Dr. Deboja Mahashweta, M17305, Dr. Asmita Ray
REE PAPER FOR 75th AIOS ANNUAL CONFERENCE 2017
(FULL TEXT)
CHIEF AUTHER: Dr. DebojaMahashweta
AIOS membership I.D M17305
Introduction
Systemic Lupus Erythematosus is a chronic, idiopathic, multisystem, inflammatory, autoimmune disease of protean manifestations and variable behaviour. It is characterized by hyperactivity of the immune system and prominent autoantibody production, wherein, organs and cells undergo damage mediated by tissue binding autoantibodies and immune complexes.(1) The depositions cause inflammation through activation of complement system resulting in disease manifestation.
The term “Lupus”, Latin for wolf, was first described in the medieval ages for some cutaneous lesions by Herbernus of Tours in 916 A.D. Later, in 1951, Cazenave coined the term “Lupus Erythematosus”.(2)
Clinically, it is an unpredictable, remitting and relapsing disease of acute or insidious onset that may involve virtually any organ of the body. The estimated incidence of SLE ranges from 1.8 to 20 per 1, 00,000 per year. In India, the incidence is 3 in 1, 00,000 per year. (3) The disease has a strong female preponderance, approximately, 9:1.However, both sexes, all ages and ethnic groups are susceptible.
The ocular manifestations of systemic lupus erythematosus are varied and the findings are consequences of
- Manifestation of lupus.
- Manifestation of complications of lupus.
- Toxicity from drugs used in the treatment of lupus and its complications.
Although eye itself is regarded as an “immune privileged organ”, SLE can affect every ocular structure, leading, if left untreated, to severe visual loss and even blindness. Ocular manifestations affecting the eye or visual system are seen in 25 – 33 % of the cases.(4)Ocular involvement may precede or follow the onset of systemic illness.
The ocular manifestations of SLE can be an indicator of potentially serious systemic disease activity. They are usually reflection of systemic disease. Thus, thepresence ofocular lupus should alert the clinician to the likely presence of disease activity elsewhere. The eye has been rightly said to be the “barometer for SLE”.(5)
Ocular features can range from lesions of eyelid, keratoconjunctivitis sicca, episcleritis, and scleritis to sight threatening complications like retinal vascular diseases and neuro-ophthalmic complication. Visual loss may occur due to lupus choroidopathy or lupus optic neuropathy. Also, there can be features related to complications of SLE like nephropathy, neuropathy, gastrointestinal complications, etc. Yet another group of ocular disease may result from the drugs used in the treatment of SLE. Steroids can lead to cataract, glaucoma, various ocular infections. Also antimalarials used may lead to certain maculopathies.
Clinical diagnosis of SLE is not confirmatory. Laboratory investigations are frequently necessary to confirm the diagnosis.Diagnosis is usually made by a careful review of the patient’s entire medical history coupled with an analysis of the results obtained in routine laboratory tests and some specialized tests related to immune status.
The study on ocular manifestations of SLE has not yet been done in this part of the country. Therefore, the present work is an attempt to study the spectrumof ocular manifestation of SLE, its incidence, age, sex and distribution and also their relation to the disease activity, complications and treatment modalities. The main purpose of the study is to highlight that proper ocular examination is important for early diagnosis, prompt initiation of therapy and better prognosis of SLE cases.
Methods
A random and prospective study was conducted on 50 patients diagnosed with systemic lupus erythematosus, attending outpatients department and also admitted in wards of various departments of a tertiary hospital for a period of one year.
RESULTS AND OBSERVATION
Incidence:
The incidence of ocular manifestation in the total number of SLE patients examined during the study period is tabulated below:
| CATEGORY | NO. OF PATIENTS |
| The total no. of patients examined during the study period | 50 |
| Total no. of patients with ocular manifestations | 36 |
Age group of study population:
In the present study, the age of the patient ranged from 20 – 60 years. The mean age of presentation is 32.2 years and median age of presentation is 30 years. Majority of the patients were in the age group of 20 – 30 years.
Table showing the age group distribution:
| AGE GROUP (IN YEARS) | NO. OF PATIENTS | PERCENTAGE |
| 20 -30 | 26 | 52 % |
| 31-40 | 16 | 32 % |
| 41-50 | 05 | 10 % |
| 51-60 | 03 | 06 % |
Sex:
In the present study, out of the 50 patients, 47 were females and 3 males. The female is to male ratio is 15.6: 1.
Race:
In the present study, out of the 50 patients, 26 were of mongoloid origin. The 24 patients of non mongoloid origin were from other different race, community and religion.
Blood pressure:
In the present study, out of the 50 patients, 20 patients were hypertensive. Among these 20 patient, some had blood pressure (BP) <140/90, but were on antihypertensive medication and the rest had BP>140/90.
Presence of systemic involvement:
Majority of patients had multisystem involvement
In the present study, all the patients had other system involvement. The number of systems involved ranged from 1 to 5. Majority of the patients had 3 systems involvement (25 in no.). The next biggest group (20 in no.) had 2 systems involved. 3 patients had involvement of 4 systems and 1 each had involvement of 1 and 5 systems.
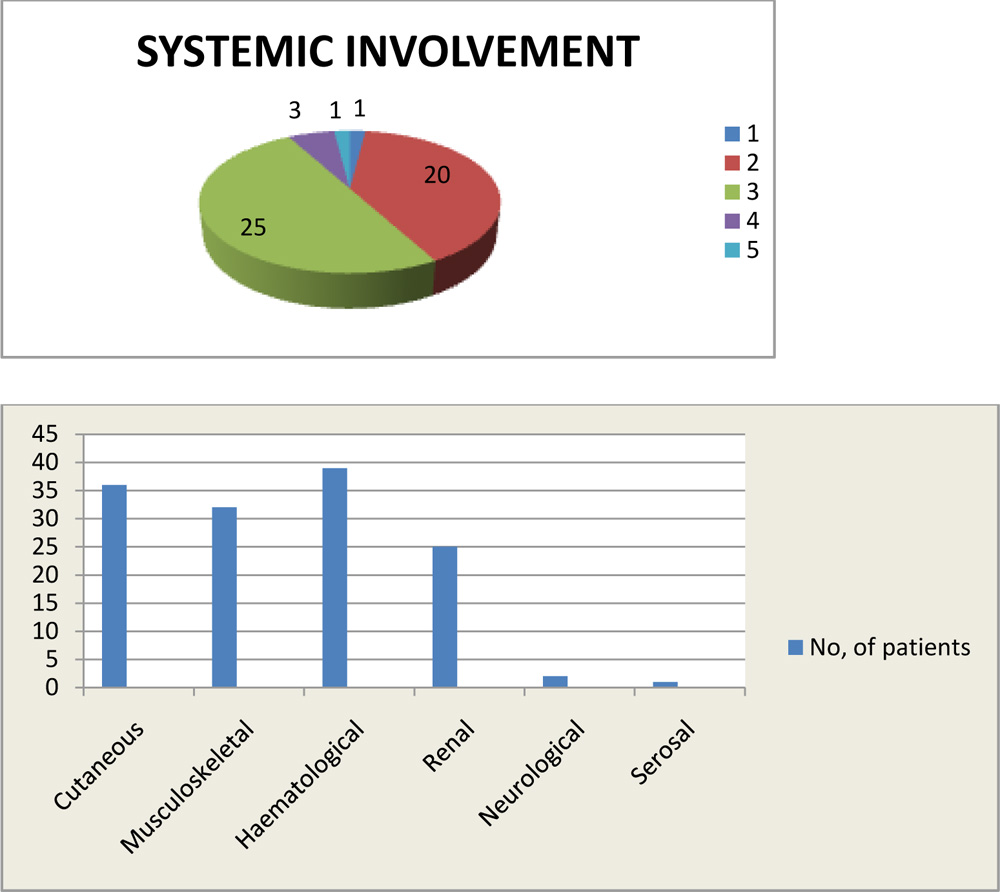
The bar diagram above shows various system involvement in SLE.
Assessment of disease activity:
The disease activity of all the SLE cases was assessed at presentation in relation to the Systemic Lupus Erythematosus Disease Activity Index (SLEDAI). Each of the patients were assigned a score depending on the number of organs involved. Each organ involvement carried a fixed score.
Score < 10: mild to moderate disease.
Score >10: greater disease activity
In the present study SLEDAI score ranged from 3 to 27. Majority of the patients had score < 10 (70 %). Rest of the patients had score > 10 (ranging from 11 to 27). The mean score was 9.42.
Vision:
In the present study, vision was recorded using Snellen’s distant vision chart. The best corrected visual acuity of the 50 patients was divided into three groups, as 6/6 – 6/12, 6/12 – 6/60 and < 6/60. Out of the 50 patients, 3 patients were in the subgroup BCVA 6/18 – 6/60 and 4 were in the subgroup of BCVA < 6/60. Out of the 43 patients in the first group (6/6 – 6/12), 8 patients had BCVA 6/12 in one or both the eyes. All these cases had bilateral posterior subcapsular cataract.
The following table shows the best corrected visual acuity in the study group:
| BEST CORRECTED VISUAL ACUITY | NO. OF PATIENTS | PERCENTAGE |
| 6/6 – 6/12 | 43 | 86 % |
| 6/18 – 6/60 | 3 | 3 % |
| < 6/60 | 4 | 8 % |
The table below shows patients with BCVA less than 6/12:
| Sl no. | Pt. No | Age ( in years) | Sex | BCVA | Cause of decreased vision |
| 1 | 1 | 28 | F | R-FC 1 ft
L-FC 1 ft |
B/L CRVO |
| 2 | 11 | 22 | F | R- 6/18 |
Lupus retinopathy |
| 3 | 21 | 20 | F | R- FC 1 ft
L- FC 1 ft |
B/L optic atrophy |
| 4 | 23 | 20 | F | R- FC 1 ft
L- FC 1 ft |
B/L CRVO |
| 5 | 47 | 59 | F | L- 6/18 |
PSC |
| 6 | 49 | 23 | F | R- FC 2 ft
L- FC 4 ft |
Lupus retinopathy |
| 7 | 50 | 35 | M | L- 6/18 | CSCR
|
Ocular manifestation:
72 % of patients (36 out of 50) had ocular involvement. Anterior segment manifestation was seen in 29 patients and posterior segment lesions in 17 patients. Thus, in the present study group, the anterior segment manifestation (58 %) was more than posterior segment (34 %).Most of the patients had combined anterior and posterior segment involvement
Anterior segment:
The anterior segment manifestations of SLE included those due to the disease and also those due to the complications of the drugs used in the treatment of the disease.
The table below shows the various anterior segment manifestations in the study group:
| ANTERIOR SEGMENT MANIFESTATION | NO. OF PATIENTS | PERCENTAGE | NO. OF EYES |
| Keratoconjunctivitis sicca | 20 | 40 % | 37 |
| Posterior subcapsular cataract | 11 | 22 % | 22 |
| Eyelid lesions | 3 | 6 % | 6 |
| Ulcerative blepheritis | 2 | 4 % | 4 |
| Subconjunctival haemorrhage | 1 | 2 % | 1 |
| Periorbital oedema | 1 | 2 % | 2 |
| Peripheral ulcerative keratitis | 1 | 2% | 1 |
| Episcleritis | 2 | 4% | 2 |
Among the 11 cases of posterior subcapsular cataract (PSC), all had bilateral involvement. This occurred as a complication of corticosteroid used in the treatment of SLE. No correlation was found between the duration of the disease and the development of PSC (p value=0.1198). Also there was no association with the dose of corticosteroids. The corticosteroid treatment included pulse methyl prednisolone (1 gram i.v daily x 3 days) or cycles of pulse dexamethasone (100mg / day x 3 days). The dexamethasone pulse cycles ranged from 14 to 24 in number among the affected patients. The age of the patients ranged from 20 to 42 years, the median value being 35 years. Thus, no such association was seen with the age of the patients.
Also, while comparing the incidence of PSC in SLE with SLE disease activity index, no association was found between PSC and disease activity.(p value = 0.7726)
Posterior segment:
The posterior segment manifestations of SLE included those due to the disease, those due to the complications of the disease and also those due to the drugs used in the disease.
| POSTERIOR SEGMENT MANIFESTATION | NO. OF PATIENTS (PERCENTAGE OF PATIENTS) | NO. OF EYES |
| Lupus retinopathy | 7 (14 %) | 14 |
| Hypertensive retinopathy | 7 (14 %) | 14 |
| CSCR | 2 (4%) | 2 |
| CRVO | 2 (4%) | 4 |
| Optic atrophy | 1 (2%) | 2 |
| Pigmentary maculopathy | 1 (2%) | 2 |
Retinal/choroidal involvement in SLE and disease activity:
The relationship between retinal or choroidal involvement in SLE and the disease activity was measured using SLE disease activity index. In the present study group, out of the 50 patients, 12 had SLEDAI score > 10, which signified greater disease activity. Out of the 12 patients, 9 had either retinopathy or choroidopathy. 38 patients had SLEDAI score <10 (signifying mild to moderate disease). Out of these 38 patients, only 2 had retinochoroidal involvement. The manifestations resulting from the drugs used in the treatment of SLE were excluded.
Table below showing association of retinochoroidal involvement with SLEDAI score:
| RETINAL/CHOROIDAL INVOLVEMENT | SLEDAI < 10 | SLEDAI >10 |
| PRESENT | 2 | 9 |
| ABSENT | 36 | 3 |
The two sided “p value” of the above table is<0.0001 %. It is considered extremely significant.
The row column association is statistically significant.
Calculation details:
Chi sq with Yate correction = 21.942
Degree of freedom = 1
Relative risk = 0.1970
95 % confidence interval – 0.05603 to 0.6994.
Treatment:
In the present study, all the patients were on corticosteroid treatment. Out of the 50 patients, 27 were on steroid only, 20 were on steroid + hydroxychloroquin and 3 were on steroid + immunosuppressant( cyclophosphomide).Out of the 27 patients who were on steroid only, 23 were on prednisolone and 5 on dexamethasone.
The table below shows the different treatment received by the patients in the study group:
| TREATMENT | NO.OF PATIENTS | PERCENTAGE |
| Steroid | 27 | 54 % |
| Steroid + HCQS | 20 | 40 % |
| Steroid + Cyclophosphomide | 3 | 6 % |
Therapy and its complications:
Out of the 50 patients in the study group, 12 patients had ocular manifestations arising from the drugs used in the treatment of SLE.
Out of the 50 patients on steroid, 11 had developed bilateralposterior subcapsular cataract and 2 patients had unilateralcentral serous chorioretinopathy.
Out of the 20 patients on steroid +HCQS, 1 had bilateralpigmentary maculopathy.
However, none of the complications had any relation with the duration of therapy.
The table below shows ocular manifestations due to drugs used in the treatment of SLE:
| MANIFESTATION | NO. OF PATIENTS | PERCENTAGE | NO. OF EYES |
| Posterior subcapsular cataract | 11 | 22 % | 22 |
| Pigmentary maculopathy | 1 | 2 % | 2 |
| CSCR | 2 | 4 % | 2 |
DISCUSSION
In the present study, 50 diagnosed patients of systemic lupus erythematosus attending the various departments of a tertiary hospital for a period of 1 year. The incidence and the spectrum of ocular manifestations of SLE among the OPD as well as the indoor cases were studied.
Incidence:
Ushiyama et al., 2000(6) reported in their study that the ocular complications are seen in around 25 – 33 % of cases of SLE.
In present study involving 50 patients, 36 patients had ocular involvement, which amounts to 72 %.The higher percentage of incidence may have been due to the following reasons:
- The study group was small.
- The ocular manifestations also included those due to the complication of the disease and those resulting from the drugs used in the treatment of the disease.
- Most of the patients included in the study were admitted patients where chance of increased disease activity was more.
Age:
In the present study, the age of fifty patients were divided into four groups, as 20- 30 years, 31-40 years, 41-50 years and 51 – 60 years. It was observed that maximum number of patients (52 %) were in the age group of 20 – 30 years. The mean age of presentation of was 32.2 years and median age of presentation was 30 years.
Yap et al., 1998 (7) in their study found the mean age of the study population as 32.9 years.
M P K Soo et al., 2000(8) in their study found the mean age of the study population as 34+11 years.
Kumar et al., 2002 (3) reported the mean age of presentation in SLE to be 36.7 years.
Thus, the present study was comparable with the earlier studies.
Sex:
Out of the 50 patients in the present study, 47 patients were female and 3 patients were male. Thus, the female : male ratio was 15.6 : 1.
Yap et al., 1998 (7) in their study found the female: male ratio to be 16.5 :1
Kumar et al., 2002 (3) in their study reported the female : male ratio as 11 : 1.
Bertoli et al., 2010 in a large multicentric study involving 588 patients reported the female : male ratio as 8.9 :1.
Thus, we find that the sex ratio of the present study was comparable to the earlier studies.
Race:
In the present study, it was observed that 52% of the patients (25 in no.) were of mongoloid origin. Rest of the patients (24 in no.) were from various other communities taken together.
A study in Hawaii showed prevalence in those of Chinese origin being 24.1 in 1,00,000, whereas that in whites was 5.8 in 100000.(9)
Also, Yap et al, 1998 (7)in his study found that out of the 70 patients included in their study,there were 55 Chinese (78%), 10 Malays (14%), 4 Indians (6%) and one Eurasian (1%).
MP Soo et al, 2000 (8) in their study found that out of the 52 patients included in their study,39 (75%) Chinese, 10 (19.2%)Malays and 3 (1.9%) Indians.
Thus, it can be concluded that SLE is more prevalent in mongoloid population, as Chinese are also considered to be mongoloid descendants.
Systemic involvement:
Systemic involvement was seen in all the cases in the present study, ranging from 1 to 5 systems. Majority of the patients (58 %) had three or more system involvement. 2 % and 40 % patient had one and two system involvement respectively.
Soo et al., 2000(8) in their study reported one and two system involvement in 4 % and 35 % patients respectively, while 60 % patients had three or more system involvement. Thus, the present study was comparable with this study.
Among the various systemic involvements, majority of the patients had cutaneous (72 %) and haematological (78 %) involvement. This was followed by musculoskeletal involvement (64 %) patients.
Gladmannet al., 1997 (10) reported the incidence of cutaneous involvement at the time of diagnosis to be 53 % and at anytime during the disease 78 %. Also he reported the incidence of haematological and musculoskeletal involvement anytime in the disease to be 85 %.
Soo et al., 2000 (8) in their study found cutaneous and musculoskeletal involvement to be most common, 90% and 86 % respectively. In their study, haematological involvement was seen in lesser number of cases.
The variation in the systemic involvement in the present study in comparison to the earlier studies may be attributed to the shorter duration of disease (mean duration of disease 1.75 years). These patients, if followed over a period of time, may present with other systemic involvements.
Assessment of disease activity:
In the present study, the disease activity was assessed using the SLE disease activity index. Out of the 50 patients, 15 had SLEDAI score more than 15, which indicated increased disease activity. Rest of the patients had score less than or equal to 10, which indicated mild to moderate disease. The mean SLEDAI score was 9.42.
Narayanan et al, 2010 (7) in their study of 50 SLE patients, reported the mean score of 10.88. Thus, the present study is comparable to the earlier study.
Vision:
In the present study, out of the 50 patients, 43 patients had best corrected visual acuity (BCVA) 6/12 or better in either of the eyes. 3 patients had BCVA between 6/18 and6/60 and 4 patients had vision worse than 6/60. Thus, 14 % patients had BCVA lessthan 6/12. Among the 43 patients in the first group, 8 patients (14 eyes) had BCVA 6/12. All these patients had cataract. In the second group, out of the 3 patients (3 eyes), one each had cataract, CSCR and lupus retinopathy. In the third group, out of 4 patients (8 eyes), 2 had CRVO, 1 had B/L optic atrophy and one had lupus retinopathy.
Yap et al., 1998(7) in his study reported BCVA less than 6/12 in 7 patients out of the 70 patients in the study group (10 %). Out of the 7 patients, two each had cataract, optic neuropathy and retinal vaso occlusive disease.
Thus, the present study is comparable with the earlier study.
Anterior segment:
The commonest anterior segment manifestation was keratoconjunctivitis sicca. It was present in 40% of the patients (20 in no.), as recorded by Schirmer’s strip with a recording of less than 10 mm.
Steinberg et al., 1971(12)reported the incidence of keratoconjunctivitis sicca (KCS) in SLE to be around 25 %.
Yap et al.,1998 (7) in their study reported the incidence of KCS as 31 %.
Jensen et al.,1999 (13) reported the incidence of KCS in SLE to be upto 35 %.
M P K Soo et al., 2000 (8) in their study found KCS in 20 % of the cases. In this study, the author reported that although one third of the patient in the study group had dry eye, most of them were asymptomatic. Also, they didn’t find any relation between KCS and age of the patient, duration of illness or severity of SLE.
In the present study also, no relation has been seen between occurrence of dry eye and age (p value=0.0632), duration (p value=0.0978), or severity of SLE. Thus, the present study is comparable to the earlier studies in regard of keratoconjunctivitis sicca in SLE.
The next common anterior segment finding in the present study is posteriorsubcapsular cataract (PSC). Out of the 50 cases, 11 had bilateral posterior subcapsular cataract (22 %). This was considered to be a complication of the use of corticosteroids in the treatment of SLE.
Yap et al.,1998 (7) reported the incidence of steroid induced PSC in SLE to be 20 %
M P K Soo et al.,2000 (8) in their study reported the incidence of PSC to be 14 %. Also they reported that there was no relation between occurrence of cataract in SLE with age of the patient, duration of illness and activity of the disease. All the cases were bilateral.
In the present study, no relationship was seen between duration of illness and PSC (p value=0.1198).No association was seen between disease activity (SLEDAI) and PSC also (p value= 0.7726). Thus, the present study is comparable to the earlier studies.
Hardieet al.,1992 (14) reported that although direct link between steroid and PSC is generally accepted, however dose dependant nature of this cataract is still controversial.
Black and colleague., 1960 (15)had reported that patients with steroid dose of less than 10mg/day of prednisolone or equivalent or duration of less than 1 year were less likely to develop lenticular changes.
In the present study,the corticosteroid treatment included pulse methyl prednisolone (1 gram i.v daily x 3 days) or cycles of pulse dexamethasone (100mg / day x 3 days). The dexamethasone pulse cycles ranged from 14 to 24 in number among the affected patients, each cycle after every 28 days. Thus, all the patients were on corticosteroid dose of more than 10 mg/day equivalent of prednisolone.
In the present study, scaly eyelid lesions and ulcerative blepheritis is seen in 6 % and 4 % of the patients respectively.
Huey et al., 1983 (16) in their study reported eyelid involvementin SLE patients in 6 % of the cases, which is similar to the present study.
In the present study, subconjunctival haemorrhage, peripheral ulcerative keratitis (PUK) is seen in one each of the patients.
Shearn and Pirofsky.,1952(17) in their study of ocular involvement of SLE in 32 patients had found subconjunctival haemorrhage in 2(6.2 %) patients
Foster et al.,1999 (18)reported peripheral ulcerative keratitis as a rare manifestation and an ominous sign of active vasculitis with higher disease activity.
In the present study, the patient who had presented with PUK had SLEDAI score of 27, which is also the highest score in the present study. This patient also had signs of increased disease activity in the posterior segment.
Out of the 50 patients in the present study, 2 (4 %) presented with episcleritis.
Frithet al., 1990 (19)had reported the occurrence of repeated episodes of episcleritis in patients with SLE and the incidence may rise upto 28%.
Posterior segment:
In the present study, 17 patients (28%) presented with posterior segment involvement. The commonest finding was lupus retinopathy (benign form), seen in 14 % of the patients. All the patients had bilateral involvement.
Stafford Brady et al., 1988 (20) reported the incidence of more benign form of SLE to be 3.3 to 28.1 % with the incidence rising with the severity of the systemic disease in the sampled population.
M PK Soo et al., 2000 (8) reported that the incidence of early retinal disease in SLE is up to 29 %, although the frequency of the findings varies according to the population being studied and the systemic disease activity.
Ushiyama et al., 2000 (6) reported in their study involving 69 patients, the incidence of lupus retinopathy to be 10 %.
Sivarajet al., 2007 (21) reported that retinal affection occurs in 7 – 26 % of the patients which is invariably bilateral.
Vasudev et al.,2010 (22) reported that lupus retinopathy may be asymptomatic in its milder forms or may result in permanent visual loss.
The classical findings like cotton wool spot, superficial haemorrhage, hard exudates were present in all the seven patients bilaterally. In this group of patients, vision loss was not severe. The vision recorded in 6 out of 7 patients ranged from 6/6 to 6/36. In one patient, due to other comorbidities, vision could not be ascertained properly.
Jabs et al., 1986 (23) reported that in contrary to milder form,severe occlusive retinopathy or vasculitis is a rare form of retinopathy in SLE. Unlike the milder, more benign forms of retinopathy, visual loss often ensues.
Gold et al., 1977 (17) reported that clinical association exists between severe retinal vascular occlusion in SLE and CNS lupus.
Foster et al., 2000 (18) in their study reported that patient with SLE retinopathy are at a higher risk of developing CNS lupus (71%) when compared with those without (13 %).
In the present study, 2 patients presented with bilateral central retinal vein occlusion. The vision of both the patients was finger counting at one foot in both the eyes. Also, in both the cases, central nervous system involvement was present. Thus, the present study also supports the association between severe vaso occlusive retinopathy in SLE and CNS lupus.
Out of the 50 patients in the present study, 2 patients (4 %) presented with central serous chrioreretinopathy.
Khng et al., 2000(24) reported that central serous chorioretinopathy (CSCR) has also been associated as a complicating event in SLE, although it is uncommon. Further, he noted that it is difficult to be sure of the exact cause for CSCR in the presence of SLE, because this group of patients is predisposed to choroidal vascular inflammation, tend to have nephritis and associated hypertension, and often are oncorticosteroid therapy.
In the present study, out of the two patients with CSCR, both were on corticosteroid therapy. However, none of them were hypertensive or had nephropathy. Thus, though hypertension and nephropathy can be ruled out as the cause for CSCR, it is difficult to conclude whether the disease per se or the corticosteroid therapy is responsible.
Klinkhoff et al.,1986 pointed out that retinal manifestation parallel disease activity elsewhere.
Sivaraj et al.,2007(21) reported that SLE retinopathy and scleritis are the hallmarks of systemic disease activity and require systemic immunosuppression.
Stafford Brady et al.,1988(20) noted that retinopathy in SLE is a marker of poor prognosis for survival, that is SLE patients with retinopathy have overall worse prognosis and decreased survival, compared to SLE patients without retinopathy.
In the present study, out of the 11 patients with retinal involvement (excluding cases of hypertensive retinopathy and one case of optic atrophy), 9 patients had SLEDAI score > 10. Only 2 patients had score< 10. Out of 39 cases without retinal involvement, only 3 patients had score >10. Rest of the patients had score < 10.This has been considered extremely significant. ( p value = 0.0001)
In the present study, 1 patient (2%) presented with bilateral optic atrophy with gross diminution of vision.
Yap et al., 1998(6) reported the incidence of neuro-ophthalmological manifestations as ranging from 3% to 30% in various studies.The clinical presentation is highly variable but generally the visual outcome is poor.
Jab et al.,1986(23) reported that the most common manifestation is that of optic nerve damage. He further noted thatthe optic nerve damage is believed to be secondary to an occlusive vasculitis of the small arterioles of the nerve, which leads to demyelination and/or axonal necrosis.
In the present study, along with pale optic disc there was marked attenuation of the vessels. This may suggest earlier retinopathy leading to marked secondary gliosis as the possible cause for optic atrophy.
Features of hypertensive retinopathywere seen in 7 (14%) patients in the present study. In the present study 20 patients were hypertensive. Out of these 20 patients, 7 had hypertensive retinopathy. The association between presence of high blood pressure and disease activity was not found to be significant (p value = 0.3447). Also, association between hypertensive retinopathy and disease activity was not significant. (p value = 1.000)
No comparative data was available in this regard.
Also, one patient in the present study had pigmentary maculopathy in both the eyes. This was considered to be a complication of hydroxychloroquin (HCQS) therapy. Out of the 20 patients on HCQS, 1 patient had developed pigmentary maculopathy.
- Peponis et al., 2006(25) reported pigmentary stippling and granular appearance as early complication of HCQS therapy.
Sivaraj et al., 2007(20) noted loss of foveal reflex and fine granular appearance as initial subtle changes which are often asymptomatic in HCQS therapy.
Mavrikakiset al., 2003 in their study of 400 patients receiving long term HCQS upto 6.5mg/kg/day, found only 2 patients affected and that after a period of 6 years.
In the present study, the patients with pigmentary changes over macula was on dose of 200 mg twice daily , with breaks in between therapy, for around 5 years.
In the present study, follow up was difficult. Most of the patients didn’t turn up for check up. In majority of the cases, either the patients were asymptomatic or they had other comorbidities for which follow up could not be done. For this reason, long term monitoring of the ocular involvement in relation to systemic status was not possible.
CONCLUSION:
According to the results obtained from the present study, it can be concluded that SLE and its ocular manifestations are not uncommon. Also, some of the ocular manifestations are associated with significant morbidity.
In our present study, ocular involvement was found in 72 % of the total SLE cases. Both anterior and posterior segment were involved with anterior being more common than posterior.
Thus, we conclude that as SLE is a multisystem disease with varied manifestations, diagnosis depends upon careful evaluation of all the systems including eye. Therefore,
ophthalmologist should include SLE in the differentialdiagnosis of many retinal vascular and neuro-ophthalmicdisorders. In addition, all patients with ocular lupus should be carefully evaluated forsystemic involvement to detect potentially treatable andpreventable complications of the disease. The ophthalmologist may play an important rolein the care of patients with SLE, since ocular inflammatorylesions may precede potentially serious extraoculardisease and may guide towards the diagnosis.
Bibliography
- Diseases of the immune system, Harrison’s Principles of Internal Medicine, 17th edition.
- Smith CD.,Cyr M. : The history of lupus erythematosus. From Hippocrates to Osler.Rheum Dis Clin North Am. 1988 Apr;14(1):1-14.
- Kumar A. Indian guidelines on the management of SLE; J Indian RheumatolAssoc 2002; 10: 80-96
- Ushiyama o, Ushiyama K, Kaorada S, Suzuki N, tada Y, Ohta et al. Retinal diseases in patients with systemic lupus erythematosus. Ann Rheum Dis 2000; 59: 705-8
- Mary Terlaak Smith. The Eye as a Barometer for SLE. Lupus International 2011.
- Ushiyama O, Ushiyama K, Koarada S, Tada Y, Suzuki N, Ohta A, Oono S,Nagasawa K. (2000) Retinal disease in patients with systemic lupus
- erythematosus. Ann Rheum Dis 59: 705-708.
- Yap EY, Au Eong KG, Fong KY, Howe HS, Boey ML, Cheah WM, FengPH. (1998) Ophthalmic manifestations in Asian patients with systemiclupus erythematosus. Singapore Med J 39: 557-559.
- Soo MP, Chow SK, Tan CT, NadiorN,Yeap SS, Hoh HB. The spectrum of ocular involvement in patients with systemic lupus erythematosus without ocular symptoms. Lupus 2000; 9: 511–514.
- Serdula MK, Rhoads GG. The frequency of systemic lupus erythematosus in different groups in Hawaii. Arthritis Rheum 1979; 22: 328 – 33.
- Gladman D. Systemic lupus erythematosus: clinical features. In:KlippelJ,WeyandC,WortmannR,editors. Primer on the rheumaticdiseases. 11th ed. Atlanta: ArthritisFoundation, 1997;267–272.
- Col K Narayanan, Col V Marwaha, Col K Shanmuganandan, GpCapt S Shankar. Correlation between Systemic Lupus Erythematosus Disease Activity Index, C3, C4 and Anti-dsDNA Antibodies. 2010. MJAFI. Vol 66, No. 2.
- Steinberg AD, Talal N. The coexistence of Sjögren’s syndrome and systemic lupus erythematosus. Ann Intern Med 1971; 74: 55–61.
- Jensen JL, Bergem HO, Gilboe IM, Husby G, Axell T. Oral and ocular sicca symptoms and findings are prevalent in systemic lupus erythematosus. J Oral Pathol Med 1999; 28: 317–322.
- HardieI, Matsunami C, Hilton A, Dyer J, Rumbach 0. Ocular complications in renal transplant recipients. Trans Aor1992;24: 177.
- Black RL, Oglesby RB, von Sallmann L, Bunim Posterior subcapsular cataracts induced by corticosteroids in patients withrheumatoid arthritis. JAM 1960; 174 150-155.
- Huey C, Jakobiec FA, Iwamoto T et al. Discoid lupus erythematosus of the eyelids. Ophthalmology 1983; 90: 1389 ± 1398.
- Gold DH, Morris DA, Henkind P. Ocular findings in systemic lupus erythematosus. Br J Ophthalmol1972; 56: 800–804.
- Foster CS. Systemic lupus erythematosus. In: Albert DM, Jacobeic FA, azar DT, Gragoudas ES, Power SM, Robinson NL editors. Priciples and practice of ophthalmology. 2nd Pennsylvania. WB Saunders company; 2000. P 4563-9
- Frith P, Burge SM, Millard PR,Wojnarowska F. External ocular findings in lupus erythematosus: a clinical and immunopathological study Br J Ophthalmol1990; 74: 163–167.
- Stafford-Brady FJ, Urowitz MB, Gladman DD, Easterbrook M. Lupus retinopathy. Patterns, associations, and prognosis. Arthritis Rheum 1988;31: 1105–1110.
- Sivaraj RR, Durrani OM, Denniston AK, Murray P, Gordon C. Ocular manifestation of systemic lupus erythematosus. Rheumatology 2007; 5: 1-6.
- VasudevAnandRao, DattaGulnarPandiar, NirupamaKasturi, V Muthkrishnanan, DM Thappa. A case to illustrate the role of ophthalmologist in systemic lupus erythematosus. Ind J Dermatol 2010; 55(3): 268-70
- Jabs DA, Hanneken AM, Schachat AP, Fine SL. Choroidopathy in systemic lupus erythematosus. Arch Ophthalmol1988; 106: 230–234.
- Khng CG, Yap EY, Au-Eong KG, Lim TH, Leong KH. Central serous retinopathy complicating systemic lupus erythematosus: a case series.Clin Experiment Ophthalmol2000; 28: 309–313.
- V Peponis, V C Kyttaris, C Tyradellis, I Vergados and N M Sitaras. Ocular manifestations of systemic lupus erythematosus: a clinical review. Lupus 2006; 15:3

