Dr. Shishir Shekhar Singh,S14445, Dr. Manish Nagpal, Dr. Hardik Jain, Dr.Navneet Mehrotra
INTRODUCTION:
In recent years, the development of anti-vascular endothelial growth factor (VEGF) agents that can be intravitreallyinjected has revolutionized the therapeutic approach for wet age-related macular degeneration (AMD).Its use for AMD was first introduced in 2005 with the off-label use of Bevacizumab (Avastin).1 This was followed in short sequence by the approval of anti-VEGF agents specifically designed for intraocular use [Ranibizumab (Lucentis) and aflibercept (Eylea)].2–4Tightly controlled multicenter randomized controlled studies have convincingly shown the treatment benefits for wet AMD patients with Ranibizumab (Lucentis, Genetech, San Francisco, California, USA), Bevacizumab (Avastin, Genetech, San Francisco, California, USA) and Aflibercept (Eylea, Bayer Healthcare Pharmaceuticals, Berlin, Germany).2-6In clinical trials, rigid treatment and follow-up protocols are usually mandated and patients are recruited according to strict inclusion and exclusion criteria to ensure validity of results.
To date, there is no indicator that would allow physicians to predict injection frequency, duration or visual acuity (VA) outcome in a particular patient. Currently, various anti-VEGF treatment protocols exist: fixed monthly or bimonthly injections,2–4 pro-re-nata (PRN) and treat-and-extend (TAE) protocol7. It is, however, often not possible to fully adhere to one of these protocols in daily practice since individual patient factors (both medical and organisational) have to be taken into account when scheduling an individual patient’s regimen.8–10
Furthermore, a heavy financial burden and compliance issues involved in routine clinical practice have led to dosing regimens that are less intensive than those used in clinical trials. It is uncertain whether identical outcomes from clinical trials will be replicated in the wider community in routine clinical practice.Here, we describe the visual outcome based efficacy of intravitreal Anti-VEGF agents (Ranibizumab and Bevacizumab) used as 3-monthly loading dose followed by PRN based therapy for recurrences over 24months duration in a population of wet AMD patients.
METHODS:
This was a single-centre, retrospective, study. The study was approved by the institutional ethics committee. Patients were informed about the off-label condition of intravitrealBevacizumab. The medical records of every patients receiving anti-VEGF injection (Ranibizumab and Bevacizumab) from February 2009 to June 2014 were reviewed.
The inclusion criteria were:
- Age > 50 years
- Diagnosed as Wet AMD based upon:
- Clinical findings on slit-lamp bio-microscopy or indirect ophthalmoscopy
- Optical coherence tomography (OCT) findings
- Fluorescein angiography (FA) findings
- Indocyanin green angiography (ICGA) findings (as and when required)
- OCT – Angiography (OCT-A) findings (as and when required)
- Follow up of at least 24 months
- Receiving 3–monthly loading dose of Anti-VEGFintravitreal injections, and further Anti-VEGF therapy on PRN basis for recurrences
- Regardless of
- Baseline best corrected visual acuity (BCVA)
- Choroidal neovascularization (CNV) characteristics:
- Size
- Location (subfoveal or juxtafoveal) and
- Composition (classic lesions including both predominantly and minimally classic types, or occult lesions).
The exclusion criteria were:
- History of photodynamic therapy (PDT) or intravitreal triamcinolone (TA) during follow-upperiod after Anti-VEGF
- Any form of combination therapy
- CNV attributable to any cause other than AMD, such as Myopic degeneration
- Either Polypoidalchoroidalvasculopathy (PCV) or Retinal angiomatous proliferation (RAP) confirmed by ICGA
- Presence of comorbid ocular conditions,particularly diabetic retinopathy, that might compromisevisual acuity.
39 eyes of 35 patients were studied. All patients received a comprehensive baseline ophthalmological examination, includingBCVA, intraocular pressure (IOP) measurements, indirect ophthalmoscopy and slit-lamp biomicroscopy. The documented findings from Spectralis(HRA + OCT) (Hidelberg Engineering) were reviewed. BCVA was recorded in Snellen decimal values and converted to the logarithm of the minimal angle of resolution (logMAR) units.
The intravitreal doses of 1.25 mg Bevacizumab or 0.5 mg Ranibizumab was injected in the operation theatre using 30-guage needle at 3-4 mm post-limbus. Prophylactic topical antibiotics were applied for 5 days after the injection. Anti-VEGF injection was given at the baseline visit, with a fixed 4-6 weekly injection regimen for the next 2months to all patients in the study. This was defined as the 3-monthly loading dose Anti-VEGF therapy.
Following the loading dose therapy, patients were re-treated on PRN basis for recurrences of CNV lesions (presence of persistent subretinal fluid, new onset of macular haemorrhage, worsening visual acuity, increased retinal thickening by OCT and/or increased leakage of CNV assessed by FA)over the 24 months study duration.
Post loading dose therapy, patients were divided into two subgroups on basis of developing recurrences requiring re-treatments or not:
- Group A: Eyes requiring retreatment on PRN basis for recurrences, n=17
- Group B: Eyes with no further recurrences, n=22
At each follow-up visit, data were collected on the patients’ BCVA, whether they received retreatment, OCT measurements (if available), and the date of visit. The visits closest to the time points at baseline, 4 and 24 months were analysed. Statistical analysis was performed using SPSS. Two-tailed paired T tests were used to compare VA between different time points (baseline, 4 and 24 months). A ‘p’ value of <0.05 was considered statistically significant.
RESULTS:
Of the wet AMDcases reviewed, 39 eyes of 35 patients met the inclusion criteria with follow-up of 24 months. All these patients received 3-monthly loading dose Anti-VEGF therapy. Out of these 39 eyes,17 eyes (Group A) developed recurrences requiring re-treatments done on PRN basis; whereas 22 eyes (Group A) did not develop any recurrences requiring re-treatments. Patientsdemographics and clinical characteristics are presented in Table 1.
Table 1: Demographics and clinical characteristics of wet AMD patients who completed 24months of follow-up
| Parameters | n (%) |
| Number of patients | 35 |
| Number of eyes | 39 |
| Male | 27 (77.14) |
| Female | 8 (22.86) |
| Age (mean) | 70.63 ± 7.77 |
| Number of injections (mean) | 4.59 ± 2.83 |
| Number of eyes required retreatments | 17 (43.59) |
| Number of eyes followed up for more than 24 months | 6 (15.38)
|
The mean baseline BCVA in logMAR units of the 39 eyes was 0.76 ± 0.40. This improved (although statistically non-significantly) to 0.66 ± 0.42 at the 4 months post 3-monthly loading dose therapy (p=0.088). The statistically non-significant improvement of visual acuity was not maintained at 24months (declined statistically significantly) (p= 0.001), and the mean BCVA in logMAR units was 0.78 ± 0.40 with the mean similar to baseline. (Figure 1, 2.1, 2.2)
Figure 1:All 39 eyes- Comparison between mean BCVA at baseline, at 4 monthspost loading dose therapy and at 24 months following PRN basis therapy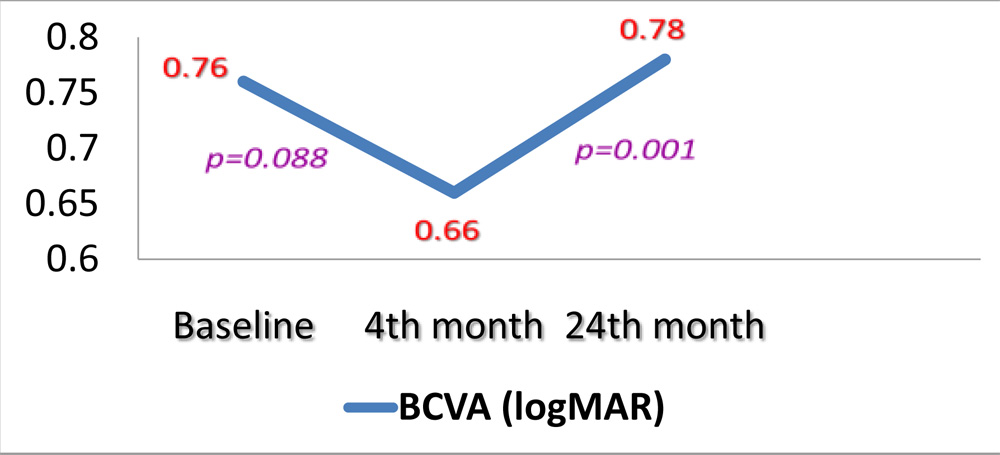
Figure 2.1:Fundus photograph OS shows greyish elevation of tissue deep to retina at macula suggestive of CNV in AMD (A). FA of OS shows a discrete, well-demarcated focal area of hyperfluorescence at macula (arrow) in early phase frame (B), with hyperfluorescence increasing in intensity and extending beyond boundaries of hyperfluorescence identified in early phase in later phase frame (C) suggestive of a classic CNV. OCT of OS shows the subretinalneovascular membrane at fovea (D).The visual acuity at presentation was OS 6/24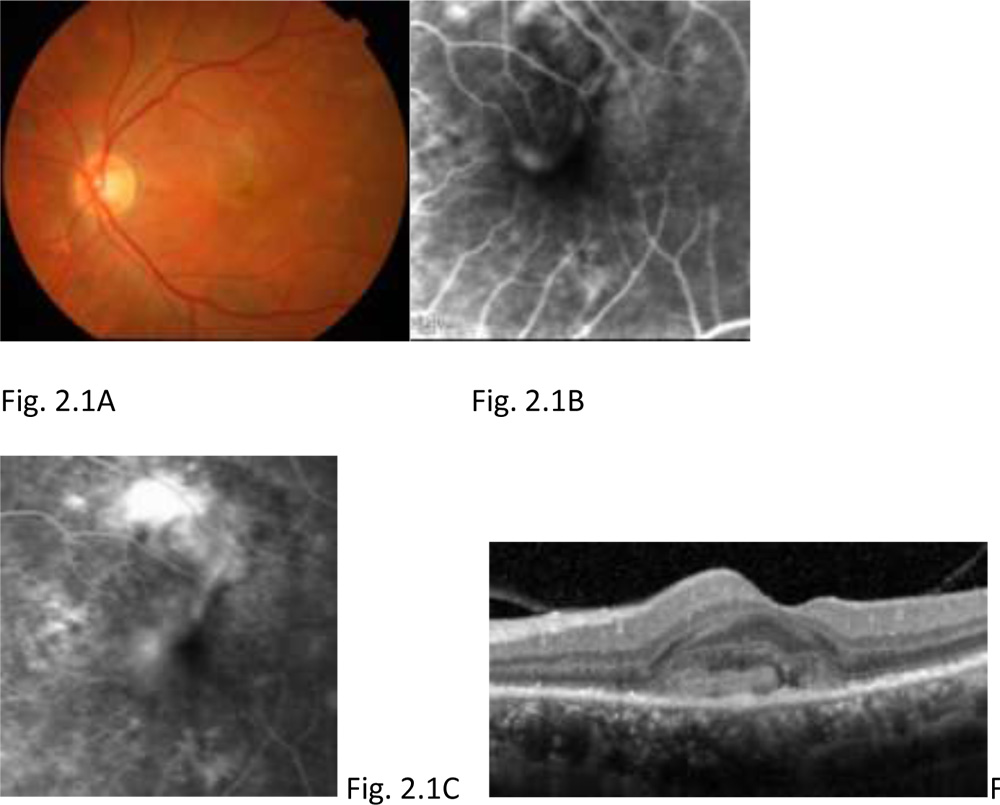
Figure 2.2: At 4 months, post 3-monthly loading dose Anti-VEGF therapy in OS. Fundus photograph OS shows resolving CNV in AMD (A). FA of OS in early (B) and late (C) phase shows an absence of the leakage from the CNV tissue compared to the pre treatment FA as seen in figure 2.1 (B & C). OCT of OS shows reduction in the subretinalneovascular membrane (D). The visual acuity at 4 months post 3-monthly loading dose therapy was 6/12
 Post 3-monthly loading dose therapy ,for the 17 eyes requiring retreatment on PRN basis for recurrences (Group A) the mean BCVA in logMAR units improved statistically non-significantly from baseline BCVA 0.57 ± 0.41 to 0.47 ± 0.34 at 4 months (p =0.209). The statistically non-significant improvement of visual acuity was not maintained at 24 months (declined statistically significantly) (p= 0.002), and the mean BCVA in logMAR units was 0.69 ± 0.35, which was a non-significant decline compared to the baseline (p= 0.290). (Figure 3, 4.1, 4.2, 4.3)
Post 3-monthly loading dose therapy ,for the 17 eyes requiring retreatment on PRN basis for recurrences (Group A) the mean BCVA in logMAR units improved statistically non-significantly from baseline BCVA 0.57 ± 0.41 to 0.47 ± 0.34 at 4 months (p =0.209). The statistically non-significant improvement of visual acuity was not maintained at 24 months (declined statistically significantly) (p= 0.002), and the mean BCVA in logMAR units was 0.69 ± 0.35, which was a non-significant decline compared to the baseline (p= 0.290). (Figure 3, 4.1, 4.2, 4.3)
Figure 3: GROUP A- Comparison between mean BCVA at baseline, at 4 monthspost loading dose therapy and at 24 months following PRN basis therapy
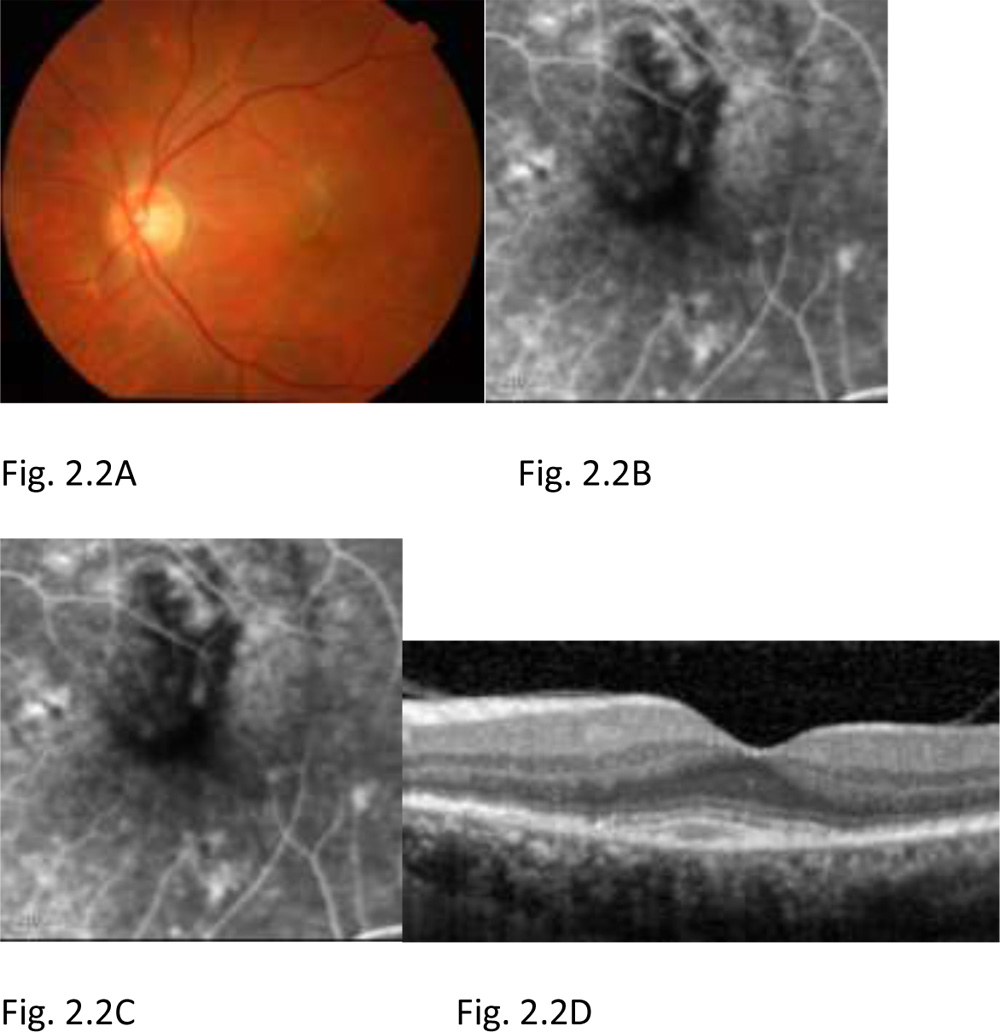
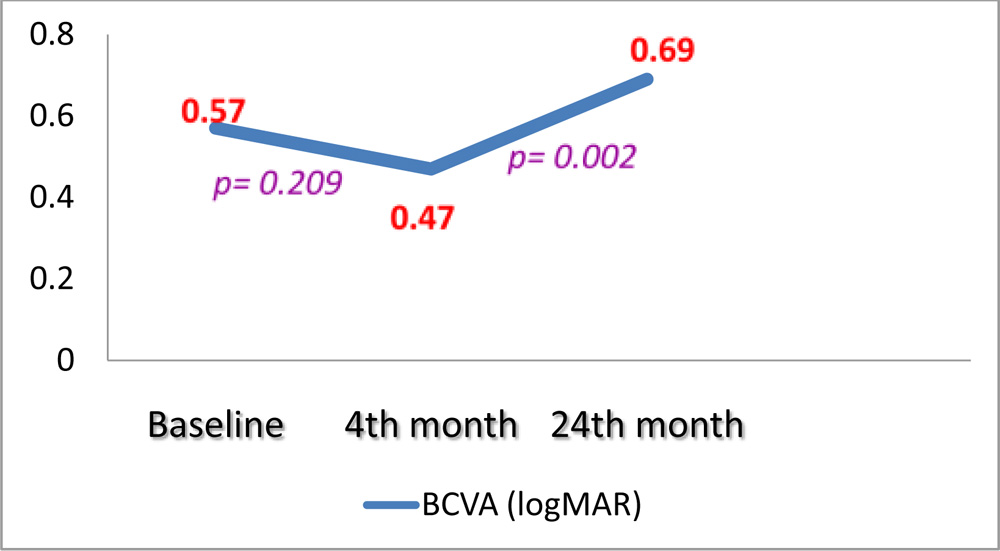 Figure 4.1: Fundus photograph OD shows greyish elevation of tissue deep to retina at macula suggestive of CNV in AMD (A). OCT of OD shows the subretinalneovascular membrane at fovea (B). The visual acuity at presentation was 6/24
Figure 4.1: Fundus photograph OD shows greyish elevation of tissue deep to retina at macula suggestive of CNV in AMD (A). OCT of OD shows the subretinalneovascular membrane at fovea (B). The visual acuity at presentation was 6/24
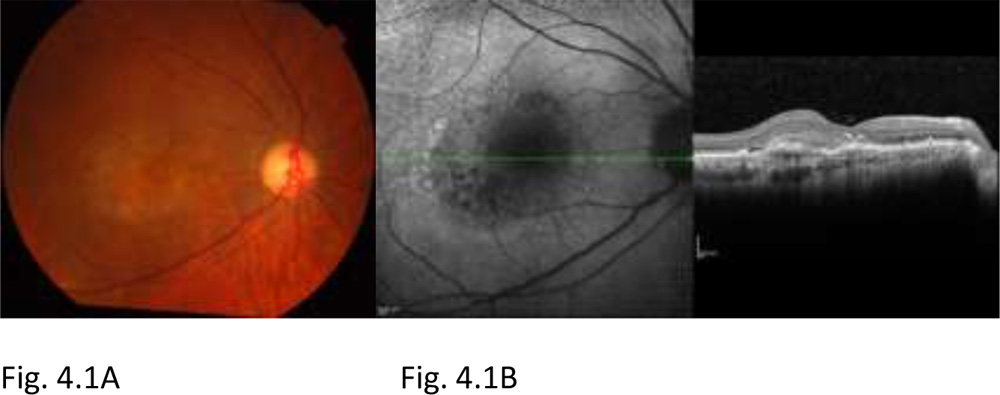 Figure 4.2: Post 3-monthly loading dose Anti-VEGF therapy, the patient developed a recurrence. Fundus photograph OD shows greyish elevation of tissue deep to retina at macula suggestive of CNV in AMD, with subretinal blood visible temporal to it (A). OCT of OD shows the scarred subretinalneovascular membrane at fovea, larger in size as compared to baseline OCT (B). FA + ICG of OD shows subretinal blocked fluorescence due to subretinal blood alongside leakage visible from recurrent scarred CNV lesion. The visual acuity at recurrence was 6/60. The patient was treated on PRN basis for recurrences.
Figure 4.2: Post 3-monthly loading dose Anti-VEGF therapy, the patient developed a recurrence. Fundus photograph OD shows greyish elevation of tissue deep to retina at macula suggestive of CNV in AMD, with subretinal blood visible temporal to it (A). OCT of OD shows the scarred subretinalneovascular membrane at fovea, larger in size as compared to baseline OCT (B). FA + ICG of OD shows subretinal blocked fluorescence due to subretinal blood alongside leakage visible from recurrent scarred CNV lesion. The visual acuity at recurrence was 6/60. The patient was treated on PRN basis for recurrences.
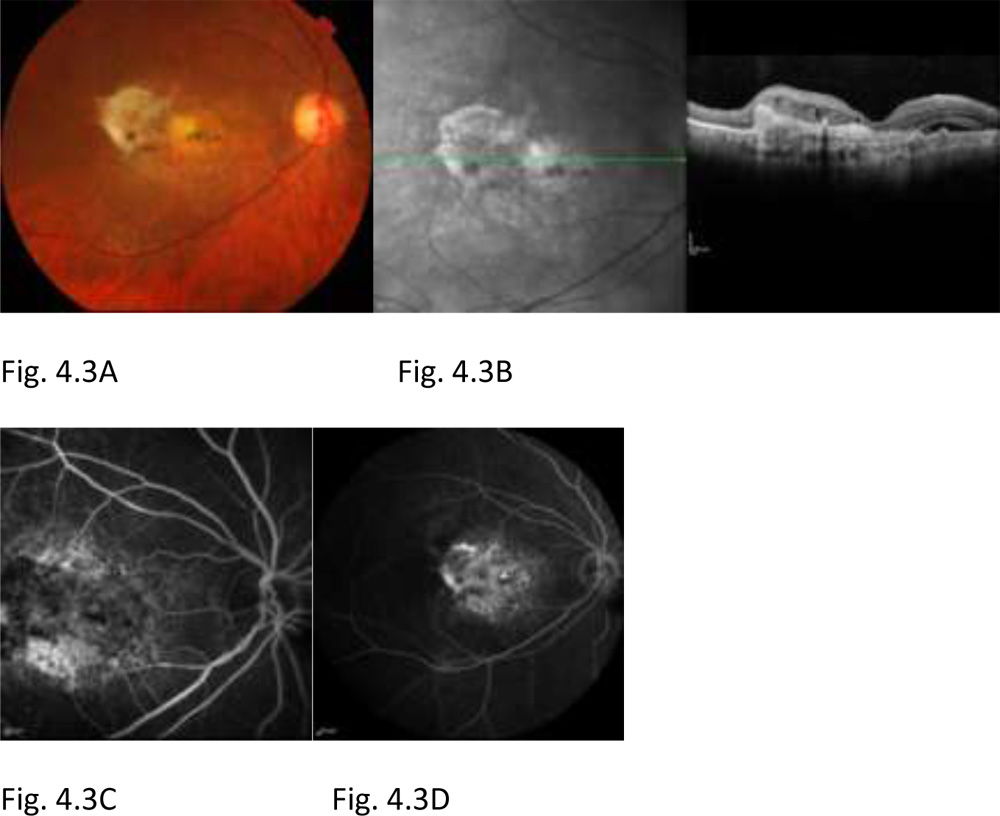
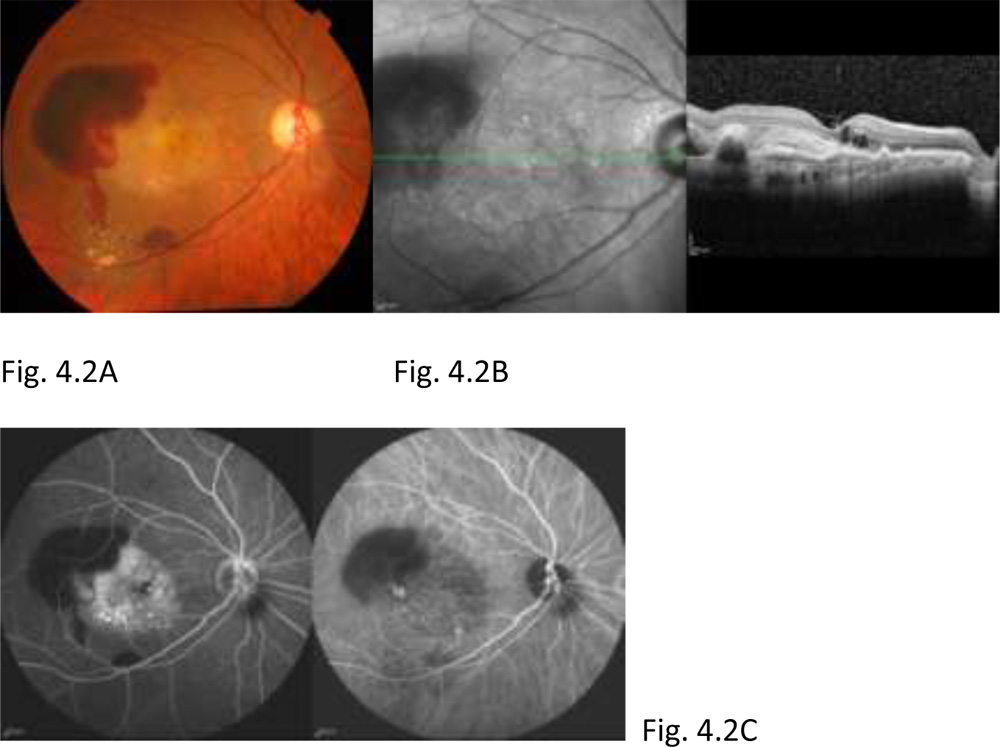 Figure 4.3: Post PRN basis Anti-VEGF therapy for recurrences until 24 months. Fundus photograph OD shows yellow-white scar tissue at macula and temporal to it (A). OCT of OD shows the subretinal scar at macula (B). FA of OD in early phase (C) shows window defects at macula and in late phase (D) shows staining of scar tissue at macula. The visual acuity at 24 months was finger counting at 4 meter despite PRN basis Anti-VEGF therapy
Figure 4.3: Post PRN basis Anti-VEGF therapy for recurrences until 24 months. Fundus photograph OD shows yellow-white scar tissue at macula and temporal to it (A). OCT of OD shows the subretinal scar at macula (B). FA of OD in early phase (C) shows window defects at macula and in late phase (D) shows staining of scar tissue at macula. The visual acuity at 24 months was finger counting at 4 meter despite PRN basis Anti-VEGF therapy
 Post 3-monthly loading dose therapy,for the 22 eyes with no further recurrences (Group B) the mean BCVA in logMAR units improved statistically non-significantly from baseline BCVA 0.91 ± 0.33 to 0.81 ± 0.42 at 4 months (p =0.248). The statistically non-significant improvement of visual acuity was maintained at 24 months (declined statistically non-significantly) (p= 0.206), and the mean BCVA in logMAR units was 0.85 ± 0.43, which was better than the baseline mean BCVA. (Figure 5, 6.1, 6.2)
Post 3-monthly loading dose therapy,for the 22 eyes with no further recurrences (Group B) the mean BCVA in logMAR units improved statistically non-significantly from baseline BCVA 0.91 ± 0.33 to 0.81 ± 0.42 at 4 months (p =0.248). The statistically non-significant improvement of visual acuity was maintained at 24 months (declined statistically non-significantly) (p= 0.206), and the mean BCVA in logMAR units was 0.85 ± 0.43, which was better than the baseline mean BCVA. (Figure 5, 6.1, 6.2)
Figure 5: GROUP B- Comparison between mean BCVA at baseline, at 4 monthspost loading dose therapy and at 24 months following PRN basis therapy
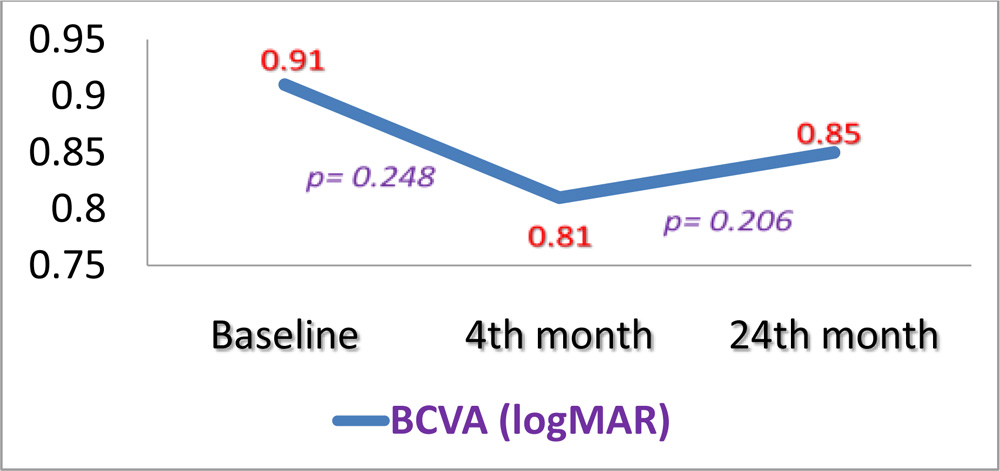 Figure 6.1:Fundus photograph OD shows greyish elevation of tissue deep to retina at macula suggestive of CNV in AMD (A). OCT of OD shows the subretinalneovascular membrane at fovea, subretinal fluid and intraretinal fluid filled cystoid spaces (B). FA of OD shows a discrete, well-demarcated focal area of hyperfluorescence at macula (arrow) in early phase frame (C), with hyperfluorescence increasing in intensity and extending beyond boundaries of hyperfluorescence identified in early phase in later phase frame (D) suggestive of a classic CNV. The visual acuity at presentation was finger counting at 3 meter.
Figure 6.1:Fundus photograph OD shows greyish elevation of tissue deep to retina at macula suggestive of CNV in AMD (A). OCT of OD shows the subretinalneovascular membrane at fovea, subretinal fluid and intraretinal fluid filled cystoid spaces (B). FA of OD shows a discrete, well-demarcated focal area of hyperfluorescence at macula (arrow) in early phase frame (C), with hyperfluorescence increasing in intensity and extending beyond boundaries of hyperfluorescence identified in early phase in later phase frame (D) suggestive of a classic CNV. The visual acuity at presentation was finger counting at 3 meter.
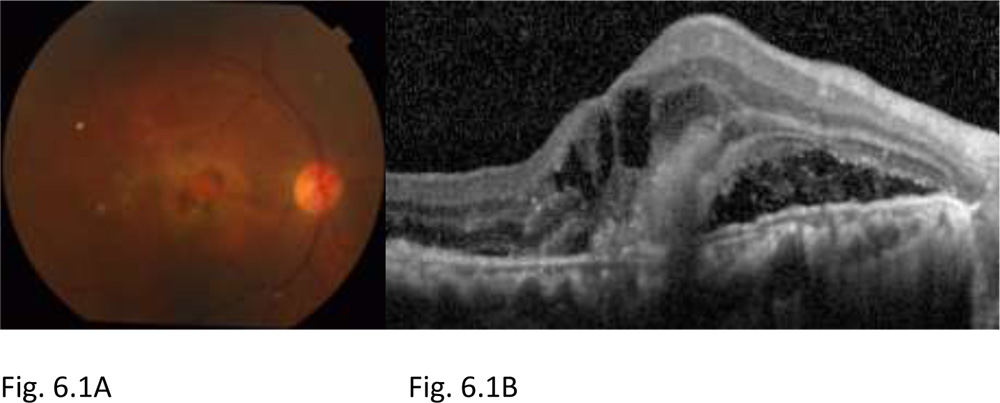
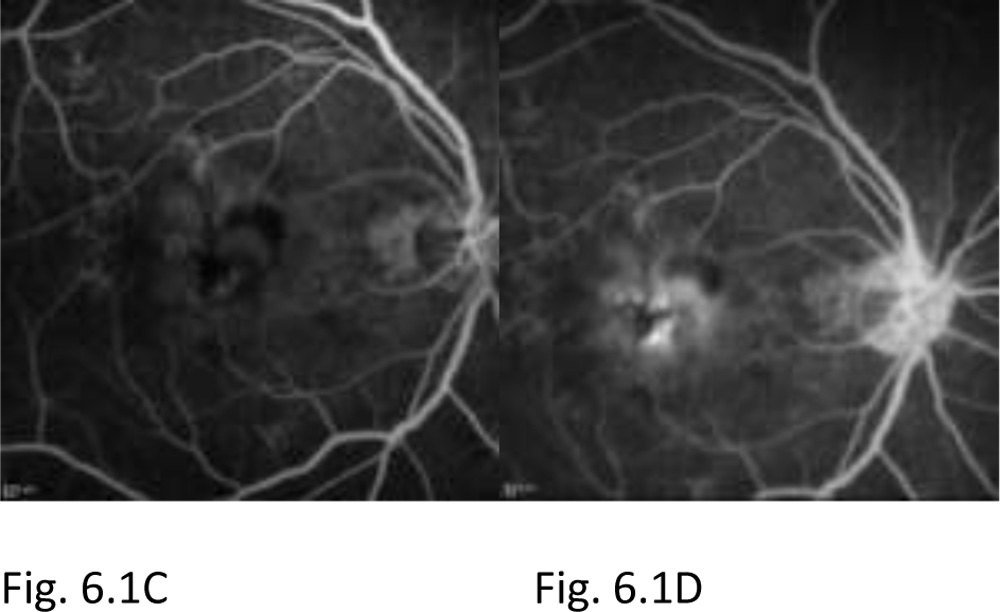
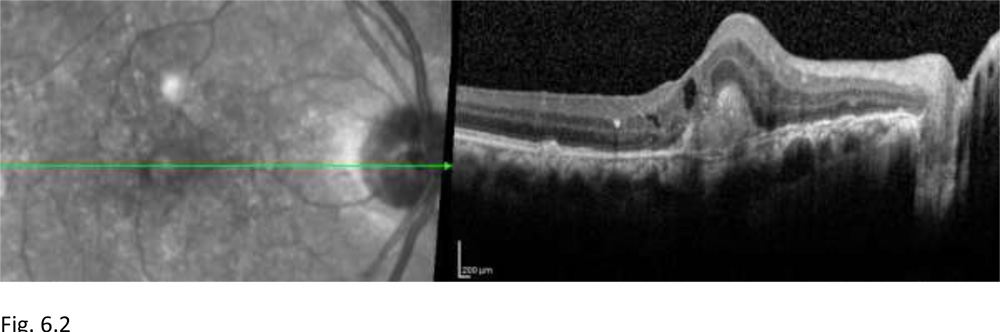
Figure 6.2: Post 3-monthly loading dose Anti-VEGF therapy at 24 months. The patient did not develop any recurrences following the loading dose therapy, thus not requiring any further Anti-VEGF therapy. OCT of OD shows reduction in the subretinalneovascular membrane, disappearance of the subretinal and intraretinal fluid pockets, residual subretinal scar at fovea can be seen.
Thus, across all 39 eyes, mean VA improved at 4 monthsfollowing loading dose therapy (although non-significantly)
Further, comparing group A and group B post 3-monthly loading dose Anti-VEGF therapy (Figure 7)
- GROUP A (eyes with PRN therapy for recurrences)
The improved mean VA at 4 months declined significantly at 24 months.
- GROUP B (no recurrences post loading dose therapy)
The improved mean VA at 4 months was maintained until 24 months.
Figure 7: Comparing mean BCVA changes in group A and group B at baseline, 4 and 24 months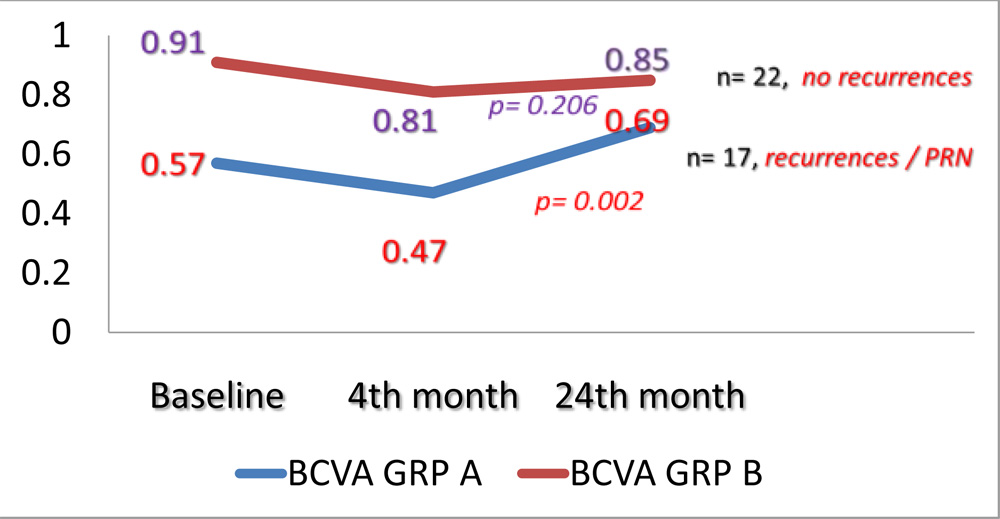 DISCUSSION:
DISCUSSION:
Heavy financial burden and compliance issues in routine clinical practice have led to dosing regimens that are less intensive than those used in clinical trials. We aimed to determine visual outcome of treatment with 3-monthly loading dose followed by PRN Anti-VEGF therapy in Wet AMD, which is usually the regimen followed in wider community in everyday practice.
After 3- monthly loading dose Anti-VEGF therapy, 22 out of 39 eyes did not develop any further recurrences requiring re-treatments; whereas 17 eyes developed recurrences which were treated on PRN basis with Anti-VEGF therapy.Across all 39 eyes, the mean baseline BCVA improved non-significantly at 4 months post 3-monthly loading dose therapy. This initial improvement in mean BCVA was maintained until 24 months in eyes with no further recurrences; whereas it was not maintained in eyes requiring re-treatments for recurrences, despite the PRN basis Anti-VEGF therapy.
Comparison of AMD treatments trial (CATT) was a two-year investigation that enrolled and randomized 1107 patients with CNV into four different treatment groups: Ranibizumab or Bevacizumab in a monthly or an as-needed regimen.5-6 Similar to the CATT study, in our study most of the change in mean visual acuity occurred during the first year, with relative little change during the second year, rather in our study the mean visual acuity declined to baseline level or worse at the end of 24 months follow up. We found that 3-monthly loading dose followed by PRN Anti-VEGF therapy for recurrences could not stabilise vision until 24 months in eyes with recurrences, despite the PRN therapy; although initial vision gained at 4 months was maintained in eyes with no recurrences until 24 months.
One of the potential reasons for less optimal visual outcome in our study compared with clinical trials is the lower mean number of injections (4.59 in 2 years). Such discrepancy in visual outcome of Anti-VEGF treatment in routine clinical practice was attributed to poorer compliance to treatment protocol, longer time intervals between follow-up visits, less number of follow-up OCTs performed and fewer reinjections. 8,11-16 The Swedish Lucentis Quality Registry found a good improvement in visual acuity after 3 injections of Ranibizumab, but this subsequently dropped back to pretreatment levels.14 Similar results were found by the WAVE study in Germany and in the French Lumiere study.8,11,15 Besides the number of injections, case selection, OCT parameters in monitoring disease activity and treatment regimens also contributed to the differences in visual outcomes in observational studies compared with clinical trials.
Thus we conclude that as eyes which will develop recurrences following 3-monthly loading dose Anti-VEGF therapy cannot be predicted, hence a more intensive dosing regimen like TAE dosing after loading dose therapy, as suggested by Spaide17,18 can be better in stabilising long term visual acuity.
Our study was limited by small sample size and being a retrospective study. The study retrospectively evaluated patients who returned for follow-up appointments, but patients who did poorly or exceptionally well may have defaulted follow-up visits.
The significance of data from retrospective studies is that they provide an indication of what is happening in routine clinical practice, in contrast to results of tightly controlled clinical trials, which may or may not be achievable in routine practice. Our study can be useful in providing patients’ expectations for visual stabilization or improvement after receiving less-than-monthly anti-VEGF, which may be a more preferable treatment regimen especially when cost and compliance is a concern. More frequent follow-up visits and may optimize efficacy in clinical practice. Thus, a dosing regimen like theTAE can be a good compromise between the monthly, bi-monthly dosing and the PRN dosing in everyday practice.
REFERENCES
- Rosenfeld PJ, Moshfeghi AA, Puliafito CA. Optical coherence tomography findings after an intravitreal injection of bevacizumab (avastin) for neovascular age-related macular degeneration. Ophthalmic Surg Lasers Imaging 2005;36:331–5.
- Brown DM, Kaiser PK, Michels M, et al. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med 2006;355:1432–44.
- Rosenfeld PJ, Brown DM, Heier JS, et al. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 2006;355:1419–31.
- Heier JS, Brown DM, Chong V, et al. Intravitrealaflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology 2012;119:2537–48.
- CATT Research Group, Martin DF, Maguire MG, Ying GS, Grunwald JE, Fine SL, Jaffe GJ. Ranibizumab and bevacizumab for neovascular age-related macular degeneration. N Engl J Med 2011;364 (20): 1897-1908.
- Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group, Martin DF, Maguire MG, Fine SL, Ying GS, Jaffe GJ, Grunwald JE, Toth C, Redford M, Ferris FL 3rd. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology 2012;119(7):1388-1398.
- Gupta OP, Shienbaum G, Patel AH, Fecarotta C, Kaiser RS, Regillo CD. A treat and extend regimen using ranibizumab for neovascular age-related macular degeneration clinical and economic impact. Ophthalmology 2010; 117(11):2134-2140.
- Finger RP, Wiedemann P, Blumhagen F, et al. Treatment patterns, visual acuity and quality-of-life outcomes of the WAVE study—a noninterventional study of ranibizumab treatment for neovascular age-related macular degeneration in Germany. ActaOphthalmol (Copenh) 2013;91:540–6.
- Thompson AC, Thompson MO, Young DL, et al. Barriers to follow-up and strategies to improve adherence to appointments for care of chronic eye diseases. Invest Ophthalmol Vis Sci 2015;56:4324–31.
- Wolf A, Kampik A. Efficacy of treatment with ranibizumab in patients with wet age-related macular degeneration in routine clinical care: data from the COMPASS health services research. Graefes Arch ClinExpOphthalmol Albrecht Von Graefes Arch FürKlinExpOphthalmol 2014;252:647–55.
- Cohen SY, Dubois L, Tadayoni R, Fajnkuchen F, Nghiem-Buffet S, Delahaye-Mazza C, Guiberteau B, Quentel G. Results of one-year’s treatment with ranibizumab for exudative age-related macular degeneration in a clinical setting. Am J Ophthalmol 2009;148(3):409-413.
- Michalova K, Wickremasinghe SS, Tan TH, Chang A, Harper CA, Downie JA, Hunyor AP, Guymer RH. Ranibizumab treatment for neovascular age-related macular degeneration: from randomized trials to clinical practice. Eye (Lond) 2009;23(8):1633-1640.
- Bandukwala T, Muni RH, Schwartz C, Eng KT, Kertes PJ. Effectiveness of intravitrealranibizumab for the treatment of neovascular age-related macular degeneration in a Canadian retina practice: a retrospective review. Can J Ophthalmol 2010;45(6):590-595.
- Hjelmqvist L, Lindberg C, Kanulf P, Dahlgren H, Johansson I, Siewert A. One-year outcomes using ranibizumab for neovascular age-related macular degeneration: results of a prospective and retrospective observational multicentre study. J Ophthalmol 2011;2011:405724.
- Heimes B, Lommatzsch A, Zeimer M, Gutfleisch M, Spital G, Dietzel M, Pauleikhoff D. Long-term visual course after anti-VEGF therapy for exudative AMD in clinical practice evaluation of the German reinjection scheme. Graefes Arch ClinExpOphthalmol 2011;249(5):639-644.
- Gillies MC, Walton R, Simpson JM, Arnold JJ, Guymer RH, McAllister IL, Hunyor AP, Essex RW, Morlet N, Barthelmes D; Fight Retinal Blindness! Project Investigators. Prospective audit of exudative age-related macular degeneration: 12-month outcomes in treatment-naive eyes. Invest Ophthalmol Vis Sci 2013;54(8):5754-5760.
- Spaide R (2007) Ranibizumab according to need: a treatment for age-related macular degeneration. Am J Ophthalmol 143: 679-680.
- Spaide RF (2009) The as-needed treatment strategy for choroidal neovascularization: a feedback-based treatment system. Am J Ophthalmol 148: 1-3.

