Dr. Riddhi Arya, A13570, Dr. Manish Nagpal, Dr. Navneet Mehrotra
Introduction
Diabetic Macular Edema (DME) is the leading cause for significant vision loss in the patients with diabetic retinopathy (DR)1. The inflammation has been noted as the leading events in disease development. It is observed that the increased permeability of macular capillaries in the course of hypoxia leads to increased levels of vascular endothelial growth factor (VEGF) and release of inflammatory factors, including chemokines, cytokines such as interleukin (IL)-6 and IL-8, and prostaglandins.2,3
The administration of intravitreal corticosteroids reduces capillary permeability and the formation of secondary macular edema due to various etiologies.4,5Corticosteroids also restrict the migration of leukocytes and inhibit the formation of VEGF factor, prostaglandins, and other proinflammatory cytokines.6,7
Dexamethasone is one of the most potent anti-inflammatory steroid. Its effect is six times stronger than intravitreal triamcinolone acetonide,8and 30 times more than cortisol.1
In this study we evaluated the efficacy of intravitrealozurdex implant in naïve and refractory DME.
Materials and Methods
Patient Selection
This is a single center, prospective, comparative, interventionalstudy carried out at a tertiary eye care center. Patients were recruited between Jan 2015 to Jan 2016 after obtaining approval from the institutional review board/ethics committee and informed consent from all patients. It involves 35 eyes of type 2 diabetes mellitus patients with diabetic retinopathy and DME. The patients were divided into two groups. Group 1 had 19 eyes of treatment naïve patients and Group 2 had 16 eyes of refractory DME.
Inclusion criteria.
Criteria for inclusion were as follows: (1) age >18 years old, (2) DME in type 2 DM (3) central foveal thickness (CFT) >270 micron (4) the patients previously treated with Anti –VEGF were included in group 2, the last injection was received 3 months prior to the recruitment
Exclusion criteria.
Exclusion criteria were as follows: (1) structural damage (including atrophy of the retinal pigment epithelium, subretinal fibrosis, laser scars, epiretinal membrane involving fovea, or organized hard exudative plaques) within a 0.5 disc diameter of the center of the macula in the studied eye precluding improvement in visual acuity following the resolution of macular edema; (2) ocular surgery in the study eye in the last six months; (3) a history of ocular inflammation or (4) glaucoma; and (5) ocular hypertension in response to steroid treatment.
DME diagnostic criteria and evaluations.
Baseline evaluation of vision was carried out before therapy. Fundus Fluorescein Angiography (FFA) was performed to evaluate the status of retinopathy, while BCVA was assessed through Snellen’s visual acuity chart converted to LogMAR, slit lamp biomicroscopy, ocular tonometry (using a Goldmannapplanation tonometer), fundus biomicroscopy, optical coherence tomography (Spectralis HRA-OCT by Heidelberg Engineering) and color fundus photography. Central foveal thickness (CFT) and enhanced depth imaging (EDI) for choroidal thickness was measured by spectral-domain OCT.
Refractory Diabetic Macular Edema: it was defined as macular edema persisted for more than six months despite treatment with anti VEGF agents. The last injection was given at least three months before starting treatment with ozurdex implant.
BCVA, CFT and EDI examinations were carried out at baseline, one month, three monthsand six months post injection.
Intraoperative procedure of intravitrealOzurdex implant. All implants were performed under sterile conditions, after preparation of the conjunctiva using 5% povidone–iodine solution, topical anesthetic with lignocaine. A 700µgm slow-release intravitrealozurdex implant was placed in the vitreous cavity from pars plana route using a customized, single-use 22-gauge applicator. All injections were performed in an operating room. Patients were treated with a topical ophthalmic antibiotic for seven days after treatment.
Results
A total of 35 eyes of 30 patients were included. The population consisted of 23 males (76.7%) and 7 females (23.3%) with a mean age of63.23 ± 12.8years ranging between 55-71 years. Group 1 had 19 eyes of treatment naïve patients and group 2 had 16 eyes of refractory diabetic macular edema(DME) treated with intravitreal anti VEGF in past (table-1).
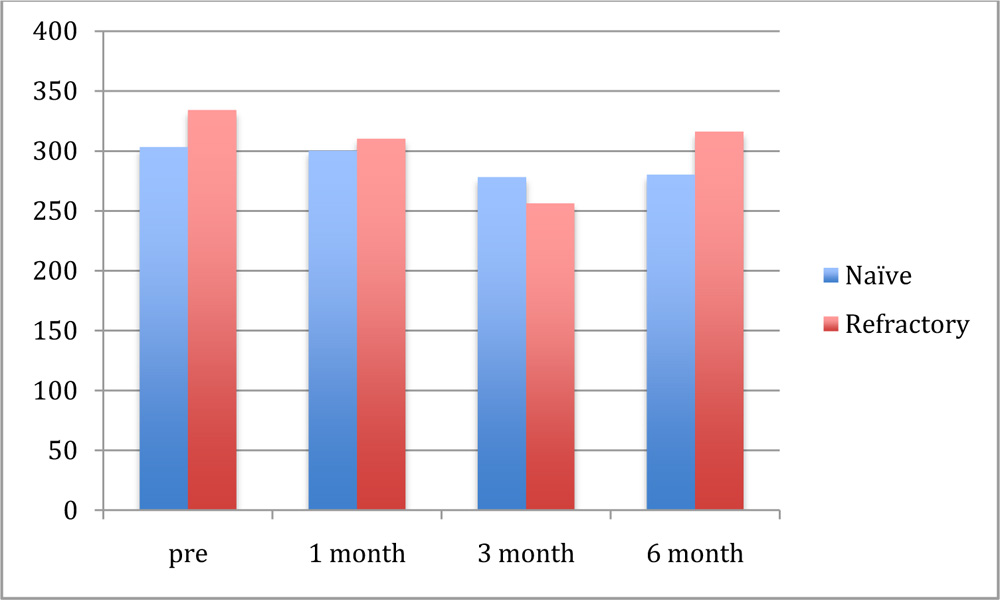
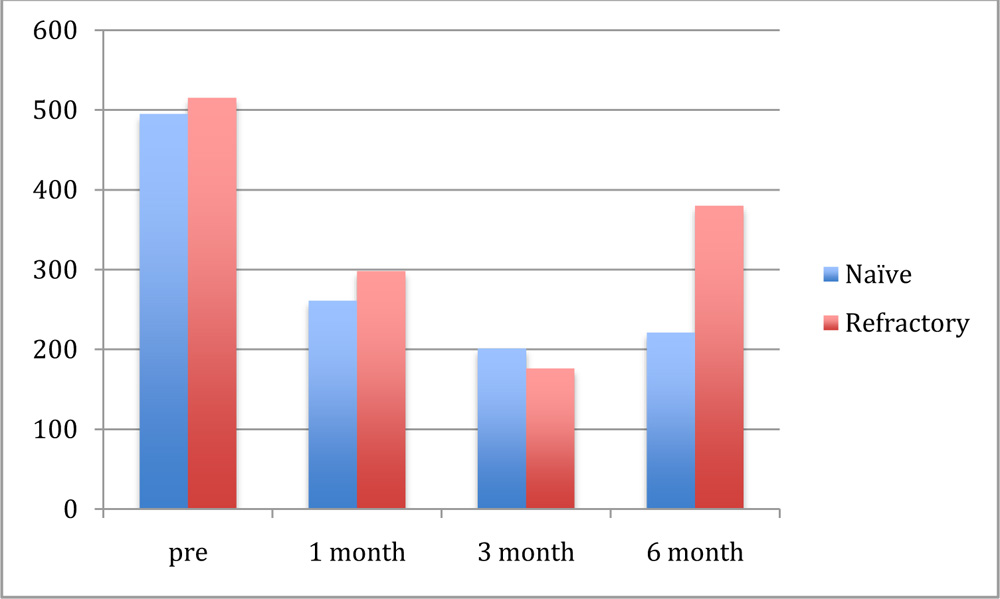
Baseline values of clinical measurement: Prior to the ozurdeximplant, both the groups showed significant macular edema. The average central foveal thickness (CFT) in group 1 was 495±240.8 µm and group 2 was 515±168.5µm. The average choroidal thickness by EDI was 303±36.7 µm and 334±90.5µmin group 1 and group 2 respectively.
Baseline snellens’ visual acuity converted to Log MAR in group 1 was0.74± 0.45 and group 2 was 0.69±0.32.
On follow up visits at 1 month, 3 month and 6 months the BCVA in LogMAR was0.6±0.75, 0.37±0.20, and 0.49± 0.32in group 1 and 0.57±0.32, 0.50±0.24, and 0.54±0.32 in group 2 respectively(Graph-1). The improvement till 3 months was statistically significant. At 6 months fall in BCVA was noted in both the groups, which was statistically insignificant. BCVA change from baseline was -0.25 logMAR in group 1 & -0.15logMARin group 2 at 6 months.
After treatment with ozurdex implant the change in CFT was noted in group 1 and group 2. It was 261±116.7µm, 201± 98.9µm and 221±104.5µm in group 1 and 298±121.3µm, 176± 101.2µm and 380± 203.2µmin group 2 at 1 month, 3 month and 6 months respectively(Graph-2). The reduction in CFT was significant (p<0.001) in group 1 till three months and showed a non significant rise(p>0.001) at 6 months. In group 2 the reduction was non significant and further increase at 6 months was noted.Central foveal thickness (CFT) change was 274 μ in group 1 and 135 μ in group 2 at 6 months.
The variation in choroidal thickness by EDI was also seen. Group 1 patient showed 300±78.3µm, 278±48.9µm and 280±32.6µm however group 2 showed 310±45.6µm, 256±79.3µm,316±88.9 µmthickness at 1 month , 3 months and 6 months. Group 1 showed a significant decrease at 3 months however this change was insignificant in group 2. Reduction in the choroidal thickness by enhanced depth imaging (EDI) was 23.3 μ and 18.2 μ in group 1 and group 2 at 6 months.
Cataract developed in 4 eyes (11.4%), which was removed surgically. 3 patients (8.57%) showed increase in IOP after intravitrealozurdexinjection which was managed medically.
None of the patient developed endophthalmitis post injection.
Discussion
This study was performed to evaluate the comparison of efficacy of intravitrealOzurdex implant in two groups of patients affected by DME. Group 1 patients which are treatment naïve showed better results as compared to group 2 patients who were refractory to treatment with anti-VEGF. In particular, we evaluated the BCVA, CFT and EDI during six months of follow-up. The results showed that ozurdex implant induced an improvement of BCVA, as measured by Snellen’s, after one and three months.There was a deterioration noticed at 6 month follow up.
In addition, we observed a reduction of CFT after one and three months from implants, while at six months, CFT values were not statistically different from baseline. EDI reduction were also seen which were significant in treatment naïve patients.
These data demonstrate that the greatest efficacy of dexamethasone is obtained within the first three months. After that, its therapeutic efficacy slowly decreases, although this effect is more pronounced in CFT than in BCVA and EDI measurements.
Other reports have also shown that the anti-inflammatory action of dexamethasone is rapid and may produce beneficial effects within the first week of treatment.4,9,10–15This effect might be attributable to the strong anti-inflammatory and anti-edema properties of the dexamethasone.
Although our findings further provide evidence for the use of Ozurdex in DME, there are some limitations to our data interpretations. First, the number of eyes examined is relatively low with a short follow-up period, and hence, it is difficult to reach definitive conclusions. Second, we acknowledge that a control group is missing. Further studies with greater cohorts of subjects and longer follow-up are required to characterize the pharmacokinetics and therapeutic efficacy of ozurdex implant in DME.
Conclusion
In conclusion,our findings suggest that the effects of ozurdex implant are more pronounced in treatment naïve patients as compared to chronic DME patients.Our study demonstrates the efficacy and safety profile of the intravitrealozurdex implant within the six-month time frame. Although individual response and metabolic state of the patient should be strictly monitored.
References:
- Richter B, Kohner E. Medical interventions for diabetic retinopathy. In: Wardnold R, Smeeth L, Henshaw K, eds. Evidence-Based Ophthalmology. London: BMJ Books; 2004:331–40.
- Antcli RJ, Marshall J. e pathogenesis of edema in diabetic maculopathy. SeminOphthalmol. 1999;14(4):223–32.
- Rechtman E, Harris A, Garzozi HJ, Ciulla TA. Pharmacologic therapies for diabetic retinopathy and diabetic macular edema. ClinOphthalmol. 2007;1: 383–91.
- Haller JA, Dugel P, Weinberg DV, Chou C, Whitcup SM. Evaluation of safety and performance of an applicator for a novel intra-vitreal dexamethasone drug delivery system for the treatment of macular edema. Retina. 2009;29:46–51.
- Pacella F, Smaldone G, Albanese G, et al. Treatment chronic macular edema in Vogt-Koyanagi Harada syndrome with dexamethasone intravitreal implant: description of three case. Senses Sci. 2015;2(1):57–63.
- Wang K, Wang Y, Gao L, Li X, Li M, Guo J. Dexamethasone inhibits leuko- cyte accumulation and vascular permeability in retina of streptozotocin-induced diabetic rats via reducing vascular endothelial growth factor and intercellular adhesion molecule-1 expression. Biol Pharm Bull. 2008;31:1541–6.
- Tamura H, Miyamoto K, Kiryu J, et al. Intravitreal injection of corticosteroid attenuates leukostasis and vascular leakage in experimental diabetic retina. Invest Ophthalmol Vis Sci. 2005;46:1440–4.
- Martidis A, Duker JS, Greenberg PB, et al. Intravitreal triamcinolone for refractory diabetic macular edema. Ophthalmology. 2002;109(5):920–7.
- Kuppermann BD, Blumenkranz MS, Haller JA, et al; Dexamethasone DDS Phase II Study Group. Randomized controlled study of an intravitreous dexamethasone drug delivery system in patients with persistent macular edema. Arch Ophthalmol. 2007;125:309–17.
- Haller JA, Kuppermann BD, Blumenkranz MS, et al; Dexamethasone DDS Phase II Study Group. randomized controlled trial of an intravitreous dexamethasone drug delivery system in patients with diabetic macular edema. Arch Ophthalmol. 2010;128:289–96.
- Rishi P, Rishi E, Kuniyal L, Mathur G. Short-term results of intravitrealdexa- methasone implant (OZURDEX®) in the treatment of recalcitrant diabetic macular edema: a case series. Oman J Ophthalmol. 2012;5(2):79–82.
- Kuppermann BD, Chou C, Weinberg DV, et al; Dexamethasone DDS Phase II Study Group. Intravitreous dexamethasone effects on di erent patterns of diabetic macular edema. Arch Ophthalmol. 2010;128(5):642–3.
- Bezatis A, Spital G, Höhn F, et al. Functional and anatomical results after a single intravitrealozurdex injection in retinal vein occlusion: a 6-month follow-up – the SOLO study. ActaOphthalmol. 2013;91(5):e340–7.
- Schmitz K, Maier M, Clemens CR, et al; German Retinal Vein Occlusion Group. Reliability and safety of intravitrealozurdex injections: the ZERO study. Ophthalmologe. 2014;111(1):44–52.
- Meyer CH, Klein A, Alten F, et al. Release and velocity of micronized dexamethasone implants with an intravitreal drug delivery system: kinematic analysis with a high-speed camera. Retina. 2012;32(10):2133–40.
Table 1 Demographic data
| Group 1 | Group2 | |
| No of patients | 19 | 16 |
| No of males | 17 | 11 |
| No of females | 2 | 5 |
| Mean Age | 61.42 ± 10.2 | 65.04 ± 15.4 |
| Study horizon | 6 months | 6 months |
Graph-1 BCVA variation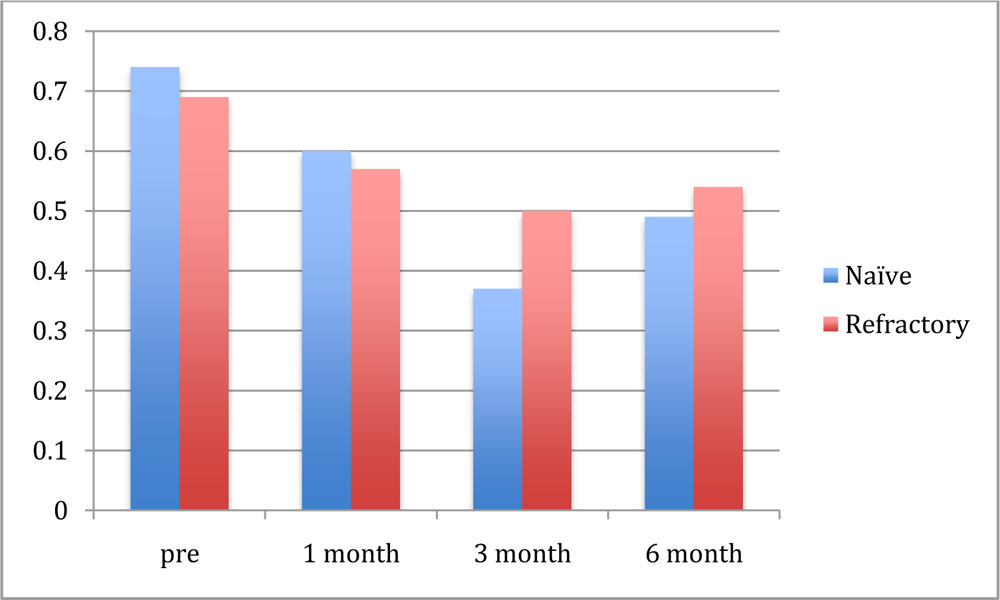
Graph 2 CFT variation
 Graph-3 EDI variation
Graph-3 EDI variation