![]()
Col . RANGACHARI AWARD
Dr. Natasha Pahuja, P16722, Dr. Rohit Shetty, Mr. Anupam Sharma, Dr. Arkasubhra Ghosh
Introduction:
Introduction: Oxidative stress has been associated with several human diseases including KC1,2 Environmentally induced oxidative stress in the stroma of KC patients causes an increase in Reactive Oxygen Species (ROS) and decrease in levels of antioxidants at a cellular level which results in degradation of the extracellular matrix and subsequent thinning of the corneal stroma1,3.Autophagy is an evolutionarily conserved mechanism which causes the degradation or clearance of long lived and misfolded proteins and damaged cellular organelles4.The failure of autophagy induction or autophagy flux (dynamics) due to oxidative stress can disturb the adaptive response and lead to autophagy mediated cell death5,6.Although, genetic and environmental factors are involved in the pathogenesis of KC, the underlying molecular mechanism of progressive corneal ectasia remains unknown. Therefore, we investigated the regulation of autophagic lysosomal pathway in the pathogenesis of KC with respect to oxidatiaaaaaaaaaaave damage in patients’ corneal epithelial tissue and in-vitro experimental conditions using human corneal epithelial (HCE) cells under chronic hyperoxic stress.
Purpose: To study inflammatory cytokines and autophagy related markers. In addition to determine whether autophagy deregulation is only restricted to the diseased or ‘cone’ area in keratoconus
Methods: Clinical examination included slit lamp examination with topographic and pachymetric evaluation on the Pentacam HR (Oculus, Germany) and the Orbscan (Orbtek, Bausch&Lomb). Clinical grades of KC were determined as per the Amsler-Krumeich classification. Total epithelium from 8- 9 mm of the cornea was debrided from the cases (keratoconus) undergoing corneal collagen cross linking (CXL) or topo-guided photo refractive keratectomy (T-PRK) followed by CXL and controls, undergoing photo refractive keratectomy (PRK) for myopia or myopic astigmatism. Gene expression from epithelium of KC (n=60) and normal cornea (n=18) from cone (ectatic) and peripheral (non ectatic) regions was studied. Differential expression of autophagy related biomarkers (LC3, LAMP1) in the cone and periphery epithelium of KC. Total ribonucleic acid (RNA) extraction was done for cells and tissues using the RNeasy Mini Kit (Qiagen) according to the manufacturer’s protocol, and quantified using a nanodrop spectrophotometer (NanoDrop 1000, Thermo Scientific). 1ug of total RNA were converted to cDNA using the Biorad iScript™ cDNA Synthesis Kit.Quantitative real-time PCR was performed using 4 μl of 10-fold diluted cDNA in a final volume of 10 μl using the SYBR Green master mix 2X (Biorad) according to the manufacturer’s instruction.
Results: The expression of autophagy signaling pathway related genes LC3A, LC3B, ATG5, ATG7, RAB7 and LAMP1in clinical grades (I to III) of KC patients’ epithelium along with the inflammatory mediators, collagen and lysyl oxidase (LOX) . The mRNA transcript and protein level expressions of the cone (ectatic) and peripheral (non-ectatic) regions in KC and control (PRK) corneal epithelium were compared and are shown in figure below.
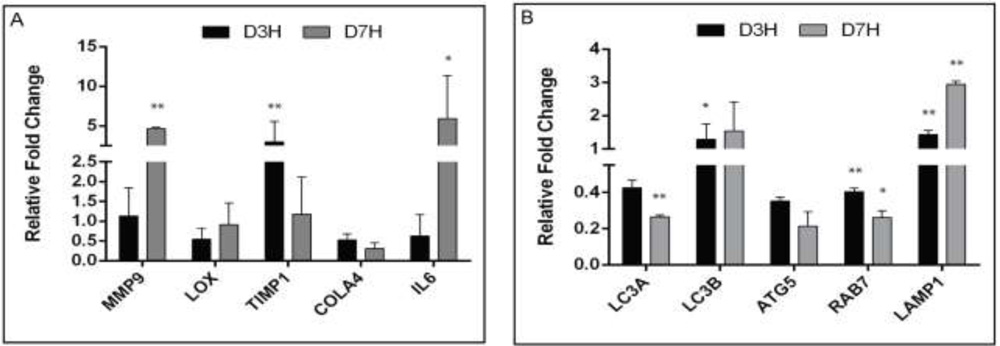
Figure: mRNA levels of autophagy markers in cone vs periphery from epithelium (keratoconus)
Conclusion: The results suggest an oxidative stress associated defective autophagy, as one of the pathogenic mechanisms in KC. Use of topical medications (that are currently available in the market for other indications) that induce autophagy can be an effective alternative strategy in the management of KC.
References:
1.Kenney, M. C. et al. Increased levels of catalase and cathepsin V/L2 but decreased TIMP-1 inkeratoconus corneas: evidence that oxidative stress plays a role in this disorder. Investigative ophthalmology & visual science46, 823-832, (2005).
2.Chwa, M. et al. Hypersensitive response to oxidative stress in keratoconus corneal fibroblasts. Investigative ophthalmology & visual science49, 4361-4369, (2008)
3.Arnal, E., Peris-Martinez, C., Menezo, J. L., Johnsen-Soriano, S. & Romero, F. J. Oxidative stress in keratoconus? Investigative ophthalmology & visual science52, 8592-8597, (2011).
4.Marino, G., Madeo, F. & Kroemer, G. Autophagy for tissue homeostasis and neuroprotection. Current opinion in cell biology23, 198-206, (2011)
5.Onodera, J. & Ohsumi, Y. Autophagy is required for maintenance of amino acid levels and protein synthesis under nitrogen starvation. The Journal of biological chemistry280, 31582-31586, (2005).
6.Kuma, A. et al. The role of autophagy during the early neonatal starvation period. Nature432, 1032-1036, (2004)


 FP1591 : Title: Novel Altered Critical Intracellular Signaling Events in Keratoconus & It’s Therapeutic Potential
FP1591 : Title: Novel Altered Critical Intracellular Signaling Events in Keratoconus & It’s Therapeutic Potential