![]()
AIOS – Om Prakash Award
Dr. Bhavna Chawla, C08835, Dr. Fahmi Hasan, Dr. Shailesh Gaikwad, Dr. Rachna Seth
Bhavna Chawla,1 Fahmi Hasan,1 Shailesh Gaikwad,2 Rachna Seth 3
1Dr Rajendra Prasad Centre for Ophthalmic Sciences
2Department of Interventional Neuroradiology
3Paediatric Oncology Division
All India Institute of Medical Sciences
New Delhi
Chief and Presenting Author
Dr Bhavna Chawla, MS
Additional Professor
Dr Rajendra Prasad Centre for Ophthalmic Sciences
All India Institute of Medical Sciences
New Delhi
Email: bhavna2424@hotmail.com
Introduction
Retinoblastoma (RB) is the most common intra-ocular malignancy seen in children. The incidence is approximately i1 in 18000 live births worldwide. The average age of presentation is 24 months in unilateral cases and 12 months in bilateral cases. One third of affected children have bilateral disease. The management of RB often presents as a challenge to the ophthalmologist. Depending on the stage of disease at diagnosis, the goals of therapy are life salvage, globe salvage and preservation of functional vision. For over a decade, systemic chemotherapy combined with focal therapy has been the mainstay of globe-preserving treatment for less advanced disease. Although this treatment approach has resulted in good outcomes, systemic chemotherapy can lead to serious toxic side effects including myelosuppression and infection. As a result, newer treatment approaches have focused on localized delivery of chemotherapy to minimize the systemic side effects of chemotherapy. One such novel approach is selective intra-arterial chemotherapy (SIAC), which delivers targeted chemotherapy to the eye harbouring the tumour via the ophthalmic artery. 1-6 The aim of this study was to evaluate the efficacy & safety of SIAC in intraocular RB.
Materials and Methods
The study was a prospective, interventional study. Prior approval was taken from the Ethics Committee of our Institute. Patients were recruited from the RB Clinic of our Centre after an informed consent. The inclusion criteria was cases of intraocular RB (Group B, C or D, according to the International Classification System), that were refractory to systemic chemotherapy. Cases with advanced intraocular Group E RB and extra-ocular disease were excluded. Demographic details such as age, gender, laterality, family history, and prior treatment history were noted. Baseline ocular examination was done under anaesthesia which included a detailed anterior segment and fundus examination. Findings noted included the size, number and location of tumours, and the presence of vitreous and/or sub-retinal seeding. RetCam documentation and B-scan ultrasonography was done. General physical and systemic examinations were carried out, and appropriate investigations were performed.
Procedure of intra-arterial chemotherapy: The SIAC procedure was carried out in the department of Interventional Neuroradiology at our Institute. Before the procedure, the child underwent a complete general physical examination, systemic examination, and investigations such as haemogram, liver and kidney function test, prothrombin time and coagulation profile. The procedure was done under general anaesthesia under aseptic conditions. The ophthalmic artery of the eye to be treated was selectively catheterized via the internal carotid artery under fluoroscopic visualization & chemotherapy was infused into the eye. The technique involved the passing of a catheter (Guiding catheter) via a femoral artery puncture on the intended side of treatment. A 5F sheath was inserted into the femoral artery, and the patient was anti-coagulated with intravenous heparin. A guide wire and a micro-catheter were then used to access the vascular supply to the eye, with special attention to the angioanatomy and blood flow patterns in each patient using fluoroscopy visualization. A selective angiogram was performed to verify the angioanatomy and placement of the microcatheter. After accessing the ophthalmic artery, melphalan drug (3-5 mg) was diluted in 30 ml of normal saline and injected manually in a pulsatile fashion over 30 mins to reach the ophthalmic artery. After completion of drug infusion, the catheter was removed and haemostasis of the femoral artery was achieved. The child was kept under observation for 24 hours to look for any procedure-related complications. Ophthalmic examination under anaesthesia was performed 3 weeks after the SIAC procedure. In case of a partial response to treatment, the SIAC procedure was repeated. After completion of treatment, all cases were followed up at two monthly intervals for a minimum period of 6 months. The main outcome measures were procedural success, tumour regression, and systemic and local complications. The rate of procedural success determined by successful catheterization of the ophthalmic artery. Efficacy of treatment was judged on the basis of tumour response to chemotherapy, the appearance of regression pattern, disappearance or calcification of vitreous seeds and/or sub retinal seeds and the absence of tumour recurrence on follow-up. The safety of treatment was assessed on the basis of systemic and/or ocular complications, if any.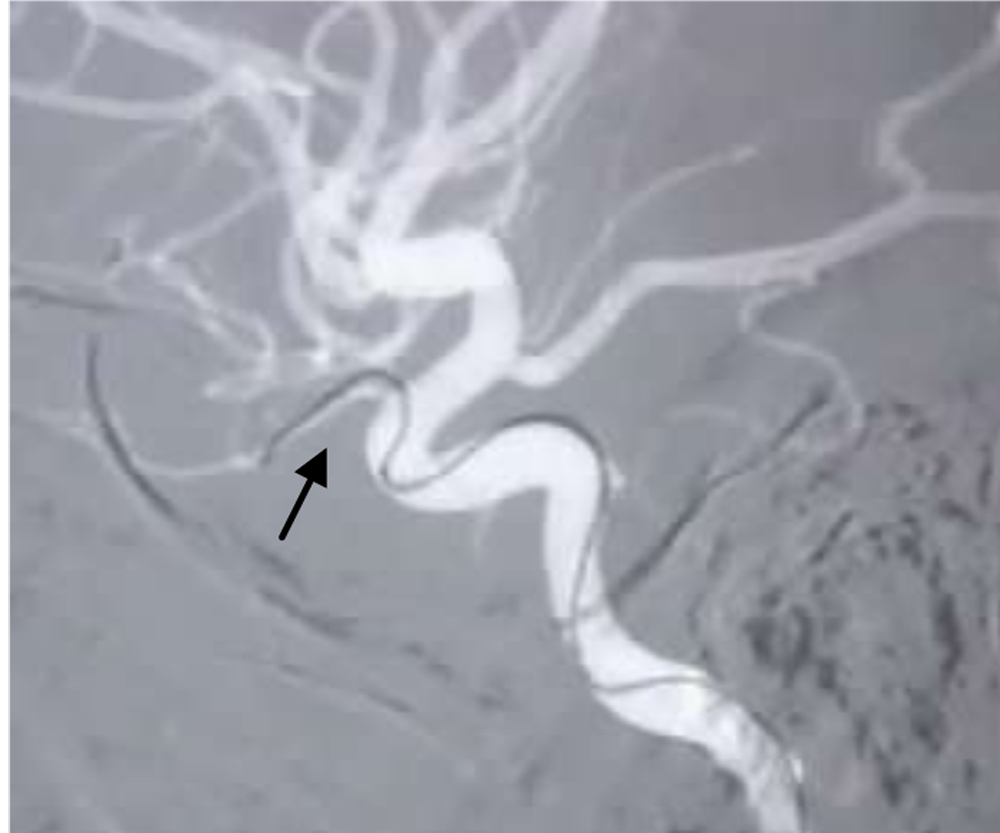
Figure 1. Selective catheterization of the ophthalmic artery (arrow) in a two year old child with retinoblastoma.
A total of 24 children (25 eyes) were recruited for the SIAC procedure during 2014-2015. These included 13 boys and 11 girls. The mean age was 3.3 years (SD±1.1). The disease was unilateral in 10 patients and bilateral in 14 patients. Eight eyes were Group B, 2 eyes were Group C, and 15 eyes were Group D RB. A total of 54 sessions of the SIAC procedure were performed. The mean number of sessions per eye was 2.2 (Range, 1-3).Ophthalmic artery catheterization was successful in 51/54 sessions (94.4%). The mean follow up was 10.8 months (SD±2.2). In two eyes (Group D ), the SIAC procedure was abandoned, hence, the efficacy of treatment was evaluated in 23 eyes. At last follow up, the tumours had regressed in 15 of the 23 eyes (65.2%). Group B eyes showed a much better tumour control rate (8/8, 100%), as compared to group C (1/2, 50%) and group D (6/13, 46.1%) eyes. No major systemic complications such as stroke, sepsis or neutropenia were noted. Local complications included patchy hair loss and skin pigmentation on the forehead, ptosis, and redness of the eye, which were transient in nature and resolved over a period of time.
Discussion
SIAC delivers targeted, high dose chemotherapy to the eye in children with RB. In 2004, the Japanese investigators Yamane & Kaneko described the technique of ‘selective ophthalmic artery infusion’ (SOAI), where a micro-balloon catheter was positioned by a trans-femoral artery approach at the cervical segment of the internal carotid artery just distal to the orifice of the ophthalmic artery.7,8 The Japanese technique of selective ophthalmic artery infusion was further developed into direct intra-arterial (Ophthalmic artery) infusion under the pioneering work of Abramson and Gobin in New York.1.2 They introduced the technique of super-selective infusion by advancing a micro-catheter into the orifice of the ophthalmic artery by a trans-femoral artery approach. Recently, studies from the West have recommended intra-arterial chemotherapy as a safe and effective treatment for advanced intra-ocular RB. SIAC has been reported to be associated with an overall success rate of 55-100% in salvaging the globe, in addition to the advantage of very low systemic toxicity. Our study shows showed an excellent rate of procedural success and satisfactory rate of globe salvage, with no significant side effects, in eyes that were otherwise destined for enucleation.
Conclusion
SIAC is effective for globe salvage in selected cases of RB after treatment failure of intravenous chemotherapy. It is a feasible and safe procedure in a multi-disciplinary setting.
References
- Abramson DH, Dunkel IJ, Brodie SE, et al. A Phase I/II Study of Direct Intra-arterial (Ophthalmic Artery) Chemotherapy with Melphalan for Intraocular Retinoblastoma: Initial Results. Ophthalmology 2008; 115: 1398-1404.
- Gobin YP, Dunkel IJ, Marr BP, et al. Intra-arterial Chemotherapy for the Management of Retinoblastoma: Four-Year Experience. Arch Ophthalmol 2011; 129: 732-737.
- Peterson EC, Elhammady MS, Quintero-Wolfe S, et al.: Selective ophthalmic artery infusion of chemotherapy for advanced intraocular retinoblastoma: initial experience with 17 tumours.J Neurosurg 2011; 114: 1603-8.
- Abramson DH, Marr BP, Dunkel IJ, et al.: Intra-arterial chemotherapy for retinoblastoma in eyes with vitreous and/or subretinal seeding: 2-year results. Br J Ophthalmol 2012; 96: 499-502.
- Muen WJ, Kingston JE, Robertson F, et al.: Efficacy and complications of super-selective intraophthalmic artery melphalan for the treatment of refractory retinoblastoma. Ophthalmology 2012; 119: 611-6.
- Shields CL, Bianciotto CG, Jabbour P, et al.: Intra-arterial chemotherapy for retinoblastoma Report No. 1, control of retinal tumours, subretinal seeds, and vitreous seeds. Arch Ophthalmol 2011; 129: 1399-406.
- Kaneko A, Suzuki S. Eye-preservation treatment of retinoblastoma with vitreous seeding. Jpn J Clin Oncol 2003; 33: 601–607.
- Suzuki S, Kaneko A. Management of intraocular retinoblastoma and ocular prognosis. Int J Clin Oncol 2004; 9: 1–6.


 FP644 : Evaluation of Selective Intra- Arterial Chemotherapy for the Treatment of Intraocular Retinoblastoma
FP644 : Evaluation of Selective Intra- Arterial Chemotherapy for the Treatment of Intraocular Retinoblastoma