Dr. Meena Chakrabarti C06675, Dr. Arup Chakrabarti
CLOSING MACULAR HOLES WITH ‘MACULAR PLUG’ WITHOUT GAS TAMPONADE AND POSTOPERATIVE POSTURING : A STUDY OF THE SD-OCT CHANGES FOLLOWING SURGERY
(FP 75)
Dr. Meena Chakrabarti MS1
Preethi Benjamin DO1,
Arup Chakrabarti MS1,
- Chakrabarti Eye Care Centre, No 102, Kochulloor, Trivandrum, India Pincode 695011 Corresponding Author:MeenaChakrabarti: Senior Consultant, Chakrabarti Eye Care Centre, No 102, Kochulloor, Trivandrum, Kerala, India, Pincode 695011
Email: drmeenachakrabarti@gmail.com
ABSTRACT
Purpose:
To study the SD-OCT changes following macular hole surgery without gas tamponade in large stage 3 and 4 macular holes of mean base diameter (MBD) 1364µm.
Methods:
18 eyes with large stage 3 or 4 macular holes and a MBD of 1364µm ± 224µm underwent modified MHS with broad ILM peel, inverted ILM flap and use of autologous gluconated blood to form a macular plug. SD-OCT evaluation was performed serially till 12 months postoperatively to assess restoration of outer retinal integrity.
Results:
Macular hole closure rate by 30th postoperative day was 100%. Closure of inner retinal layers was observed in all patients on day one. The defect in the outer retinal layer (RPE, Ellipzoid Zone, ELM) observed on first postoperative day was smaller than the preoperative base diameter and persisted for 7 to 30 days. Persistent defect in ellipsoid zone was seen in 2 patients up to 12 months. Floating redundant ILM was observed in 4 eyes.
Conclusion:
Large stage 3 and 4 MH closed slowly and completely by the 1st postoperative month in patients who underwent surgery by this modified method.
Introduction:
Idiopathic macular hole is one of the major vitreoretinal interface disorders that cause metamorphopsia and poor central vision in the elderly. They are most common in women1, 2 and have an estimated incidence of 7.8 new cases per 100,000 population per year3.
The success of macular hole surgery has improved significantly over the past 20 years since Kelly and Wendel4 first reported successful closure of idiopathic macular holes with pars planavitrectomy and fluid-air exchange in 1991. The surgical technique has evolved constantly with greatly improved anatomic and functional results and the macular hole closure rate has increased steadily from 58%4 to over 95% in recently reported studies6-12.
Taking into account the controversies regarding use of air or gas and duration of postoperative posturing, the purpose of this study was to determine the efficacy of modified techniques of macular hole surgery without intraoperative fluid air exchange, air or gas tamponade or postoperative posturing in large stage 3 and 4 macular holes. Other modification in techniques included a broad ILM peel, inverted ILM flap repositioning (ILMR) and use of autologous gluconated blood clumps (AGBC) as a macular plug to close the macular hole.We studied the SD-OCT changes following macular hole surgery without gas tamponade in large stage 3 and 4 macular holes of mean base diameter (MBD) 1364µm
Method:
A retrospective consecutive chart review of all patients with stage 3 and stage 4 macular holes, and a mean base diameter ≥ 1000µm who underwent macular hole surgery by a single surgeon (MC) at Chakrabarti Eye Care Centre, India from 2013 November to 2014 November wasperformed. The institutional review board of Chakrabarti Eye Care Centre, Trivandrum, India, approved this retrospective study. This study included only patients with large stage 3 and 4 macular holes ≥ 1000µm mean base diameter. Patients with history of retinal detachment, proliferative retinopathy, any kind of retinal surgery, diabetic retinopathy, vitreous haemorrhage, retinal vascular occlusions, uveitis, trauma, optic atrophy or glaucoma were excluded from the study. We also excluded all patients with incomplete case records and inadequate follow-up data. The data collected from the patient case records included age, sex, preoperative best corrected visual acuity measured in Snellens and converted into log MAR units, preoperative lens status, mean base diameter of the macular hole, any variation in surgical technique, postoperative OCT findings, postoperative best corrected visual acuity in log MAR units, intraocular pressure as well as whether cataract surgery was necessary in the postoperative period.
Optical Coherence Tomography was performed before and after surgery in all eyes using Cirrus 500 (Carl Zeiss, USA) Spectral Domain OCT machine with macular volume scan protocol (macular cube volume 518×518 and 200×200) as well as HD 5 line Raster enhanced depth imaging (EDI) scan protocol to detect any defect or discontinuity in the macular area and also to assess restoration of outer retinal layer integrity at various previously specified periods of time in the postoperative period.
The modified macular hole surgery in all these patients had the following steps
- 23 gauge parsplanavitrectomy (Geuder S3 VIP, Germany) under retrobulbaranesthesia in all patients
- Delineation of posterior cortical vitreous and posterior hyaloid face with preservative free triamcinolone acetonide (Aurocort, Aurolab, India). Posterior vitreous detachment (PVD) was induced by the suction of the 23 gauge cutter. In eyes with pre-existing PVD vitrectomy was carried out without using Triamcinolone acetonide.
- Epiretinal membrane peeling whenever present.
- Broad area of ILM peeling extending from arcade to arcade after staining the retinal surface with Brilliant Blue G (Ocublue Plus 0.05%, Aurolab, India) in 18 eyes (Fig 1). The concentration of Ocublue used was 0.05% and the exposure time was approximately 30 seconds. In 2 eyes, autologous gluconated blood clumps (AGBC) was used for ILM peeling. The autologous gluconated blood clumps solution was prepared immediately before starting the surgery by adding 1 ml of 5% Glucose to 2cc of autologous blood drawn from the patients’antecubitalvein inthecubital fossa with strict attention to aseptic precautions during blood withdrawal.
- A broad area of ILM peel was achieved by performing a double maculorrhexis. After circumferential ILM peeling, the ILM flap raised by the inner rhexiswas left attached to the edges of the macular hole and was inverted and gently nudged under the hole in multiple layers. The flap raised by the outer rhexis was removed. The free floating edge of this attached ILM flap, if in excess was trimmed down using the 23 g cutter before inverting the flap to fill the hole. An attempt was made to gently approximate the edges of the hole by stroking the margins of the hole with a blunt 23g spatula. The inverted ILM flap waslayered over with AGBC. The AGBC and ILM formed a loose plug closing the macular hole. (Fig 2).The macular plug thus formed comprised of multiple layers of inverted ILM layered over with autologous gluconated blood clumps.
- The peripheral vitrectomy was completed and the peripheral retina scrutinized carefully for any retinal breaks
- No fluid – air exchange or gas tamponade was performed and the patients were allowed to adopt any comfortable posture.
All patients were reviewed postoperatively on day 1, 7, 30 and thereafter at 2 monthly intervals for 12 months. At each postoperative visit, BCVA, non contactapplanation tonometry (Pulsair, Keeler); dilated slit lamp evaluation for lens changes, biomicroscopic examination of macula using 78 D lens (Volk); indirect ophthalmoscopy, fundus photography as well as Optical Coherence Tomography were performed.
For the purpose of this study, the outcome measures were defined as follows:
- Anatomical success was closure of macular hole defined as absence of neurosensory defect over the fovea in the spectral domain OCT images.
- Functional success was defined as a 2 step improvement in the log MAR visual acuity from the preoperative baseline vision by the 30th postoperative day review.
- Restoration of outer retinal layer integrity was assessed by restoration of the 4 hyperreflective lines that are seen in the outer retinal layers of a SD – OCT image namely RPE ( Retinal Pigment Epithelium); COST line(Cone Outer Segments Tip Line),Ez: (Ellipsoid Zone) or Inner Segment – Outer Segment Junction, ELM: (External Limiting Membrane).
SPSS software version 17.0 (SPSS, Inc, Chicago, IL) was used to perform statistical analysis of the data and a p value ≤ 0.05 was considered as statistically significant.
Results:
The study population included 18 eyes of 18 patients who underwent repair of macular hole by a modified surgical technique from 2013 – 2014
The preoperative baseline characteristics were as follows (Table: 1).The patients were in the age group ranging from 40 – 79 years (Mean ± SD = 62.8 years ± 7.3). The study group included 6 males and 12 females. The median preoperative visual acuity was6/60 (1.0logMAR units). Preoperative visual acuity of ≤ 6/60 (≤ 1.0 log MAR units) was present in 69.53% of patients. 76.92% patients were phakic and 23.10% patients were pseudophakic. 76.92% had stage 3 macular holes while 23.10% had stage 4 macular holes. The base diameter of the macular holes as measured from Spectral Domain OCT images ranged from 1000µm to 1728µm, with a mean base diameter of 1364 ± 349µm SD. The mean duration of symptoms before the diagnosis and surgery was 6.4 months ± 3.1. 46.3% of patients had vitreoschisis detected pre or intra operatively. Epiretinal membranes were present in 23.10% patients and were removed during surgery.
All patients were reviewed on day 1, 7th and 30thpost operative days and thereafter at 3 monthly intervals for 12 months. At each postoperative visit a best corrected visual acuity in log MAR units, slit lamp examination to assess progression of lens changes, 78 D slit lamp biomicroscopic evaluation of macula, fundus imaging and 3D spectral domain OCT were performed. 34.61% had a closed macular hole on the first postoperative day, and 80% of the holes were closed by the 7thpost operative day. By the 1st month follow up review the hole closure rate was 100%.
The functional efficacy of this procedure was judged by a 2 step improvement of best corrected visual acuity at 2 points of time in the post operative period. Table 2 gives the distribution of BCVA at different time intervals. The median BCVA improved from the preoperative value of 1.0 logMAR units to 0.5logMAR units at 1 month and 12 monthspostoperatively. While only 15.38% of patients had a visual acuity ≥6/24 preoperatively; 77.91% and 100% of patients respectively could achieve a visual acuity ≥6/24(0.5 log MAR) at 1 month and 12 months postoperatively. It was observed that the relationship between the preoperative and postoperative 1 month and 12 months BCVA was statistically significant (p= 0.000) by the Wilcoxon signed rank test. However the improvement in BCVA from the 1st to 12th postoperative month was not statistically significant. (Table 3)
Taking into account both hole closure as well as 2 step improvement in log MAR visual acuity in the postoperative period, success rate of 100% was seen at postoperative 1 month period and maintained at the 12th month postoperative follow up.
3 out of the 18 phakic eyes needed cataract surgery within the 12 months postoperative follow up period. The mean IOP was 15.7 mm ±4.75 and none of the patients required anti – glaucoma medications in the postoperative period. No significant intraoperative or postoperative complications were noted.
All patients underwent serial OCT evaluation using the 518 × 518 and 200 × 200 volume scan protocols of SD OCT (Cirrus 500, Carl Zeiss Meditech USA) as well as the HD 5 line Raster enhanced depth imaging protocol. The OCT scan images were analysed for restoration of outer retinal layer integrity by observing the restoration of the four hyper reflective lines in the outer retinal layers namely RPE, COST line, EZ and ELM.Total restoration of the outer retinal integrity was observed in all patients by the end of the first postoperative month in 18 eyes and partially in 2 eyes. These two eyes had a small outer retinal layer defect which persisted at the 12th month follow up visit and was not associated with non improvement in vision. It was also observed that although the inner retinal layer closure could be achieved on the first postoperative day in all eyes, the defect in the outer retinal layers though smaller persisted for varying periods of time from 7th to the 30th postoperative day. In two patients, the small defect in the ellipsoid zone persisted up to 12 months.
Discussion:
The success of macular hole surgery has improved significantly over the past 20 years since Kelly and Wendel4 first reported successful closure of idiopathic macular holes with parsplanavitrectomy, and fluid air exchange in 1991. The surgical technique has evolved constantly with improved hole closure rate from 58% to over 95% with various modifications in the surgical strategy. The surgical procedure for closure of macular holes has several controversial steps with respect to the staining and peeling of ILM, selection of intraocular tamponade agents and the optimum duration of face down posturing or whether posturing is necessary in the postoperative period. Review of available literature shows a general consensus in performing fluid air exchange and using gas or air endotamponadeintraoperatively. Studies advocating no- or- reduced duration postoperative posturing15-22 have advocated phacovitrectomy as a primary procedure to tackle the expected cataract progression or have excluded phakic eyes.23
Optical Coherence Tomography studies on macular holes in the postoperative period have demonstrated early closure of macular holes .Themost importantdeterminant factors to achieve macular hole closure is therefore to allow the MH to remain dry long enough to achieve apposition which naturally occurs within 1 week. Whether it is achieved by staying up straight, head bend in reading posture, or totally avoiding a supine position or face down posture appears to be immaterial as long as there is a sufficiently large intraocular gas bubble for the required period of time.
Majority of patients who undergo macular hole surgery are elderly and have associated co- morbid condition such as arthritis, higher risk for thrombo-embolic and neurological complication and are usually living alone and fending for themselves. All these factors contribute to poor patient compliance and reduction in the effectiveness of any form of postoperative posturing.
Taking all these factors into consideration and also considering the fact that the compliance in our part of the Asian continent is exceptionally poor, we had eliminated fluid air exchange, endotamponade and postoperative posturing altogether. Our results show that this modification in the surgical technique did not compromise the hole closure rate or functional recovery, and at the same time reduced the incidence of cataract progression and secondary postoperative intraocular pressure spike.
34.61% had a closed macular hole by day 1, 80% by day 7 and100% by the 30th postoperative day review. These results compare favourably to results published for MHS with gas tamponade and no face down positioning where a closure rate varying from 60%27 to 100%28 have been reported by various authors.29-31
In all 18 patients who underwent macular hole surgery without gas tamponade or postoperative posturing, a broad area of internal limiting membrane extending across the arcade was peeled. Iezzi32 et al have demonstrated the efficacy of no face down positioning and broad ILM peel in the surgical repair of idiopathic macular holes with MBD 609.6 ± 226.2µm. The authors used non expansile SF6 (20%) gas and no face down positioning, and maintenance of a reading posture with eye decline at 450 downward gaze without neck or face position adjustments for 3 to 5 days post operatively. Broad ILM peeling of a taut ILM will help remove all tangential tractional forces on the macula, allowing the retina to relax sufficiently. In our series of 26 cases, review of recorded videos of all patients demonstrated that there was a significant shrinkage of the hole size, a change in the rounded taut contour of the hole to a crenated appearance of the hole edges as well as the ability to re-approximate the hole edges (a slit- like defect) by gently massaging with a blunt 23g spatula from the superior and the inferior quadrants. This maneuver was not associated with retinal pigment epithelium mottling or compromised functional recovery in the post operative period.
Despite good visual and anatomic results following macular hole surgery reported by a number of authors, OCT evaluation demonstrate that 19% – 39% of the so called closed holes are actually flat open holes with bare retinal pigment epithelium. Large macular holes usually close this way. ZofiaMichalewskaet al9 reported on the inverted ILM flap technique to avoid a flat open appearance in large macular holes. Michalewska and co authors hypothesized that the inverted ILM flap technique stimulates proliferation of glial cells that fill the macular hole thereby enchancing macular hole closure rates and improving postoperative visual acuity.
In the first few months after macular hole surgery with inverted ILM flap, SD OCT demonstrated an improvement in foveal contour and increased tissue coverage of the macular hole.32
An important limitation of this technique is that the inverted ILM may detach during the intraoperative maneuvers or may detach spontaneously during fluid air exchange33 and hence newer techniques for maintaining the inverted ILM flap on the macular surface are necessary. Chi-Chun Lai et al31reported on a modification of inverted ILM flap repositioning in highly myopic eyes with macular hole retinal detachment (MHRD). They utilized a modified combination therapy, vitrectomy with inverted ILM repositioning (ILMR) combined with autologous blood clot and gas tamponade with one day prone positioning to treat MHRD in highly myopic eyes. Chi-Chun Lai et al33 claimed that the main advantages of this technique are that the inverted ILMR would fill the entire hole and provide a stronger bridge for proliferation of glial cells. The ILMR and the autologous blood clump would become a plug that will seal the macular hole in minutes. An additional advantage is the presence of growth factors in the platelets of the blood clot that can promote healing.
In our series of 18 patients with large stage 3 or 4 macular holes, 100% hole closure was achieved with utilisation of the inverted ILM flap and AGBC to form a macular plug . Our technique varied from the technique used by Nawrocki33 et al and Chi- Chun Lai31et al in the following aspects.
While harvesting the ILM flap for inverted ILM repositioning we took care to ensure that a broader area of attached ILM was present around the margin of the hole by performing a 2 stage maculorrhexis.(Fig 1) When the ILM flap repositioning maneuver was performed usually this area of attached ILM separated to the margins of the hole.
Multiple layered ILM flap repositioning was performed. Postoperative OCT images in 2/15 eyes showed that a part of the ILM had dislodged from the well of the hole and was floating in the vitreous cavity still attached to the edge of the hole while the hole appeared closed.A slightly elevated hyperreflective tissue probably representing the macular plug was observed on first postoperative day in OCT images.
No obvious signs of retinal toxicity in the form of retinal pigmentary degeneration were observed. The blood clump solution is cost effective, easily available and has in addition growth factors to promote healing. The possible risk of infection with use of autologous blood products should not be ignored and strict aseptic precautions should be followed during blood withdrawal and mixing with 5% glucose. Potential contamination can be further reduced by preparing the AGBC solution immediately before vitrectomy.
Eliminating postoperative face – down positioning and the complications of gas tamponade without compromising the macular hole closure rate has significant advantages for the patient. Although this study has intrinsic limitations because of the retrospective nature of its design and small sample size, our data provides strong evidence in asmall series of patients with large stage 3 and 4 macular holes that comparable or even better hole closure rates can be achieved without gas tamponade. The technique used could have been simplified if AGBC alone was used for delineating the posterior hyaloid face for PVD induction, visualization for ILM peeling and to develop the macular plug after ILM flap inversion. A larger series of cases utilizing this technique will provide strong and conclusive data on the efficacy of macular hole surgery without gas tamponade or postoperative positioning.
References:
- Wang S, Xu L, Jonas J B. Prevalence of full thickness macular holes in urban and rural adult Chinese: the Beijing Eye Study. Am J Ophthalmol 2006; 141: 589-591
- Jackson T L, Donachie P H, Sparrows J M, Johnston R L: The United Kingdom National Ophthalmology Database Study of Vitreoretinal surgery: report 2; macular hole. Ophthalmology 2013 Mar; 120(3): 629-634.
- Mc Cannel C A, Ensminger J L, Diehl N N, Hodge D N. Population based incidence of macular holes. Ophthalmology 2009 Jol; 116(7): 1366-1369.
- Kelly N E, Wendel R T. Vitreous surgery for idiopathic macular holes: Results of a pilot study. Arch Ophthalmol 1991; 109: 654-659.
- Sakamoto T, Ishibashi T. Visualising vitreous in vitrectomy Graefers Arch ClinExpOPhthalmol 2009; 247: 1153-1163
- Fabian I D, Moisseiev J. Suturelessvitrectomy: evolution and current practices. Br J Ophthalmol 2011; 95: 318-324
- Park D W, Sipperley J O, Sneed S R, Dugel P U, Jacobsen J. Macular hole surgery with internal limiting membrane peeling and intravitreous air. Ophthalmology 1999; 106: 1392
- Brooks H L Jr. Macular hole surgery with and without internal limiting membrane peeling. Ophthalmology 2000; 107: 1939-1948; discussion 1948-1949
- ZofiaMichalewska, JanuszMichalewski, Jerzy Nawrocki et al. Macular hole closure after vitrectomy: The Inverted ILM flap technique. Ophthalmology 2010; Vol 47 Issue 10: 2018-2025
- Bainbridge J, Herbert E, Gregor Z. Macular Holes: Vitreo retinal relationships and surgical approaches. Eye (Lond) 2008; 22: 1301-1309
- Thompson J T, Glaser B M, Sjaarda R N, Murphy R P, Hanham A. Effects of Intraocular bubble duration in the treatment of macular holes by vitrectomy and transforming growth factor – beta2. Ophthalmology 1994; 101: 1195-1200
- Thomson J T, Smiddy W E, Glaser B M, Sjaarda R N, Flynn H W Jr. Intraocular tamponade duration and success of macular hole surgery. Retina 1996; 16: 373-382.
- Berger, Jeffrey W, Brucker, Alexander J. The magnitude of the bubble beyond pressure: implications for macular hole surgery. Retina 1998; Vol 18(1): 84-85
- Thompson J T, Sjaarda R n, Glaser B M et al. Increased intraocular pressure after macular hole surgery. Am J Ophthalmol 1996; 121: 615-622.
- Tornambe P E, Poliner L S, Grote k et al. Macular hole surgery without face down positioning. A Pilot study. Retina 1997; 17: 179-185.
- Sato Y, Isomac T. Macular hole surgery with internal limiting membrane removal, air tamponade and one day positioning. Jpn J Ophthalmol 2003; 83: 289-292.
- Krohn J. Duration of face down positioning after macular hole surgery: a comparison between 1 week and 3 days. Acta Ophthalmol Scand 2005; 83: 289-292.
- Jaycock P, Bunce C, Xing W et al. Outcomes of macular hole surgery: implications for surgical management and clinical governance. Eye 2005; 19:879-884.
- Guillaubey A, Malvitte L, La fontaine P O et al. Comparison of face – down and seated position after idiopathic macular hole surgery: a randomized clinical trial. Am J Ophthalmol 2008; 146(1): 128-134.
- Lange C A, Membrey L, Ahmed N et al. Pilot randomized controlled trial of face down positioning following macular hole surgery. Eye (Lond) 2012; 26: 272-7.
- Tadayoni R, Vicant E, Devin F et al. A randomized controlled trial of alleviated positioning after small macular hole surgery. Ophthalmology 2011; 118: 150-5
- Solebo A L, Lange C A, Bunce A, Bainbudge J W. Face down positioning or posturing after macular hole surgery. Cochrane Database System Review 2011; (12): CD 008228.
- Iezzi R, Kapoor KG. No face-down positioning and broad internal limiting membrane peeling in the surgical repair of idiopathic macular holes. 2013;120:1998–2003.
- Jumper J M, Gallemore R P, McCuen et al. Features of macular hole closure in the early postoperative period using Optical Cohence Tomography. Retina 2000; 20: 232-237.
- Eckardt C, Eckert T, Eckardt U et al. Macular hole surgery with air tamponade and Optical Coherence Tomography – based to duration of face down positioning. Retina 2008; 28: 1087-1096
- Verma D, Jalabi M W, Watt W G, Naylor G. Evaluation of posturing in macular hole surgery. Eye 2002 Nov; 16(6): 701-704
- Ryan E A, Lee S, Chern S. Use of intra vitreal autologous blood to identify posterior cortical vitreous in macular hole surgery. Arch Ophthalmol 1995; 113: 822-823.
- Gaudric A, Massin P, Paques M et al. Autologous platelet concentrates for treatment of full thickness macular holes. Graefes Arch ClinExp Ophthalmology. 1995; 233: 549-554.
- Lai C C, Wu Wc, Chuang L H, Yeung L et al. Prevention of Indocyanine green toxicity to retinal pigment epithelium with whole blood in stain assisted macular hole surgery. Ophthalmology 2005; 112: 1409-1414.
- Lai C C, Hwang Y S, Liu L. Blood assisted internal limiting membrane peeling for macular hole repair. Ophthalmology 2009; 116: 1525-1530
- Chi – Chun Lai, Yen Po Chen, Nan – Kai Wang et al. Vitrectomy with internal limiting membrane repositioning and autologous blood for macular hole retinal detachment in highly myopic eyes.Published online http://www.aaojournal.org/article/s0161-6420 (15) 00539-4/ full text
- Raymond Iezzi, Kapil G Kapoor. No face – down positioning and broad internal limiting membrane peeling in the surgical repair of idiopathic macular holes. Ophthalmology 2013; 120: 1998-2003
- Nawrocki J, MichalewskaZ. Spectral domain OCT for macular holes. In Holz F G, Spaide R F ed Medical Retina: Focus on retinal imaging. Berlin: springer essentials in Ophthalmology. Krieglstein G K; Weinreb R N, series ed.LEGENDS
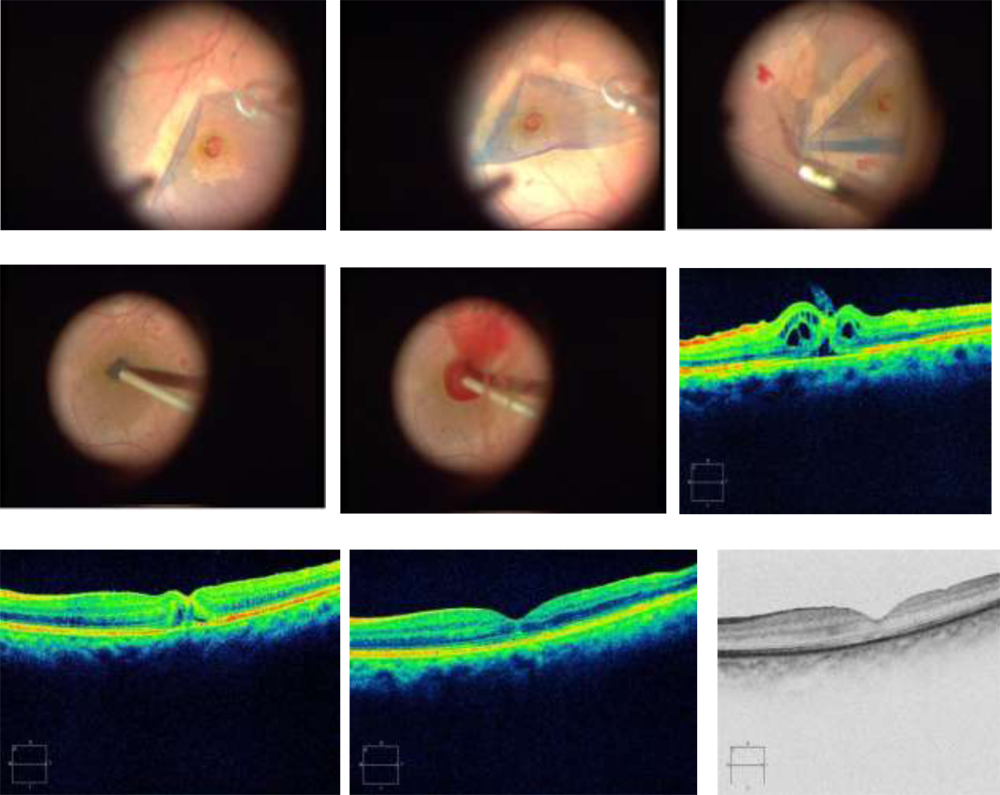
Fig 1 (a-i): Demonstrating the surgical technique and postoperativefollow- up SD-OCT images
(a – c ): Demonstrating the technique of double maculorrhexis to achieve a broad area of ILM peeling. Brilliant Blue G isused to stain the ILM and a Finesse Flex Loop (Greishaber) issused to raise the ILM flap.(d): The ILM flapraised by the innermaculorrhexisisgentlyinverted and repositedinto the well of the macularhole and gentlynudgedunder the holeedges. (e) Autologousgluconatedbloodclumpsislayered over the inverted ILM flap to form a macularplugclosing the macularhole . (f): SD-OCT image on the first postoperativeday shows closure of the innerlayers of the hole.Note the persistence of a smalltriangularshapeddefect in the outerretinallayers, and cystoid changes in the retina. A small tag of loose ILM isseenprotrudinginto the vitreouscavity (yellowarrow). (g): By the 7th postoperativeday the outerretinal layer defect has becomesignificantlysmaller,withmarkedreduction in the cystoid changes.(h &i): Complete holeclosurewithrestoration of outerretinalintegrityisachieved by the 14th postoperativeday.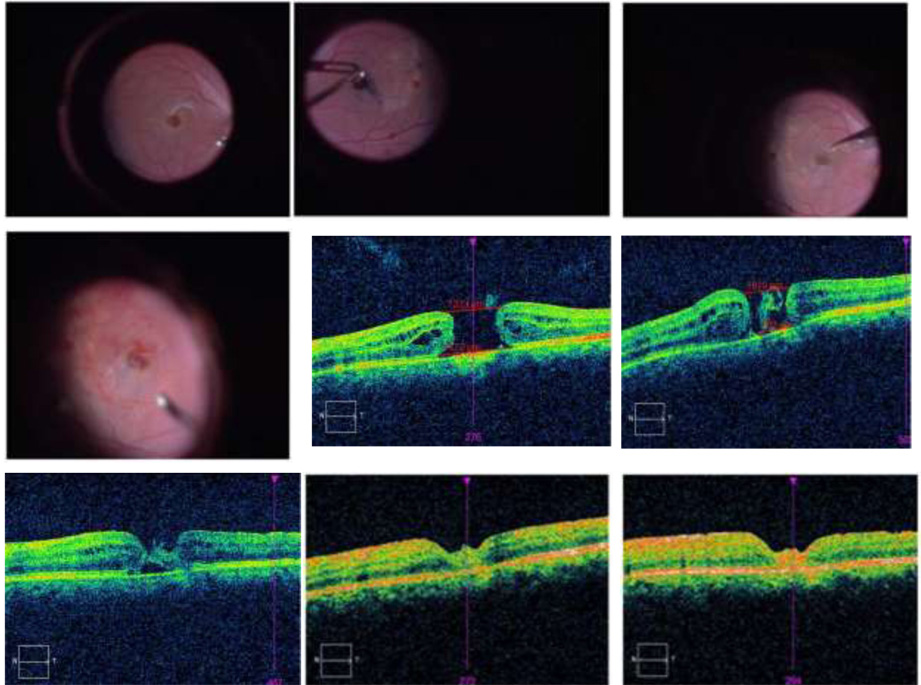 Fig 2 a-m: (a-d): Intraoperative still photography of modified macular hole surgery in a 40 year
Fig 2 a-m: (a-d): Intraoperative still photography of modified macular hole surgery in a 40 year
old laser technician who sustained an accidental laser burn while handling Q switched 1064 nm Nd YAG laser in a dermatology clinic.Note the presence of a partial posterior vitreous detachment around a large full thickness macular hole of base diameter 1428 microns. (b): Peeling of ILM stained with BBG using ILM forceps. (c): ILMR – Internal limiting membrane repositioning. (d): End of surgery showing the hole closed with inverted LM layered over with autologous gluconated blood clumps. (e): Preoperative SD-OCT image showing a full thickness macular hole of base diameter 1428 microns.(f): Postoperative day 1 image shows that the hole is smaller with hyper reflective tissue in the gap. (g): On day 7 the hole is partially closed by bridging tissue.A defect in the outer retinal layers is seen. (h): Day 14 shows partial restoration of foveal contour. (i) & (j) : Adequate hole closure with restoration of outer retinal integrity. Vision improved from 6/60 to 6/9. (k-m): Fundus pictures on day 1,7 and 30 in the postoperative period
TABLE 1: PREOPERATIVE BASELINE CHARACTERISTICS
| FACTORS | NO: OF CASES n= 26 (%) |
| 1. Mean Age ± SD | 62.8 ± 7.3 (Range : 40-79 yrs) |
| 2. Sex | |
| Male : | 6 (23.10%) |
| Female: | 20 (76.92%) |
| 3. Median BCVA | 6/60 (1.0 log MAR units) |
| ≤ 6/60 (1.0 log MAR) | 18 (69.53%) |
| ≥6/60 | 8 (30.47%) |
| 4. Lens Status | |
| Phakic | 20 (76.9%) |
| Pseudophakic | 6 (23.10%) |
| 5. Stage of Macular Hole | |
| Stage 3 | 20 (76.92%) |
| Stage 4 | 6 (23.10%) |
| 6. Hole size (Mean basal diametre) | 892.8 ± 349 µm SD |
| 7. Large holes ≥ 1000µm BD | 18 (69.23%) |
| 8. Duration | 6.4 ± 3.1months |
| 9. Vitreoschisis | 46.3% |
| 10. Epiretinal Membranes | 6 (23.10%) |
TABLE 2: DISTRIBUTION OF BCVA AT DIFFERENT INTERVALS OF TIME
| BCVA | PREOPERATIVE | POST OP 1 MONTH | POST OP 12 MONTHS | ||||
| logMAR | Snellen | No | % | No | % | No | % |
| 0.0 | 6/6 | 0 | 0.0% | 1 | 7.7 % | 3 | 11.54% |
| 0.2 | 6/9.2 | 0 | 0.0% | 0 | 3.84% | 5 | 23.07% |
| 0.3 | 6/12 | 0 | 0.0% | 3 | 23.07% | 4 | 15.38% |
| 0.5 | 6/18 | 4 | 15.38% | 6 | 38.46% | 4 | 30.77% |
| 0.6 | 6/24 | 1 | 0.0% | 2 | 3.84% | 2 | 19.23% |
| 0.8 | 6/36 | 1 | 0.0% | 2 | 7.7% | 0 | 0 % |
| 1.0 | 6/60 | 10 | 42.11% | 2 | 7.7% | 0 | 0 % |
| 1.08 | 5/60 | 2 | 7.7% | 2 | 7.7% | 0 | 0 % |
| 1.17 | 11.54% | 0 | 0 % | 0 | 0 % | ||
| 1.30 | 15.38% | 0 | 0 % | 0 | 0 % | ||
| 1.48 | 0 | 0.0% | 0 | 0 % | 0 | 0 % | |
| 2.00 | 0 | 0.0% | 0 | 0 % | 0 | 0 % | |
| MEDIAN | 1.00 | 0.50 | 0.50 | ||||
TABLE 3: EFFECTIVENESS OF TREATMENT ON BCVA
| BCVA | MEAN | SD | MEDIAN | PAIR | Z# | P |
| Preoperative | 1.00 | 0.32 | 1.00 | Pre op & post op 1 month | 4.66 | 0.000 |
| Post OP 1 month | 0.50 | 0.38 | 0.50 | Pre op & post op 12 months | 4.66 | 0.000 |
| Post OP 12 months | 0.50 | 0.39 | 0.50 |
(Significant at 0.01 level. Wilcoxon signed Rank test.)

