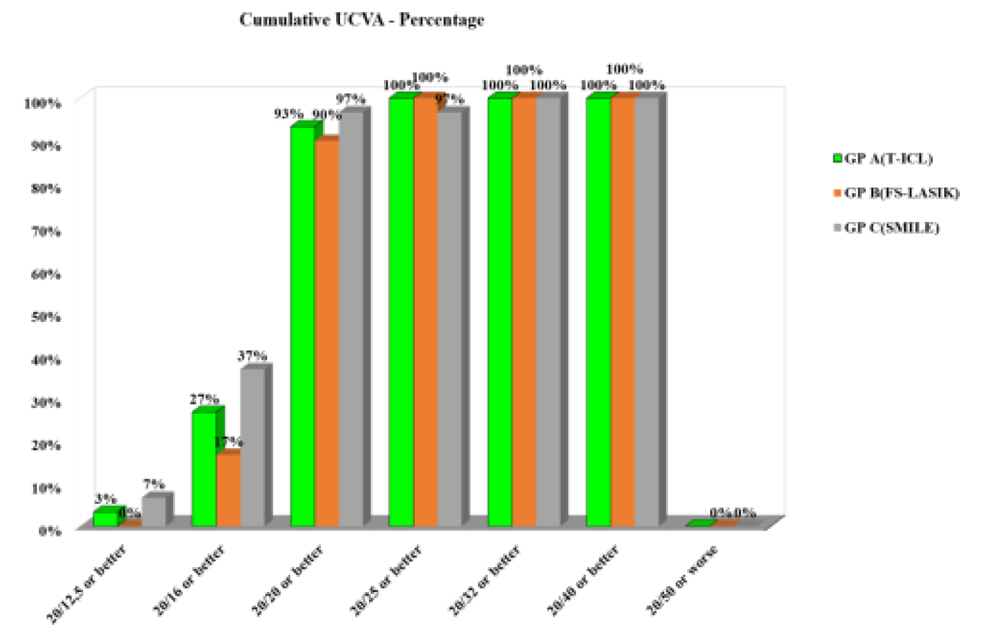 Figure 3. Predictability of Spherical Equivalent (SE) correction with the three treatment modalities at 1 year
Figure 3. Predictability of Spherical Equivalent (SE) correction with the three treatment modalities at 1 year
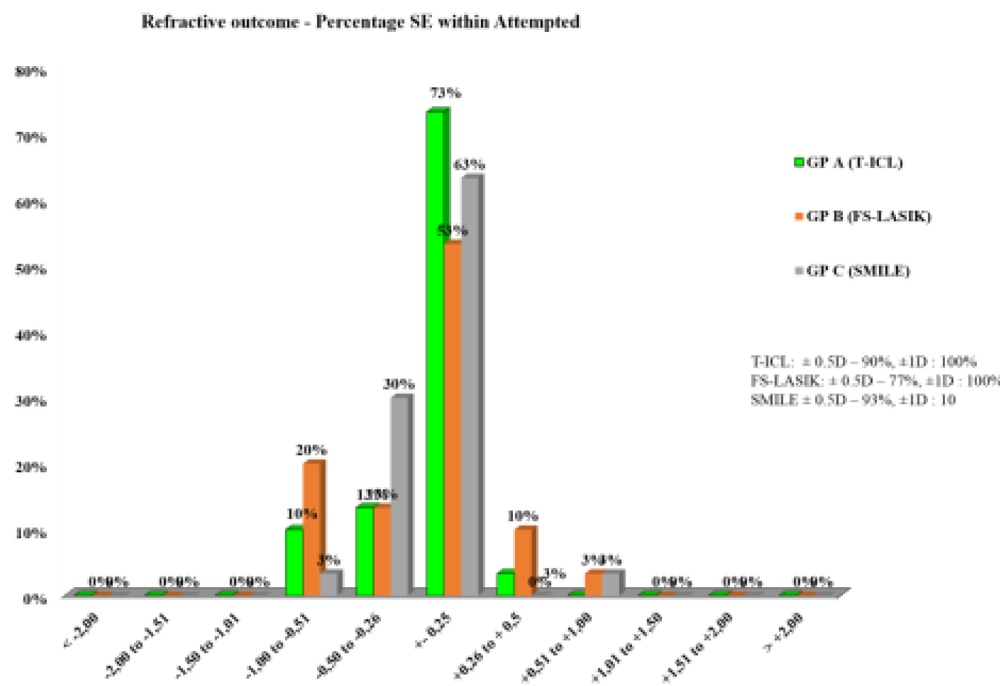 Figure4. Predictability- scatter (attempted v/s achieved) astigmatism correction with the three modalities at 1 year
Figure4. Predictability- scatter (attempted v/s achieved) astigmatism correction with the three modalities at 1 year
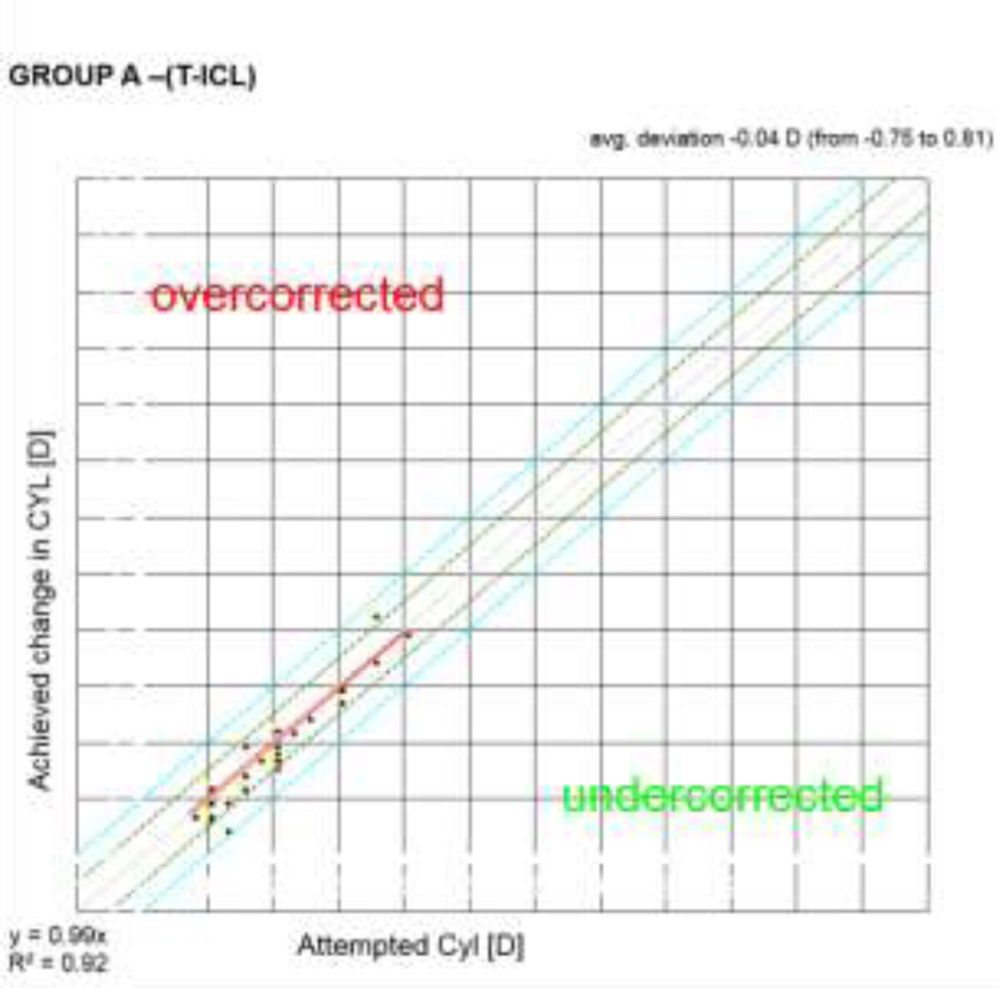
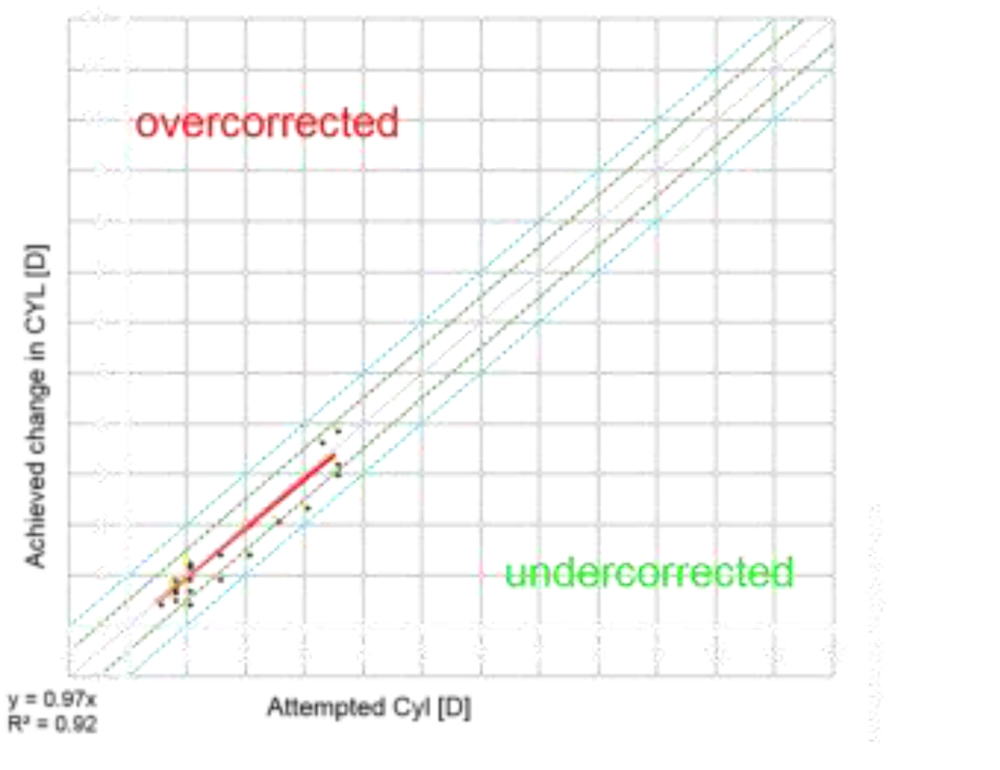
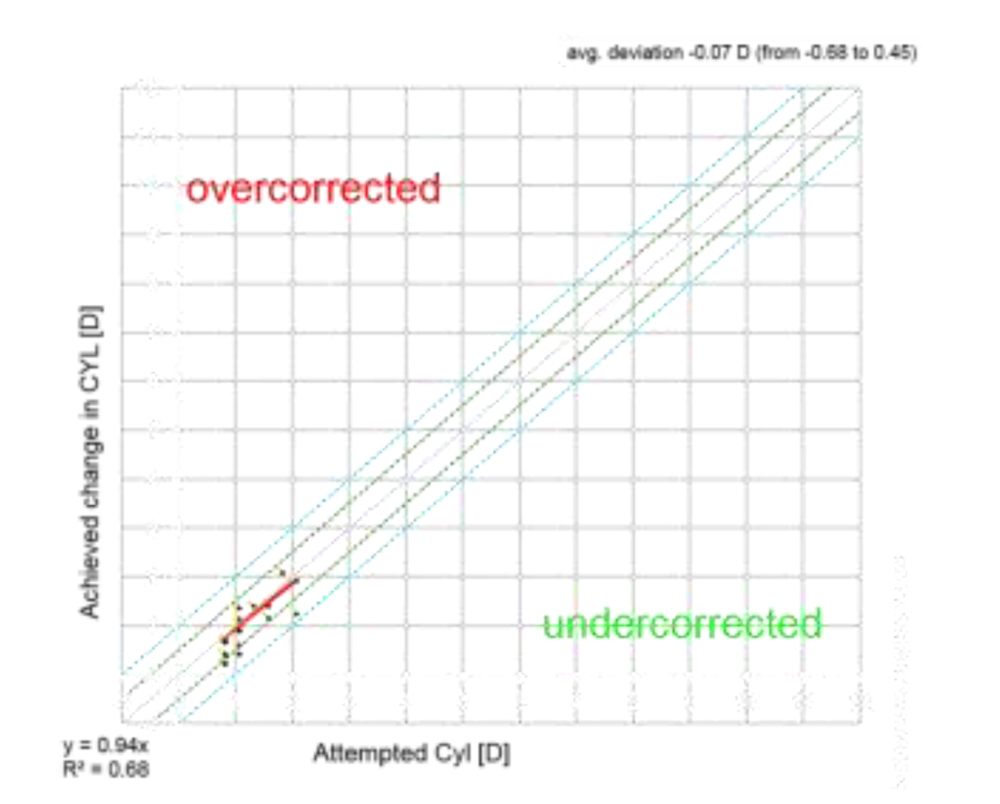 Figure 5. Stability of astigmatism correction with the three treatment modalities
Figure 5. Stability of astigmatism correction with the three treatment modalities 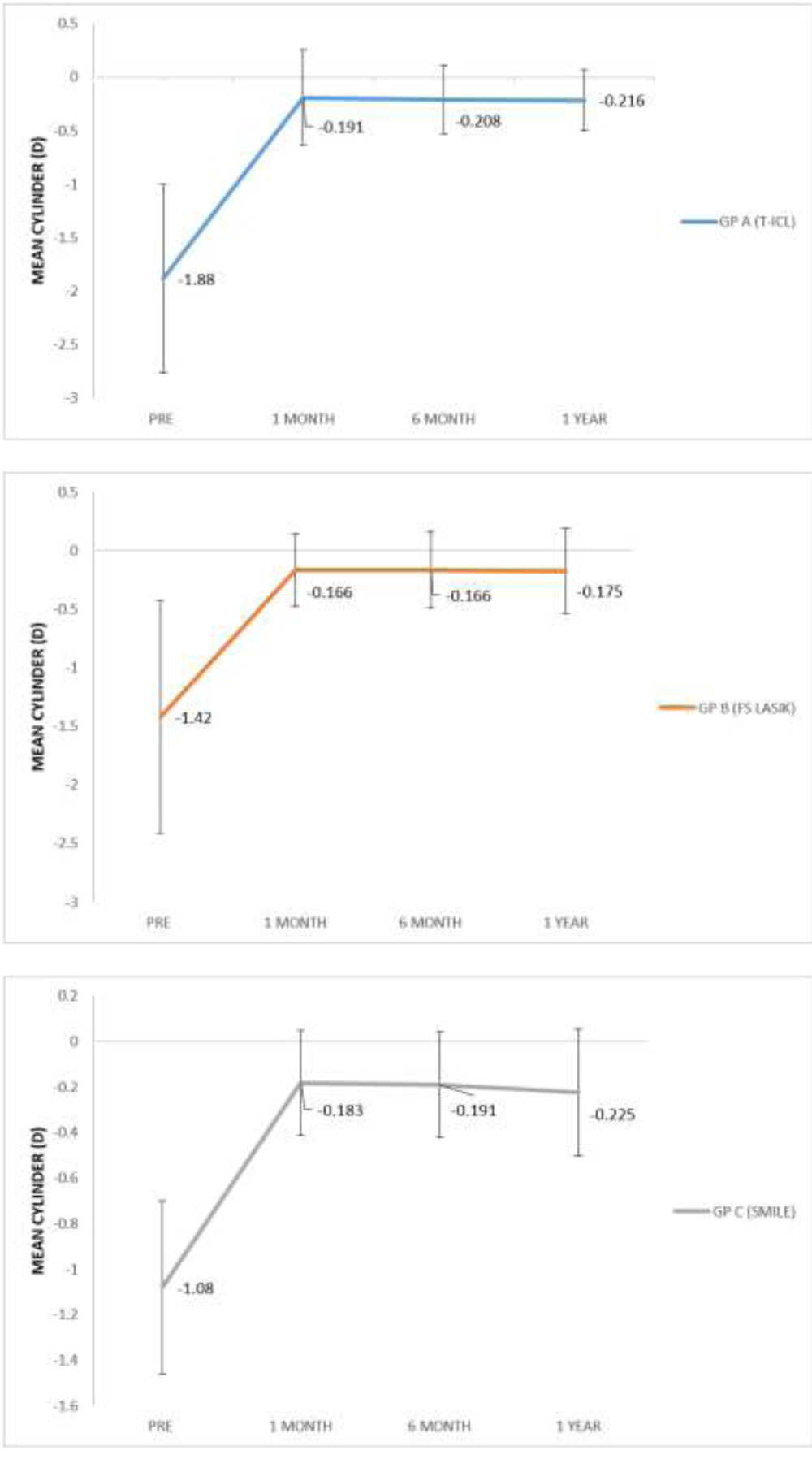 Figure 6.Contrast sensitivity with Functional Acuity Contrast Test (FACT) chart with (A) T-ICL (B) FS-LASIK(C) ReLEx SMILE over time
Figure 6.Contrast sensitivity with Functional Acuity Contrast Test (FACT) chart with (A) T-ICL (B) FS-LASIK(C) ReLEx SMILE over time 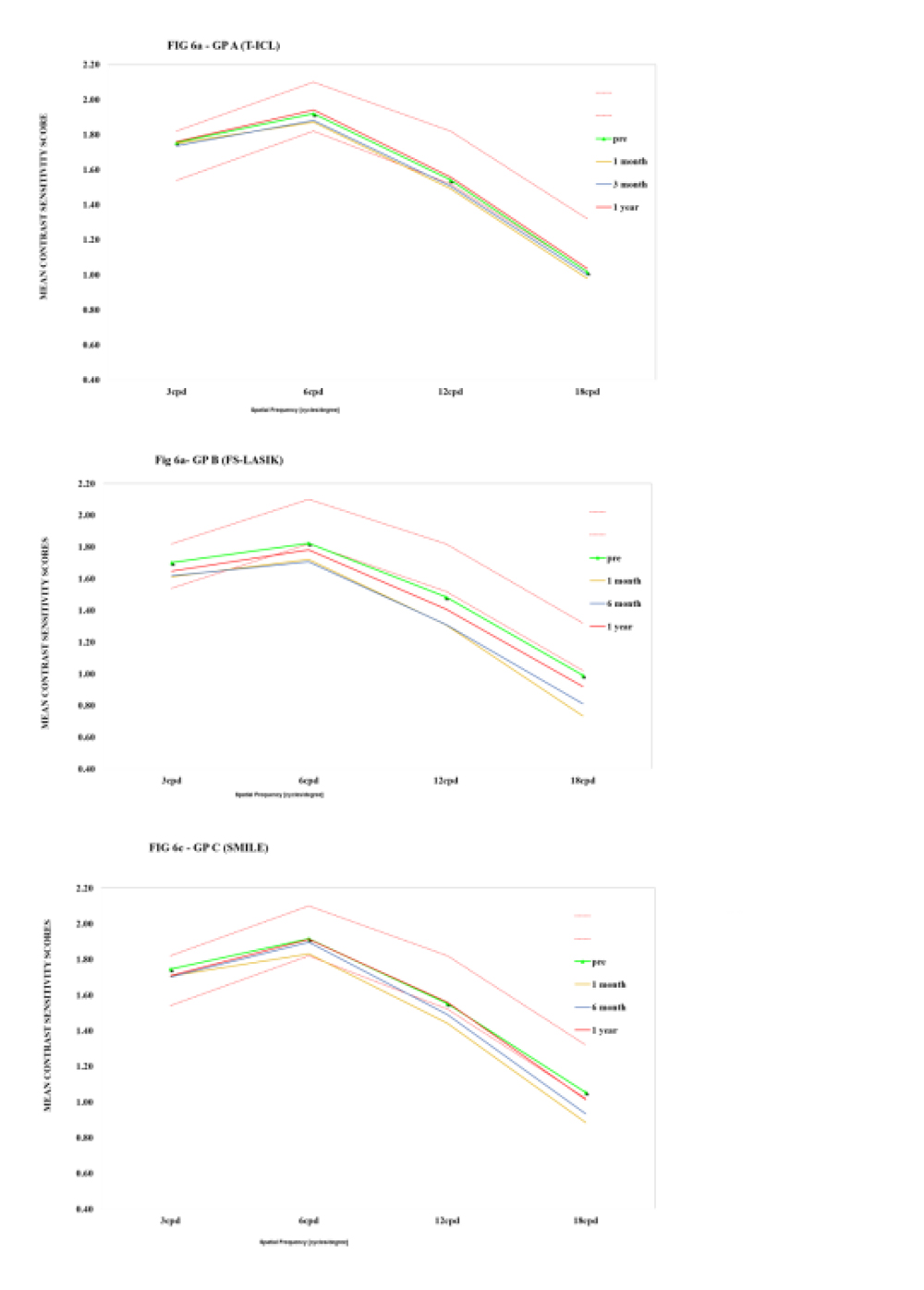 Table 1. Pre-operative baseline characteristics of patients included in the three study groups
Table 1. Pre-operative baseline characteristics of patients included in the three study groups
| Mean±SD | Gp A
(T-ICL) |
Gp B
(FS-LASIK) |
Gp C
(SMILE) |
p-value |
| Age(years) | 26.43±2.4 | 27.63±5.04 | 28.93±5.17 | 0.096 |
| SE(D) | -5.98 D ± 1.15 | -5.43 D ± 1.22 | -4.58 D ± 1.59 | 0.063 |
| Cyl(D) | -1.88 D ± 0.88 | -1.42 D ± 0.99 | -1.08 D ± 0.38 | 0.001* |
| CDVA(LogMAR) | 0.032 ± .048 | 0.007 ±0.016 | -0.01 ±0.043 | 0.000* |
| ECD(cells/mm2) | 3087 ± 245 | 3011 ± 243 | 2951 ± 330 | 0.510 |
| TBUT(sec) | 11.50 ± 1.27 | 11.43 ± 1.47 | 11.26 ± 1.22 | 0.784 |
| HOA(RMS,µ) | 0.260± 0.14 | 0.115 ± 0.05 | 0.120 ± 0.06 | 0.000* |
| FACT(cpd) | ||||
| A(1.5) | 1.79 ± 0.5 | 1.76 ± 0.09 | 1.75 ± 0.06 | 0.157 |
| B(3) | 1.76 ± 0.07 | 1.73 ± 0.13 | 1.75 ±0.08 | 0.392 |
| C(6) | 1.93 ± 0.09 | 1.87 ± 0.19 | 1.91 ± 0.09 | 0.211 |
| D(12) | 1.60 ± 0.10 | 1.58 ± 0.22 | 1.58 ± 0.16 | 0.890 |
| E(18) | 1.08 ± 0.15 | 1.11 ± 0.24 | 1.07 ± 0.27 | 0.814 |
| *p-value using Analysis of Variance (ANOVA)
SE-Sperical equivalent, CDVA-Corrected distance visual acuity, ECD- Endothelial cell density, TBUT- Tear film breakup time, HOA(RMS)-Higher order aberratons (root mean square), FACT- Functional contrast aquity test |
||||
Table 2. Post-operative visual and refractive outcomes with the three surgical modalities at 1 year
| Mean±SD | Gp A(TICL) | Gp B(FS-LASIK) | Gp C(SMILE) | p-value | |
| UDVA(LogMAR) | |||||
| 1month | -0.002 ± 0.055 | 0.0073 ± 0.06 | -0.021 ± 0.062 | 0.115 | |
| 6 months | -0.009 ± 0.043 | -0.002 ± 0.054 | -0.028± 0.055 | 0.230 | |
| 1 year | -0.022 ± 0.021 | -0.011 ± 0.069 | -0.027 ± 0.069 | 0.626 | |
| CDVA(LogMAR)
Pre |
0.032 ± 0.048 |
0.007 ± 0.016 |
-0.008 ± 0.038 |
0.000* |
|
| 1 month | -0.035 ± 0.047 | -0.019 ± 0.065 | -0.04 ± 0.067 | 0.390 | |
| 6 months | -0.055 ± 0.066 | -0.042 ± 0.088 | -0.052 ± 0.076 | 0.780 | |
| 1 year | -0.071 ± 0.079 | -0.06 ± 0.10 | -0.064 ± 0.082 | 0.880 | |
| SE(D)
Pre |
-5.98 D ± 1.15 |
-5.43 D ± 1.22 |
-4.58 D ± 1.59 |
0.063 |
|
| 1month | -0.143 ± 0.22 | -0.159 ± 0.43 | -0.137 ±0.20 | 0.962 | |
| 6 months | -0.159 ± 0.23 | -0.209 ± 0.46 | -0.171 ± 0.20 | 0.823 | |
| 1 year | -0.164 ± 0.20 | -0.225 ± 0.43 | -0.208 ± 0.24 | 0.733 | |
| CYL(D)
Pre |
-1.88 D ± 0.88 |
-1.42 D ± 0.99 |
-1.08 D ± 0.38 |
0.001* |
|
| 1 month | -0.191 ± 0.45 | -0.166 ± 0.31 | -0.183 ± 0.23 | 0.943 | |
| 6 months | -0.208 ± 0.32 | -0.166 ± 0.33 | -0.191 ± 0.23 | 0.863 | |
| 1 year | -0.216 ± 0.28 | -0.175 ± 0.36 | -0.225 ± 0.28 | 0.803 | |
| *p-value using Analysis of Variance (ANOVA)
UDVA-Uncorrected distance visual acuity CDVA-Corrected distance visual acuity, SE(D)-Spherical equivalent(Dioptres), CYL(D)- Cylinder(Dioptres) |
|||||
Table 3. Higher order aberrations, Endothelial cell density, Tear fim break up time and Contrast Sensitivity values at 1 year with the three treatment modalities
| T-ICL | FS-LASIK | SMILE | p- value* | ||
| HOA(RMS,µ) | pre | 0.260± 0.14 | 0.115 ± 0.05 | 0.120 ± 0.06 | 0.000 |
| 1 year | 0.150 ± 0.07 | 0.163 ± 0.14 | 0.126 ± 0.07 | 0.355 | |
| p-value† | 0.007 | 0.038 | 0.688 | ||
| ECD(cells/mm2) | Pre | 3977 ± 334 | 3030 ± 281 | 2845 ± 666 | 0.510 |
| 1 year | 2808 ± 315 | 3025 ± 310 | 2840 ± 070 | 0.683 | |
| p-value† | 0.068 | 0.922 | 0.972 | ||
| TBUT(sec) | Pre | 11.50 ± 1.27 | 11.43 ± 1.47 | 11.26 ± 1.22 | 0.784 |
| 1 year | 11.33 ± 1.2 | 9.4 ± 1.47 | 10.7 ± 1.08 | 0.000 | |
| p-value† | 0.344 | 0.000 | 0.064 | ||
| FACT(cpd) | |||||
| A(1.5) | Pre | 1.79 ± 0.5 | 1.76 ± 0.09 | 1.75 ± 0.06 | 0.157 |
| 1 year | 1.79 ± 0.60 | 1.70 ± 0.09 | 1.75 ± 0.08 | 0.000 | |
| p-value† | 0.573 | 0.007 | 0.988 | ||
| B(3) | Pre | 1.76 ± 0.07 | 1.73 ± 0.13 | 1.75 ±0.08 | 0.392 |
| 1 year | 1.78 ± 0.08 | 1.65 ± 0.12 | 1.72 ± 0.10 | 0.000 | |
| p-value† | 0.161 | 0.000 | 0.249 | ||
| C(6) | Pre | 1.93 ± 0.09 | 1.87 ± 0.19 | 1.91 ± 0.09 | 0.211 |
| 1 year | 1.97 ± 0.06 | 1.78 ± 0.15 | 1.92 ± 0.13 | 0.000 | |
| p-value† | 0.033 | 0.002 | 0.700 | ||
| D(12) | Pre | 1.60 ± 0.10 | 1.58 ± 0.22 | 1.58 ± 0.16 | 0.890 |
| 1 year | 1.61 ± 0.12 | 1.44 ± 0.17 | 1.58 ± 0.16 | 0.000 | |
| p-value† | 0.352 | 0.000 | 0.970 | ||
| E(18) | Pre | 1.08 ± 0.15 | 1.11 ± 0.24 | 1.07 ± 0.27 | 0.814 |
| 1 year | 1.13 ± 0.14 | 0.97 ± 0.19 | 1.10 ± 0.20 | 0.002 | |
| p-value† | 0.004 | 0.000 | 0.579 | ||
| *p-value using Analysis of Variance (ANOVA), †p-value using paired t-test
HOA(RMS)-Higher order aberratons (root mean square), ECD- Endothelial cell density, TBUT- Tear film breakup time, , FACT- Functional contrast acuity test |
|||||
References
- Knorz MC, Wiesinger B, Liermann A, Seiberth V, Liesenhoff H. Laser in situ keratomileusis for moderate and high myopia and myopic astigmatism. Ophthalmology. 1998;105(5):932-40.
- Barsam A, Allan BD. Excimer laser refractive surgery versus phakic intraocular lenses for the correction of moderate to high myopia. Cochrane Database Syst Rev. 2014(6):CD007679.
- Huang D, Schallhorn SC, Sugar A, Farjo AA, Majmudar PA, Trattler WB, et al. Phakic intraocular lens implantation for the correction of myopia: a report by the American Academy of Ophthalmology. Ophthalmology. 2009;116(11):2244-58.
- Sanders D, Vukich JA. Comparison of implantable collamer lens (ICL) and laser-assisted in situ keratomileusis (LASIK) for low myopia. Cornea. 2006;25(10):1139-46.
- Sari ES, Pinero DP, Kubaloglu A, Evcili PS, Koytak A, Kutluturk I, et al. Toric implantable collamer lens for moderate to high myopic astigmatism: 3-year follow-up. Graefes Arch Clin Exp Ophthalmol. 2013;251(5):1413-22.
- Reinstein DZ, Carp GI, Archer TJ, Lewis TA, Gobbe M, Moore J, et al. Long-term Visual and Refractive Outcomes After LASIK for High Myopia and Astigmatism From -8.00 to -14.25 D. J Refract Surg. 2016;32(5):290-7.
- Reinstein DZ, Carp GI, Lewis TA, Archer TJ, Gobbe M. Outcomes for Myopic LASIK With the MEL 90 excimer laser. J Refract Surg. 2015;31(5):316-21.
- Elies D, Alonso T, Puig J, Gris O, Guell JL, Coret A. Visian toric implantable collamer lens for correction of compound myopic astigmatism. J Refract Surg. 2010;26(4):251-8.
- Liu M, Chen Y, Wang D, Zhou Y, Zhang X, He J, et al. Clinical Outcomes After SMILE and Femtosecond Laser-Assisted LASIK for Myopia and Myopic Astigmatism: A Prospective Randomized Comparative Study. Cornea. 2016;35(2):210-6.
- Zhang Y, Shen Q, Jia Y, Zhou D, Zhou J. Clinical Outcomes of SMILE and FS-LASIK Used to Treat Myopia: A Meta-analysis. J Refract Surg. 2016;32(4):256-65.
- Chan TC, Ng AL, Cheng GP, Wang Z, Ye C, Woo VC, et al. Vector analysis of astigmatic correction after small-incision lenticule extraction and femtosecond-assisted LASIK for low to moderate myopic astigmatism. Br J Ophthalmol. 2016;100(4):553-9.
- Zhang J, Wang Y, Wu W, Xu L, Li X, Dou R. Vector analysis of low to moderate astigmatism with small incision lenticule extraction (SMILE): results of a 1-year follow-up. BMC Ophthalmol. 2015;15:8.
- Sheetal Brar., et al. “Incidence & Factors Responsible for Implantable Collamer Lens (ICL) Explantation & Outcomes of Further Management -5 Year Retrospective Study”. EC Ophthalmology 1 (2015): 231-239.
- Ganesh S, Gupta R. Comparison of visual and refractive outcomes following femtosecond laser- assisted lasik with smile in patients with myopia or myopic astigmatism. J Refract Surg. 2014;30(9):590-6.
- Hashem AN, El Danasoury AM, Anwar HM. Axis alignment and rotational stability after implantation of the toric implantable collamer lens for myopic astigmatism. J Refract Surg. 2009;25(10 Suppl):S939-43.
- Hasegawa A, Kojima T, Isogai N, Tamaoki A, Nakamura T, Ichikawa K. Astigmatism correction: Laser in situ keratomileusis versus posterior chamber collagen copolymer toric phakic intraocular lens implantation. J Cataract Refract Surg. 2012;38(4):574-81.
- Reinstein DZ, Gobbe M, Gobbe L, Archer TJ, Carp GI. Optical Zone Centration Accuracy Using Corneal Fixation-based SMILE Compared to Eye Tracker-based Femtosecond Laser-assisted LASIK for Myopia. J Refract Surg. 2015;31(9):586-92.
- Mori T., et al. “Factors affecting rotation of a posterior chamber collagen copolymer toric phakic intraocular lens”. Journal of Cataract Refractive Surgery 4 (2012): 568-573
- Alio JL, Toffaha BT, Pena-Garcia P, Sadaba LM, Barraquer RI. Phakic intraocular lens explantation: causes in 240 cases. J Refract Surg. 2015;31(1):30-5
- Pedersen IB, Ivarsen A, Hjortdal J. Three-Year Results of Small Incision Lenticule Extraction for High Myopia: Refractive Outcomes and Aberrations. J Refract Surg. 2015;31(11):719-24.
- Kamiya K, Shimizu K, Saito A, Igarashi A, Kobashi H. Comparison of optical quality and intraocular scattering after posterior chamber phakic intraocular lens with and without a central hole (Hole ICL and Conventional ICL) implantation using the double-pass instrument. PLoS One. 2013;8(6):e66846.
- Igarashi A, Kamiya K, Shimizu K, Komatsu M. Visual performance after implantable collamer lens implantation and wavefront-guided laser in situ keratomileusis for high myopia. Am J Ophthalmol. 2009;148(1):164-70 e1.
- Perez-Vives C, Dominguez-Vicent A, Ferrer-Blasco T, Pons AM, Montes-Mico R. Optical quality of the Visian Implantable Collamer Lens for different refractive powers. Graefes Arch Clin Exp Ophthalmol. 2013;251(5):1423-9.
- Miao H, Tian M, Xu Y, Chen Y, Zhou X. Visual Outcomes and Optical Quality After Femtosecond Laser Small Incision Lenticule Extraction: An 18-Month Prospective Study. J Refract Surg. 2015;31(11):726-31.
- Gyldenkerne A, Ivarsen A, Hjortdal JO. Comparison of corneal shape changes and aberrations induced By FS-LASIK and SMILE for myopia. J Refract Surg. 2015;31(4):223-9.
- Lazaridis A, Droutsas K, Sekundo W. Topographic analysis of the centration of the treatment zone after SMILE for myopia and comparison to FS-LASIK: subjective versus objective alignment. J Refract Surg. 2014;30(10):680-6.

