Dr. Harshavardhan
V K, Dr. Sumita Mohapatra, Dr. Parul Priyambada,
Dr. Nilamadhab Pradhan
PRESENTING AUTHOR: DR. HARSHAVARDHAN V K
CO-AUTHORS: PROF (DR) SUMITA MOHAPATRA, DR. PARUL PRIYAMBADA, DR. NILAMADHAB PRADHAN
Introduction:
Alzheimer’s disease, a neurodegenerative disorder is the most common cause of dementia in the elderly. Approximately 10% of all patients over the age of 70 have significant memory loss, and in more than half the cause is Alzheimer’s disease. It presents with an insidious onset of memory loss followed by a slowly progressive dementia over several years. The cognitive changes in AD follow a characteristic pattern beginning with memory loss and spreading to language and visuospatial deficit. In mild cognitive impairment (MCI) memory loss falls 1.5 standard deviation below normal on standardized memory tests. MCI is now considered as the prodromal stage of AD. The pathogenesis of neurodegeneration revolves around formation of extracellular senile plaques due to amyloid beta deposition (Aβ) and formation of intracellular neurofibrillary tangles (NFTs) due to hyperphosphorylation of tau proteins. The neurodegeneration occurs mostly in medial temporal lobe, lateral temporal cortex and nucleus basalis. Visual disturbances in AD have been well documented and attributed to neurodegeneration in the primary and associative visual cortex.
Recent studies have shown that neurodegeneration involves retinal ganglion cells and their axons in optic nerve. Also, histopathological lesions associated with AD like neuronal loss, beta-amyloid proteins, neurofibrillary tangles, and granulovacuolar degenerations have not only been found in the brain but also in the neuroretina.
Alzheimer’s disease is characterized by its slow progression, where neurodegeneration starts several years before full blown clinical manifestations. This provides a window period for early diagnosis during the symptom less period and during the prodromal MCI. However until recently the diagnosis of Alzheimer’s has been only possible after symptomatic onset and it has been largely a diagnosis of exclusion. Early diagnosis of Alzheimer’s has been a challenge.
So far the diagnosis of Alzheimer’s has been on basis of symptoms, neuroimaging studies, CSF evaluation. Neuroimaging studies like CT and MRI show no specific changes especially in the early disease. Functional imaging studies show hypoperfusion or hypometabolism in the posterior tempero-parietal lobes. The EEG in AD is normal and shows no specific slowing. CSF examination is invasive and shows a reduced level of Aβ and increased levels of hyperphosphorylated tau proteins. However there is considerable overlap of these values with that of normal population, and hence CSF examination too is a non specific diagnostic tool. A recent tool is blood apo E genotyping, apo E4 allele of apo E gene on chromosome 19 confers increased risk for AD. Despite these diagnostic modalities, only a high probability of AD can be established.
Recently, eye has been shown to provide a window of opportunity for early diagnosis. The retina being in continuation with the central nervous system both anatomically and embryologically shows neurodegenerative changes in the ganglion cells and nerve fiber layer which if detected can provide a diagnostic modality for Alzheimer’s disease.
In the past two decades, several studies have searched for in vivo evidence of retinal involvement in AD. Optical coherence tomography (OCT), is one such sophisticated non invasive imaging study which can assess morphological changes in retina in AD. The OCT technique for the measurement of peripapillary RNFL has been proven useful for the detection of significant reduced retinal thickness in patients of AD.
Aim of study:
To evaluate retinal nerve fiber layer (RNFL) thickness in Alzheimer’s disease using Spectralis optical coherence tomography (OCT).
Materials and methods:
This is a prospective study which included right eyes of 30 diagnosed cases of AD, meeting the inclusion criteria in the age group of 67-83 years, in the time period of April 2015- March 2016. The diagnosis of AD was made on basis of MRI findings and mini-mental state examination (MMSE). The control group consisted of right eyes of 30 patients visiting the outdoor for routine eye examination, mainly for refractive errors.
Inclusion criteria:
- Patients diagnosed with Alzheimer’s disease by MRI findings and MMSE.
- Best corrected visual acuity of 20/40 or better.
Exclusion criteria:
- Patients with other known disease affecting the optic nerve like glaucoma, AION, MS.
- Patients with unclear media like those with dense cataract.
- Patients with optic disc anomalies.
- Patients with history of IOP elevations.
Both cases and controls underwent detailed ocular examination, including measurement of best corrected visual acuity, slit lamp examination, IOP measurement by applanation tonometry, central corneal thickness and fundoscopy.
All subjects underwent OCT scanning using commercially available equipment (Spectralis OCT, Heidelberg Engineering) and an ophthalmic evaluation on the
same day. The peripapillary RNFL thickness of all subjects were measured. Only those images with good quality were included.
Results:
The age difference between two groups was not statistically significant (p=0.74). The average RNFL thickness in Alzheimer’s disease patients was found to be 111.9±8.25 µm while it was 117.97±9.29 in control group. Using unpaired, Student’s t-test, the average RNFL thickness in AD patients was found to be significantly less than in controls (p<0.01).
Figure 1 : Graph showing age distribution in both groups
Figure 2: Avg. RNFL Thickness (µm) in patients with AD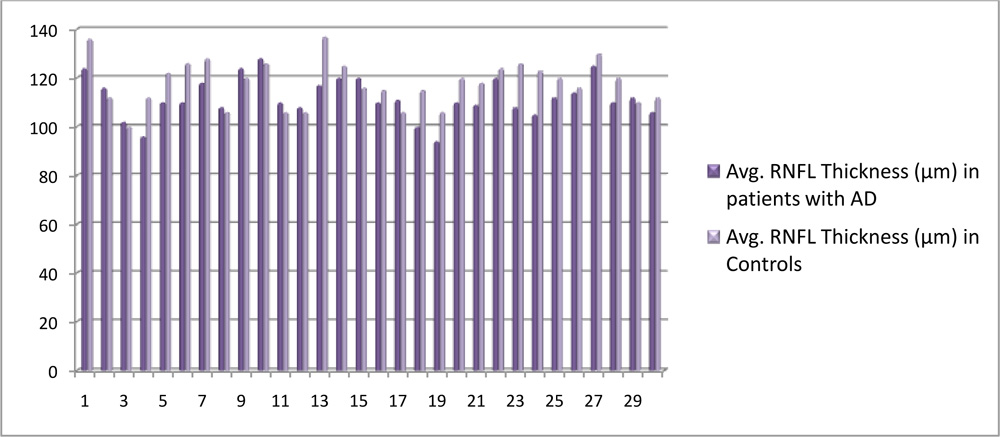
Figure 3: Comparison of average RNFL thickness in two groups
Discussion:
RNFL thickness in a group of 30 Alzheimer’s disease patients’ right eye was evaluated using Spectralis OCT (Heidelberg Engineering). A control group consisting of 30 healthy individuals was taken and evaluated for the same. The age difference between the two groups was not statistically significant. The average RNFL thickness of the two groups were compared. The average RNFL thickness in Alzheimer’s disease patients (111.9 ± 8.25 µm) was found to be significantly less than that of the control group (117.97 ± 9.29 µm) (p<0.01). It was hence concluded that RNFL thickness is reduced in patients of Alzheimer’s disease as compared to healthy individuals.
Conclusion:
The average RNFL thickness was found to be significantly less in patients of AD compared to controls and hence RNFL measurement by OCT can be used as an early and important tool to aid the diagnosis of AD. Additionally, RNFL thickness changes can be used to monitor progression of AD.
References:
1.Kanamori, A., Michael F.T. Escano, Eno, A., Nakamura, M., Maeda, H., Seya, R., . . . Negi, A. (2003). Evaluation of the effect of aging on retinal nerve fiber layer thickness measured by optical coherence tomography.Ophthalmologica,217(4), 273-8.
2.Cunha LP, Lopes LC, Costa-Cunha LVF, Costa CF, Pires LA, Almeida ALM, et al. (2016) Macular Thickness Measurements with Frequency Domain-OCT for Quantification of Retinal Neural Loss and its Correlation with Cognitive Impairment in Alzheimerʼs Disease. PLoS ONE 11(4): e0153830.doi:10.1371/journal.pone.0153830
3.Seeley W, Miller BL.Alzheimer’s disease and other dementias. In: Kasper DL, Braunwald E, Fauci AS, et al., eds. Harrison’s Principles of Internal Medicine. 19th ed. ed. New York: McGraw-Hill; April 2015.
4.Oktem, E. O., Derle, E., Kibaroglu, S., Oktem, C., Akkoyun, I., & Can, U. (2015). The relationship between the degree of cognitive impairment and retinal nerve fiber layer thickness.Neurological Sciences, 36(7), 1141-1146. doi:http://dx.doi.org/10.1007/s10072-014-2055-3
5.Dehabadi, M. H., Davis, B. M., Wong, T. K., & Cordeiro, M. F. (2014). Retinal manifestations of alzheimer’s disease.Neurodegenerative Disease Management, 4(3), 241-252. doi:http://dx.doi.org/10.2217/nmt.14.19
6.Coppola, G., Renzo, A. D., Ziccardi, L., Martelli, F., Fadda, A., Manni, G., . . . Parisi, V. (2015). Optical coherence tomography in alzheimer’s disease: A meta-analysis.PLoS One, 10(8) doi:http://dx.doi.org/10.1371/journal.pone.0134750
7.Salobrar-Garcia, E., Hoyas, I., Leal, M., de Hoz, R., Rojas, B., Ramirez, A. I., . . . Ramirez, J. M. (2015). Analysis of retinal peripapillary segmentation in early alzheimer’s disease patients.BioMed Research International,doi:http://dx.doi.org/10.1155/2015/636548
8.Jones-odeh, E., & Hammond, C. J. (2015). How strong is the relationship between glaucoma, the retinal nerve fibre layer, and neurodegenerative diseases such as alzheimer’s disease and multiple sclerosis?Eye, 29(10), 1270-1284. doi:http://dx.doi.org/10.1038/eye.2015.158
9.Kesler, A., Vakhapova, V., Korczyn, A. D., Naftaliev, E., & Neudorfer, M. (2011). Retinal thickness in patients with mild cognitive impairment and alzheimer’s disease.Clinical Neurology and Neurosurgery, 113(7),5236.doi:http://dx.doi.org/10.1016/j.clineuro.2011.02.014

